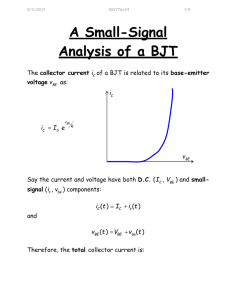
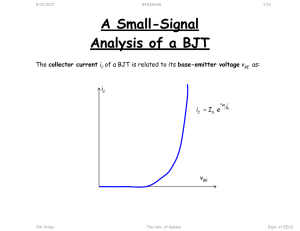
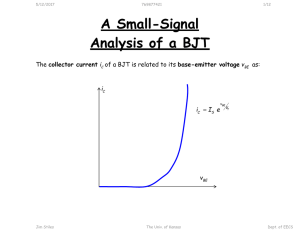
A New Synthetic Method for Nanoscale Metal-
... organic matter, usually wood, peat or even coconut shells. This pyrolysis gives rise to inorganic carbon with pores in its structure. Since its first discovery, the process has lived a strong evolution and now, activated carbon materials with high surface areas (up to 3000 m2·g-1) can be fabricated. ...
... organic matter, usually wood, peat or even coconut shells. This pyrolysis gives rise to inorganic carbon with pores in its structure. Since its first discovery, the process has lived a strong evolution and now, activated carbon materials with high surface areas (up to 3000 m2·g-1) can be fabricated. ...
Complete Solution Manual
... An electrolytic cell uses electrical energy to produce a chemical change. The process of electrolysis involves forcing a current through a cell to produce a chemical change for which the cell potential is negative. Electrical work is used to force a nonspontaneous reaction to occur. The units for cu ...
... An electrolytic cell uses electrical energy to produce a chemical change. The process of electrolysis involves forcing a current through a cell to produce a chemical change for which the cell potential is negative. Electrical work is used to force a nonspontaneous reaction to occur. The units for cu ...
Descriptive Inorganic Chemistry
... but as a part of essential scientific knowledge with applications throughout science and our lives. Because of a need for greater contextualization, we have added more features and more applications. In many colleges and universities, descriptive inorganic chemistry is offered as a sophomore or junio ...
... but as a part of essential scientific knowledge with applications throughout science and our lives. Because of a need for greater contextualization, we have added more features and more applications. In many colleges and universities, descriptive inorganic chemistry is offered as a sophomore or junio ...
CHAPTER 4 REACTIONS IN AQUEOUS SOLUTIONS
... Strategy: Recall that strong acids and strong bases are strong electrolytes. They are completely ionized in solution. An ionic equation will show strong acids and strong bases in terms of their free ions. Weak acids and weak bases are weak electrolytes. They only ionize to a small extent in solution ...
... Strategy: Recall that strong acids and strong bases are strong electrolytes. They are completely ionized in solution. An ionic equation will show strong acids and strong bases in terms of their free ions. Weak acids and weak bases are weak electrolytes. They only ionize to a small extent in solution ...
Thyristor Theory and Design Considerations
... circuits using them. One of the most important is to respect the devices’ rated limits on rate of change of voltage and current (dv/dt and di/dt). If these are exceeded, the thyristor may be damaged or destroyed. On the other hand, it is important to provide a trigger pulse large enough and fast eno ...
... circuits using them. One of the most important is to respect the devices’ rated limits on rate of change of voltage and current (dv/dt and di/dt). If these are exceeded, the thyristor may be damaged or destroyed. On the other hand, it is important to provide a trigger pulse large enough and fast eno ...
thesis_revised
... layered structures. It is expected that by substituting Bi3+ (ionic radius = 0.103 nm) ion with a slightly smaller rare earth cation such as Sm3+ ion (ionic radius = 0.098 nm) could significantly increase the structural distortion in the crystal structure and thereby enhancing the polar properties. ...
... layered structures. It is expected that by substituting Bi3+ (ionic radius = 0.103 nm) ion with a slightly smaller rare earth cation such as Sm3+ ion (ionic radius = 0.098 nm) could significantly increase the structural distortion in the crystal structure and thereby enhancing the polar properties. ...
