Chemistry, Biology
... Paper 1 will be based on the Physics and Chemistry sections of the syllabus. Paper 2 will be based on the Physics section of the syllabus. Paper 3 will be based on the Chemistry section of the syllabus. Paper 5 will be based on the Physics and Chemistry sections of the syllabus. Science (Physics, Bi ...
... Paper 1 will be based on the Physics and Chemistry sections of the syllabus. Paper 2 will be based on the Physics section of the syllabus. Paper 3 will be based on the Chemistry section of the syllabus. Paper 5 will be based on the Physics and Chemistry sections of the syllabus. Science (Physics, Bi ...
Physics, Chemistry
... Paper 1 will be based on the Physics and Chemistry sections of the syllabus. Paper 2 will be based on the Physics section of the syllabus. Paper 3 will be based on the Chemistry section of the syllabus. Paper 5 will be based on the Physics and Chemistry sections of the syllabus. Science (Physics, Bi ...
... Paper 1 will be based on the Physics and Chemistry sections of the syllabus. Paper 2 will be based on the Physics section of the syllabus. Paper 3 will be based on the Chemistry section of the syllabus. Paper 5 will be based on the Physics and Chemistry sections of the syllabus. Science (Physics, Bi ...
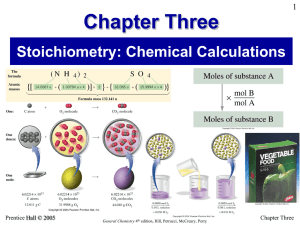
Chapter Three
... sodium, 22.67% sulfur, and 45.02% oxygen. Find the empirical formula for this compound. • HINT = Whenever you are given %s, we assume we ALWAYS have a 100-gram sample, so immediately convert you %s to grams. ...
... sodium, 22.67% sulfur, and 45.02% oxygen. Find the empirical formula for this compound. • HINT = Whenever you are given %s, we assume we ALWAYS have a 100-gram sample, so immediately convert you %s to grams. ...
The Mole Concept A. Atomic Masses and Avogadro`s Hypothesis 1
... e.g. If 1 L of nitrogen reacts with 3 L of hydrogen to form ammonia, then its formula is NH3. If 2 L of hydrogen reacts with 1 L of oxygen to form water, then its formula is H2O. So if we want to make a particular compound, all we need to do is react volumes of gases in the ratio given by their form ...
... e.g. If 1 L of nitrogen reacts with 3 L of hydrogen to form ammonia, then its formula is NH3. If 2 L of hydrogen reacts with 1 L of oxygen to form water, then its formula is H2O. So if we want to make a particular compound, all we need to do is react volumes of gases in the ratio given by their form ...
BSc Honours chemistry CBCS Syllabus 2016-17
... (ii) Estimation of oxalic acid and sodium oxalate in a given mixture. (iii) Estimation of Fe(II) with K2Cr2O7 using internal (diphenylamine, anthranilic acid) and external indicator. Reference Book ...
... (ii) Estimation of oxalic acid and sodium oxalate in a given mixture. (iii) Estimation of Fe(II) with K2Cr2O7 using internal (diphenylamine, anthranilic acid) and external indicator. Reference Book ...
laman web smk raja perempuan, ipoh
... form bonds with electron acceptors (as Lewis acids) 17. explain how nucleophiles such as OH-, NH3 , H2O, Br- , Iand carbanion have Lewis base properties, whereas electrophiles such as H+, NO2+, Br2 , AICI3 , ZnCl2 , FeBr3, BF3, and carbonium ions have Lewis acid properties 18. explain induction effe ...
... form bonds with electron acceptors (as Lewis acids) 17. explain how nucleophiles such as OH-, NH3 , H2O, Br- , Iand carbanion have Lewis base properties, whereas electrophiles such as H+, NO2+, Br2 , AICI3 , ZnCl2 , FeBr3, BF3, and carbonium ions have Lewis acid properties 18. explain induction effe ...
Alberta Chemistry 20-30 Sample CAB Questions - McGraw
... central atom is surrounded by three shared pairs of electrons. According to VSEPR theory, these electrons should be as far apart as possible, so that the electrostatic force of repulsion between them is the minimum. In such a case, the three electron pairs arrange themselves in trigonal planar geome ...
... central atom is surrounded by three shared pairs of electrons. According to VSEPR theory, these electrons should be as far apart as possible, so that the electrostatic force of repulsion between them is the minimum. In such a case, the three electron pairs arrange themselves in trigonal planar geome ...
Worked solutions to the problems
... our warnings cannot be comprehensive - your students will still need your careful supervision. We have also not included specific details for handling or disposal of the products of these lab exercises, as these will vary greatly from country to country, but we know that you will employ best-practic ...
