CHE 110 Dr. Nicholas Bizier Office DS 337b email
... Lysine is an amino acid which has the following elemental composition: C, H, O, N. In one experiment, 2.175 g of lysine was combusted to produce 3.94 g of CO2 and 1.89 g H2O. In a separate experiment, 1.873 g of lysine was burned to produce 0.436 g of NH2. The molar mass of lysine is 150 g/mol. Dete ...
... Lysine is an amino acid which has the following elemental composition: C, H, O, N. In one experiment, 2.175 g of lysine was combusted to produce 3.94 g of CO2 and 1.89 g H2O. In a separate experiment, 1.873 g of lysine was burned to produce 0.436 g of NH2. The molar mass of lysine is 150 g/mol. Dete ...
Ch3
... Solve the following conversions How many atoms of silver are in 3.50 moles of silver? Determine the number of moles of carbon disulfide in 34.75 grams of CS2. Determine the number of sulfur atoms in 34.75 grams of CS2. Copyright McGraw-Hill 2009 ...
... Solve the following conversions How many atoms of silver are in 3.50 moles of silver? Determine the number of moles of carbon disulfide in 34.75 grams of CS2. Determine the number of sulfur atoms in 34.75 grams of CS2. Copyright McGraw-Hill 2009 ...
4) What is the term for the procedure of collecting data and recording
... A) atomic notation B) atomic number C) atomic mass D) mass number E) none of the above What is the term for the value which indicates the number of protons for an atom of a given element? A) atomic notation B) atomic number C) atomic mass D) mass number E) none of the above What is the term for the ...
... A) atomic notation B) atomic number C) atomic mass D) mass number E) none of the above What is the term for the value which indicates the number of protons for an atom of a given element? A) atomic notation B) atomic number C) atomic mass D) mass number E) none of the above What is the term for the ...
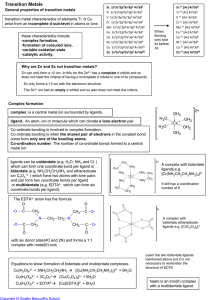
Transition Metals
... Zn can only form a +2 ion. In this ion the Zn2+ has a complete d orbital and so does not meet the criteria of having a incomplete d orbital in one of its compounds. Sc only forms a +3 ion with the electronic structure The Sc3+ ion had an empty d orbital and so also does not meet the criteria ...
... Zn can only form a +2 ion. In this ion the Zn2+ has a complete d orbital and so does not meet the criteria of having a incomplete d orbital in one of its compounds. Sc only forms a +3 ion with the electronic structure The Sc3+ ion had an empty d orbital and so also does not meet the criteria ...
Unit 5 Organic Chemistry
... Life as we know it is based on carbon chemistry (Figure 1). Therefore, it is not surprising that the early definition of organic chemistry was related to compounds obtained only from living things. Today, organic chemistry is a major branch of chemistry that deals with compounds of carbon, excluding ...
... Life as we know it is based on carbon chemistry (Figure 1). Therefore, it is not surprising that the early definition of organic chemistry was related to compounds obtained only from living things. Today, organic chemistry is a major branch of chemistry that deals with compounds of carbon, excluding ...
Safety Quiz - WordPress.com
... Which is easier to separate, a homogeneous or heterogeneous mixture? Explain your answer, giving a specific example. Heterogeneous mixtures are easier to separate because you can see the different components in the mixture. For example, it is easy to separate a mixture of candies because you can det ...
... Which is easier to separate, a homogeneous or heterogeneous mixture? Explain your answer, giving a specific example. Heterogeneous mixtures are easier to separate because you can see the different components in the mixture. For example, it is easy to separate a mixture of candies because you can det ...
Thermal Decomposition of the Non-Interstitial Hydrides for the
... Hydrogen dominates the 15-billion-year tale of our universe (see, e.g., ref 1). It is by far-and-away the most abundant element in the cosmos, of which it makes up 88.6% of the composition by weight. The chemical evolution of the stars depends crucially upon the advance of hydrogen fusion. Bewilderi ...