PART 6-ICHO-26-30
... 1.4 pH in the blood of a person changed from 7.40 to 7.00 due to lactic acid formed during physical activity. Let an aqueous solution having pH = 7.40 and [ HCO3- ] = 0.022 represent blood in the following calculation. How many moles of lactic acid have been added to 1.00 dm3 of this solution when i ...
... 1.4 pH in the blood of a person changed from 7.40 to 7.00 due to lactic acid formed during physical activity. Let an aqueous solution having pH = 7.40 and [ HCO3- ] = 0.022 represent blood in the following calculation. How many moles of lactic acid have been added to 1.00 dm3 of this solution when i ...
Part 2-ICHO-26-30
... 1.4 pH in the blood of a person changed from 7.40 to 7.00 due to lactic acid formed during physical activity. Let an aqueous solution having pH = 7.40 and [ HCO3- ] = 0.022 represent blood in the following calculation. How many moles of lactic acid have been added to 1.00 dm3 of this solution when i ...
... 1.4 pH in the blood of a person changed from 7.40 to 7.00 due to lactic acid formed during physical activity. Let an aqueous solution having pH = 7.40 and [ HCO3- ] = 0.022 represent blood in the following calculation. How many moles of lactic acid have been added to 1.00 dm3 of this solution when i ...
HMC980LP4E - Analog Devices
... power-up and power-down sequencing of the targeted amplifier. The HMC980LP4E can provide auto-bias solution to virtually any amplifier in the market (both enhancement and depletion type) with a quiescent current of up to 1.6A and a supply voltage of up to 16.5V. The HMC980LP4E has an integrated nega ...
... power-up and power-down sequencing of the targeted amplifier. The HMC980LP4E can provide auto-bias solution to virtually any amplifier in the market (both enhancement and depletion type) with a quiescent current of up to 1.6A and a supply voltage of up to 16.5V. The HMC980LP4E has an integrated nega ...
Chapter 4 - Chemistry
... Strategy: Recall that strong acids and strong bases are strong electrolytes. They are completely ionized in solution. An ionic equation will show strong acids and strong bases in terms of their free ions. Weak acids and weak bases are weak electrolytes. They only ionize to a small extent in solution ...
... Strategy: Recall that strong acids and strong bases are strong electrolytes. They are completely ionized in solution. An ionic equation will show strong acids and strong bases in terms of their free ions. Weak acids and weak bases are weak electrolytes. They only ionize to a small extent in solution ...
Second-Order Differential Equations
... associated initial value problems, we discuss a few of the many mathematical models based on second-order equations. The chapter closes with a look at transfer functions, which are used to analyze and design mechanical and electrical oscillators. ...
... associated initial value problems, we discuss a few of the many mathematical models based on second-order equations. The chapter closes with a look at transfer functions, which are used to analyze and design mechanical and electrical oscillators. ...
mcdonald (pam78654) – HW 1: High School Concepts – laude
... M1 = 12.0 M V2 = 2.0 L M2 = 0.40 M A portion of the 12.0 M HCl solution will be diluted with water to form 2.0 L of the 0.4 M HCl solution. All of the moles of HCl in the new dilute solution must come from the more concentrated solution. (There is no HCl in the water!) We use the desired volume and ...
... M1 = 12.0 M V2 = 2.0 L M2 = 0.40 M A portion of the 12.0 M HCl solution will be diluted with water to form 2.0 L of the 0.4 M HCl solution. All of the moles of HCl in the new dilute solution must come from the more concentrated solution. (There is no HCl in the water!) We use the desired volume and ...
Nanofluidic circuitry
Nanofluidic circuitry is a nanotechnology aiming for control of fluids in nanometer scale. Due to the effect of an electrical double layer within the fluid channel, the behavior of nanofluid is observed to be significantly different compared with its microfluidic counterparts. Its typical characteristic dimensions fall within the range of 1–100 nm. At least one dimension of the structure is in nanoscopic scale. Phenomena of fluids in nano-scale structure are discovered to be of different properties in electrochemistry and fluid dynamics.