... our warnings cannot be comprehensive - your students will still need your careful supervision. We have also not included specific details for handling or disposal of the products of these lab exercises, as these will vary greatly from country to country, but we know that you will employ best-practic ...
Chap 3 - HCC Learning Web
... This equation, PM = dRT, is derived from the ideal gas equation PV = nRT where P is pressure in atm, V is volume in leter, n is mole, R is the ideal gas constant, 0.082 atm.l/mol.K, and T is temperature in Kelvin. Since n = mole = mass in gram /molar mass, we can re-write PV = nRT to PV= (m/M)RT whe ...
... This equation, PM = dRT, is derived from the ideal gas equation PV = nRT where P is pressure in atm, V is volume in leter, n is mole, R is the ideal gas constant, 0.082 atm.l/mol.K, and T is temperature in Kelvin. Since n = mole = mass in gram /molar mass, we can re-write PV = nRT to PV= (m/M)RT whe ...
p-BLOCK ELEMENTS - einstein classes
... C6H5 · H + RCOCl + AlCl3 RCOC6H5 + H+ + [AlCl4]— AlCl3 is also used to catalyse the reaction to make ethyl bromide (which is used to make the petrol additive ...
... C6H5 · H + RCOCl + AlCl3 RCOC6H5 + H+ + [AlCl4]— AlCl3 is also used to catalyse the reaction to make ethyl bromide (which is used to make the petrol additive ...
Chap 3 - HCC Learning Web
... This equation, PM = dRT, is derived from the ideal gas equation PV = nRT where P is pressure in atm, V is volume in leter, n is mole, R is the ideal gas constant, 0.082 atm.l/mol.K, and T is temperature in Kelvin. Since n = mole = mass in gram /molar mass, we can re-write PV = nRT to PV= (m/M)RT whe ...
... This equation, PM = dRT, is derived from the ideal gas equation PV = nRT where P is pressure in atm, V is volume in leter, n is mole, R is the ideal gas constant, 0.082 atm.l/mol.K, and T is temperature in Kelvin. Since n = mole = mass in gram /molar mass, we can re-write PV = nRT to PV= (m/M)RT whe ...
File
... electron with relative ease to form M+ cations when in ionic compounds. They all are easily oxidized. Therefore, in order to prepare the pure metals, alkali metals must be produced in the absence of materials (H2O, O2) that are capable of oxidizing them. The method of preparation is electrochemical ...
... electron with relative ease to form M+ cations when in ionic compounds. They all are easily oxidized. Therefore, in order to prepare the pure metals, alkali metals must be produced in the absence of materials (H2O, O2) that are capable of oxidizing them. The method of preparation is electrochemical ...
Calculations and the Chemical Equation
... The Mole Concept and Atoms Atoms are exceedingly small, yet their masses have been experimentally determined for each of the elements. The periodic table provides atomic masses in atomic mass units (amu). A more practical unit for defining a "collection" of atoms is the mole, Avogadro's number of pa ...
... The Mole Concept and Atoms Atoms are exceedingly small, yet their masses have been experimentally determined for each of the elements. The periodic table provides atomic masses in atomic mass units (amu). A more practical unit for defining a "collection" of atoms is the mole, Avogadro's number of pa ...
coordination of some monodentate and hybrid multident ate
... A range of low-valent late transition-metal triarylphosphine complexes have been prepared and characterised by a combination of *H, [P and 19F (as appropriate) NMR and IR spectroscopies and mass spectrometry. Some of these complexes have been isolated as single crystals and characterised by X-ray di ...
... A range of low-valent late transition-metal triarylphosphine complexes have been prepared and characterised by a combination of *H, [P and 19F (as appropriate) NMR and IR spectroscopies and mass spectrometry. Some of these complexes have been isolated as single crystals and characterised by X-ray di ...
Chemical Reactions - 2012 Book Archive
... and carbon-14. One mole of carbon still has 6.022 × 1023 carbon atoms, but 98.89% of those atoms are carbon-12, 1.11% are carbon-13, and a trace (about 1 atom in 10 12) are carbon-14. (For more information, see Section 1.6 "Isotopes and Atomic Masses".) Similarly, the molar mass of uranium is 238.03 ...
... and carbon-14. One mole of carbon still has 6.022 × 1023 carbon atoms, but 98.89% of those atoms are carbon-12, 1.11% are carbon-13, and a trace (about 1 atom in 10 12) are carbon-14. (For more information, see Section 1.6 "Isotopes and Atomic Masses".) Similarly, the molar mass of uranium is 238.03 ...