... Hydrogen dominates the 15-billion-year tale of our universe (see, e.g., ref 1). It is by far-and-away the most abundant element in the cosmos, of which it makes up 88.6% of the composition by weight. The chemical evolution of the stars depends crucially upon the advance of hydrogen fusion. Bewilderi ...
Stoichiometry Notes
... produce Cu2O. Support your answer with calculations. (c) Calculate the maximum number of moles of Cu2O produced when the reaction proceeds to completion. ...
... produce Cu2O. Support your answer with calculations. (c) Calculate the maximum number of moles of Cu2O produced when the reaction proceeds to completion. ...
12_chemistry_impq_CH13_amines_02
... The hydrogen attached to nitrogen in sulphonamide is strongly acidic due to the presence of strong electron withdrawing sulphonyl group. Hence, it is soluble in alkali. (b) In the reaction with secondary amine, N,N-diethylbenzenesulphonamide is formed. ...
... The hydrogen attached to nitrogen in sulphonamide is strongly acidic due to the presence of strong electron withdrawing sulphonyl group. Hence, it is soluble in alkali. (b) In the reaction with secondary amine, N,N-diethylbenzenesulphonamide is formed. ...
B - eko.olunet.org
... Purity Grade of Compounds 3. In chemical experiments, the purity of the starting material and the composition of impurities/additives are of great importance. For his experiments, Thomas needed KBr with at least 95.0% purity. In order to determine the purity of an available inorganic compound, he w ...
... Purity Grade of Compounds 3. In chemical experiments, the purity of the starting material and the composition of impurities/additives are of great importance. For his experiments, Thomas needed KBr with at least 95.0% purity. In order to determine the purity of an available inorganic compound, he w ...
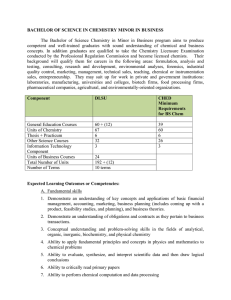
BACHELOR OF SCIENCE IN CHEMISTRY MINOR IN BUSINESS
... This course covers topics in metabolism. The areas to be discussed include bioenergetics, the design and regulation of metabolic pathways and specific molecular processes involved in the synthesis and degradation of major cellular components—the carbohydrates, lipids, proteins and nucleic acid. 3 un ...
... This course covers topics in metabolism. The areas to be discussed include bioenergetics, the design and regulation of metabolic pathways and specific molecular processes involved in the synthesis and degradation of major cellular components—the carbohydrates, lipids, proteins and nucleic acid. 3 un ...
Chemistry - College Catalog
... Comprehensive General Chemistry or Honors General Chemistry forgo partial or full AP credit. Note that no credit is given for IB chemistry. Accreditation. The Department of Chemistry also administers accreditation examinations for Comprehensive General Chemistry I, II, III and Organic Chemistry I, I ...
... Comprehensive General Chemistry or Honors General Chemistry forgo partial or full AP credit. Note that no credit is given for IB chemistry. Accreditation. The Department of Chemistry also administers accreditation examinations for Comprehensive General Chemistry I, II, III and Organic Chemistry I, I ...
Chapter 8
... 1) Matter is composed of small indivisible particles called atoms. All atoms of a particular elements are identical. Atoms of different elements differ in their chemical and physical properties. (Chapter 2). 2) Compounds are composed of molecules. Each molecule contains the same number of atoms of e ...
... 1) Matter is composed of small indivisible particles called atoms. All atoms of a particular elements are identical. Atoms of different elements differ in their chemical and physical properties. (Chapter 2). 2) Compounds are composed of molecules. Each molecule contains the same number of atoms of e ...
13- and 14-membered macrocyclic ligands containing
... same experimental conditions, and the corresponding values are also collected in Tables 1 and 2. The literature values for dota and dotp with the same metal ions are also listed for comparison reasons, however recommended values for these two ligands do not exist [36]. Indeed, the very high values o ...
... same experimental conditions, and the corresponding values are also collected in Tables 1 and 2. The literature values for dota and dotp with the same metal ions are also listed for comparison reasons, however recommended values for these two ligands do not exist [36]. Indeed, the very high values o ...
w_4-3 Chemistry of Nitrogen Compounds
... For the theoretical r value of 1.5, the chlorine dose is 7.6 ppm/ppm ammonia nitrogen and for the typical actual r value of 1.75, the chlorine dose is 8.9 ppm/ppm ammonia nitrogen. However, ammonia itself is typically not present in a chlorine treated pool, but rather in the form of a chloramine. If ...
... For the theoretical r value of 1.5, the chlorine dose is 7.6 ppm/ppm ammonia nitrogen and for the typical actual r value of 1.75, the chlorine dose is 8.9 ppm/ppm ammonia nitrogen. However, ammonia itself is typically not present in a chlorine treated pool, but rather in the form of a chloramine. If ...
Chapter 4: Experimental Techniques
... is selected for the analysis (e.g. 217.0 or 283.3 nm for Pb). The metal being analysed is not usually in its elemental form, and so the first step is digestion (decomposition) of the sample and of a series of standards. The standards contain known concentrations of the metal being analysed and are u ...
... is selected for the analysis (e.g. 217.0 or 283.3 nm for Pb). The metal being analysed is not usually in its elemental form, and so the first step is digestion (decomposition) of the sample and of a series of standards. The standards contain known concentrations of the metal being analysed and are u ...
quantitative_chemistry
... number for us, because if we weigh out a mass in grams that is the same as an element’s atomic mass or a molecule’s formula mass in amu, we will always have 6.022 × 1023 atoms or formula units. The number 6.022 × 1023 is known as Avogadro’s number (NA) (or the Avogadro constant), after the Italian p ...
... number for us, because if we weigh out a mass in grams that is the same as an element’s atomic mass or a molecule’s formula mass in amu, we will always have 6.022 × 1023 atoms or formula units. The number 6.022 × 1023 is known as Avogadro’s number (NA) (or the Avogadro constant), after the Italian p ...
hydrogen storage
... recombine to form molecules inside the tube and arrange themselves in a concentric pattern. The hydrogen pressure inside the SWNT increases with the number of injected atoms and reaches 35 GPa for 90 atoms (5 mass%). This simulation does not show condensation of hydrogen inside the nanotube. The cri ...
... recombine to form molecules inside the tube and arrange themselves in a concentric pattern. The hydrogen pressure inside the SWNT increases with the number of injected atoms and reaches 35 GPa for 90 atoms (5 mass%). This simulation does not show condensation of hydrogen inside the nanotube. The cri ...
RedOx notes:
... Which elements have specific rules? Which element(s) do(es) not have rules? Use rule 8 or 9 from above to calculate these. ...
... Which elements have specific rules? Which element(s) do(es) not have rules? Use rule 8 or 9 from above to calculate these. ...
Chapter 1: Chemistry: The Study of Change
... 12.0 Write the formulas for the following compounds: (a) rubidium nitrite, (b) potassium sulfide, (c) sodium hydrogen sulfide, (d) magnesium phosphate, (e) calcium hydrogen phosphate, (f) potassium dihydrogen phosphate, (g) iodine heptafluoride, (h) ammonium sulfate, (i) silver perchlorate, (j) boro ...
... 12.0 Write the formulas for the following compounds: (a) rubidium nitrite, (b) potassium sulfide, (c) sodium hydrogen sulfide, (d) magnesium phosphate, (e) calcium hydrogen phosphate, (f) potassium dihydrogen phosphate, (g) iodine heptafluoride, (h) ammonium sulfate, (i) silver perchlorate, (j) boro ...