F:\Users\Steven\Documents\Chemistry\CHEM120\Problem Set
... b) Calculate the final concentration of the silver after all the precipitate (solid) has formed. 2) When 75 mL of 0.20M Na3PO4 is added to 125 mL of 0.30 M Zn(NO3)2 a white solid forms. a) Please write the NET ionic reaction that occurred. b) How many grams of solid were made? c) What is the concent ...
... b) Calculate the final concentration of the silver after all the precipitate (solid) has formed. 2) When 75 mL of 0.20M Na3PO4 is added to 125 mL of 0.30 M Zn(NO3)2 a white solid forms. a) Please write the NET ionic reaction that occurred. b) How many grams of solid were made? c) What is the concent ...
expected output
... information on trends, global and local distribution; Justification of importance of course Biology of HIV/AIDS: Overview of immune system, natural immunity to HIV/AIDS; the AIDS Virus and its life Cycle, disease progression (epidemiology), transmission and diagnosis. Treatment and Management: Nutri ...
... information on trends, global and local distribution; Justification of importance of course Biology of HIV/AIDS: Overview of immune system, natural immunity to HIV/AIDS; the AIDS Virus and its life Cycle, disease progression (epidemiology), transmission and diagnosis. Treatment and Management: Nutri ...
Chemistry Honours - SCS Autonomous College
... molecules N2, O2, C2, B2, F2, CO, NO, and their ions; HCl, BeF2, CO2, (idea of s-p mixing and orbital interaction to be given). Formal charge, Valence shell electron pair repulsion theory (VSEPR), shapes of simple molecules and ions containing lone pairs and bond pairs of electrons, multiple bonding ...
... molecules N2, O2, C2, B2, F2, CO, NO, and their ions; HCl, BeF2, CO2, (idea of s-p mixing and orbital interaction to be given). Formal charge, Valence shell electron pair repulsion theory (VSEPR), shapes of simple molecules and ions containing lone pairs and bond pairs of electrons, multiple bonding ...
expected output
... information on trends, global and local distribution; Justification of importance of course. Biology of HIV/AIDS: Overview of immune system, natural immunity to HIV/AIDS; the AIDS Virus and its life Cycle, disease progression (epidemiology), transmission and diagnosis. Treatment and Management: Nutr ...
... information on trends, global and local distribution; Justification of importance of course. Biology of HIV/AIDS: Overview of immune system, natural immunity to HIV/AIDS; the AIDS Virus and its life Cycle, disease progression (epidemiology), transmission and diagnosis. Treatment and Management: Nutr ...
here
... their atoms. Since those atoms are indivisible, they obviously cannot be destroyed, so the total number of atoms in the entire system must stay the same. If the total number of atoms stays the same, the total amount of matter stays the same, and therefore, the mass stays the same. Now let’s consider ...
... their atoms. Since those atoms are indivisible, they obviously cannot be destroyed, so the total number of atoms in the entire system must stay the same. If the total number of atoms stays the same, the total amount of matter stays the same, and therefore, the mass stays the same. Now let’s consider ...
Stoichiometry - coercingmolecules
... We measure ordinary objects either by counting or weighing them, depending on which method is more convenient ...
... We measure ordinary objects either by counting or weighing them, depending on which method is more convenient ...
Chapter 1 Introduction: Matter and Measurement
... Compounds • The less electronegative atom is usually listed first. • A prefix is used to denote the number of atoms of each element in the compound (monois not used on the first element listed, however.) Matter And Measurement ...
... Compounds • The less electronegative atom is usually listed first. • A prefix is used to denote the number of atoms of each element in the compound (monois not used on the first element listed, however.) Matter And Measurement ...
Study Guide for Chapter 22 - Hydrocarbon Compounds
... b. Alkyl groups on end carbons are part of the chain; pentane. c. The chain wasn’t numbered so the substituent has the lowest possible number; 2-methylbutane. d. The methyl group on carbon 4 is part of the chain; 3-methylpentane 55. a. Ethyne has one triple C–C bond and two single C–H bonds. b. All ...
... b. Alkyl groups on end carbons are part of the chain; pentane. c. The chain wasn’t numbered so the substituent has the lowest possible number; 2-methylbutane. d. The methyl group on carbon 4 is part of the chain; 3-methylpentane 55. a. Ethyne has one triple C–C bond and two single C–H bonds. b. All ...
Calculations with Chemical Formulas and Chemical Reactions
... The calculation of either weight is straightforward; it is the sum of the individual atomic weights of all atoms or ions present in one particle of the substance. The average atomic weight of an ...
... The calculation of either weight is straightforward; it is the sum of the individual atomic weights of all atoms or ions present in one particle of the substance. The average atomic weight of an ...