Specification Status: RELEASED PolyZen GENERAL DESCRIPTION
... PolyZen Zener diode was selected due to its relatively flat voltage vs current response. This helps improve output voltage clamping, even when input voltage is high and diode currents are large. An advanced feature of the PolyZen devices is that the Zener diode is thermally coupled to a resistively ...
... PolyZen Zener diode was selected due to its relatively flat voltage vs current response. This helps improve output voltage clamping, even when input voltage is high and diode currents are large. An advanced feature of the PolyZen devices is that the Zener diode is thermally coupled to a resistively ...
Specification Status: RELEASED PolyZen GENERAL DESCRIPTION
... PolyZen Zener diode was selected due to its relatively flat voltage vs current response. This helps improve output voltage clamping, even when input voltage is high and diode currents are large. An advanced feature of the PolyZen devices is that the Zener diode is thermally coupled to a resistively ...
... PolyZen Zener diode was selected due to its relatively flat voltage vs current response. This helps improve output voltage clamping, even when input voltage is high and diode currents are large. An advanced feature of the PolyZen devices is that the Zener diode is thermally coupled to a resistively ...
Answers - Scioly.org
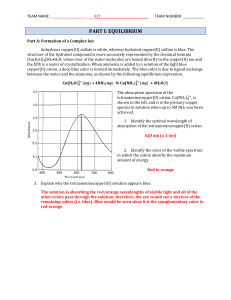
... NH4+ and Cu2+ ions. The reaction mixture will reestablish a new equilibrium position based on the value of Kf since the concentrations of the reactants and products have changed. ...
... NH4+ and Cu2+ ions. The reaction mixture will reestablish a new equilibrium position based on the value of Kf since the concentrations of the reactants and products have changed. ...
Ohms Law - ClassNet
... Resistance can be explained as the amount of resistance voltage has to overcome in a circuit for current to flow. The byproduct of resistance is heat we’ll talk about that later. Let’s now look at why copper is used as a carrier for electricity. To find out what makes it so special we have to unders ...
... Resistance can be explained as the amount of resistance voltage has to overcome in a circuit for current to flow. The byproduct of resistance is heat we’ll talk about that later. Let’s now look at why copper is used as a carrier for electricity. To find out what makes it so special we have to unders ...
Using molecular dynamics to quantify the electrical double layer and
... modern electronics such as memory-backup systems and electric vehicles [1-3]. These devices have power density capabilities that exceed conventional energy systems. Charge/discharge cycles can be performed very fast, and EDLCs can withstand more than 1 million operational cycles [1,3]. EDLC behavior ...
... modern electronics such as memory-backup systems and electric vehicles [1-3]. These devices have power density capabilities that exceed conventional energy systems. Charge/discharge cycles can be performed very fast, and EDLCs can withstand more than 1 million operational cycles [1,3]. EDLC behavior ...
chapter 4 - reactions in solution
... It is very essential to life. The human body is made up of about 65% water. o It provides essential environments for biochemical reactions that keep us alive; o It helps to maintain a constant body temperature; o moderates the Earth’s temperature; It is widely used in industrial cooling system, powe ...
... It is very essential to life. The human body is made up of about 65% water. o It provides essential environments for biochemical reactions that keep us alive; o It helps to maintain a constant body temperature; o moderates the Earth’s temperature; It is widely used in industrial cooling system, powe ...
Introduction to Electricity
... circuit, and back to the negative side of the battery. This was the convention established when electricity was first discovered, but it is incorrect! Electron Flow is what actually happens. The electrons flow out of the negative side of the battery, through the circuit, and back to the positive sid ...
... circuit, and back to the negative side of the battery. This was the convention established when electricity was first discovered, but it is incorrect! Electron Flow is what actually happens. The electrons flow out of the negative side of the battery, through the circuit, and back to the positive sid ...
Physics_AP_B_Evans_Day_09_Period_2
... connected in series with the external resistance R. r causes the potential difference between the terminals to drop below the maximum EMF. This actual voltage is the terminal voltage. ...
... connected in series with the external resistance R. r causes the potential difference between the terminals to drop below the maximum EMF. This actual voltage is the terminal voltage. ...
Skill Sheet 19-B Chemical Formulas
... Chemical formulas have two important parts: chemical symbols for the elements in the compound and subscripts that tell how many atoms of each element are needed to form the compound. The chemical formula for water, H2O, tells us that a water molecule is made of the elements hydrogen (H) and oxygen ( ...
... Chemical formulas have two important parts: chemical symbols for the elements in the compound and subscripts that tell how many atoms of each element are needed to form the compound. The chemical formula for water, H2O, tells us that a water molecule is made of the elements hydrogen (H) and oxygen ( ...
Class Notes
... The law of conservation of mass (aka: the law of conservation of matter) says that during a chemical reaction or a physical change mass is conserved; mass is neither created nor destroyed. This implies that the atoms that were there in the reactants (before the chemical change) must be there in the ...
... The law of conservation of mass (aka: the law of conservation of matter) says that during a chemical reaction or a physical change mass is conserved; mass is neither created nor destroyed. This implies that the atoms that were there in the reactants (before the chemical change) must be there in the ...
Ch2hon ppt part 3
... Empirical - Simplified ratio Molecular - How many atoms in a single molecule (not always a simplified ratio, glucose = C6H12O6) Structural - Specifies which atoms are bonded to each other (organic chem) ...
... Empirical - Simplified ratio Molecular - How many atoms in a single molecule (not always a simplified ratio, glucose = C6H12O6) Structural - Specifies which atoms are bonded to each other (organic chem) ...
HighFour Chemistry Round 1 Category C: Grades 9 – 10 Thursday
... equivalent to the atomic number because the number of protons remains unchanged during reactions. Referring to Table 24 (Periodic Table) of the Chemistry Data Booklet, the element with an atomic number of 15 is phosphorus, P. ...
... equivalent to the atomic number because the number of protons remains unchanged during reactions. Referring to Table 24 (Periodic Table) of the Chemistry Data Booklet, the element with an atomic number of 15 is phosphorus, P. ...
CP Chemistry Midterm Study Guide
... 21. How many grams of MgCl2 are in 500 mL of a 3.2 M solution? 22. How many atoms of sulfur do you have if you have 4 moles? 23. Convert 500 grams of magnesium to moles. 24. If we have 6.02x1023 molecules of oxygen, how many moles do we have? 25. One mole of carbon (C) atoms contains _______________ ...
... 21. How many grams of MgCl2 are in 500 mL of a 3.2 M solution? 22. How many atoms of sulfur do you have if you have 4 moles? 23. Convert 500 grams of magnesium to moles. 24. If we have 6.02x1023 molecules of oxygen, how many moles do we have? 25. One mole of carbon (C) atoms contains _______________ ...
Ionization methods - 2-CI - Florida International University
... depends on the relative affinities of the conjugate base of the reactant ion (CH 4, NH3 and so on) and the compound M. • Decrease in PA (proton affinity) of the conjugate base (or increase in acidity of the reactant gas ion) causes increase in fragmentation because more energy is transferred to them ...
... depends on the relative affinities of the conjugate base of the reactant ion (CH 4, NH3 and so on) and the compound M. • Decrease in PA (proton affinity) of the conjugate base (or increase in acidity of the reactant gas ion) causes increase in fragmentation because more energy is transferred to them ...
Nanofluidic circuitry
Nanofluidic circuitry is a nanotechnology aiming for control of fluids in nanometer scale. Due to the effect of an electrical double layer within the fluid channel, the behavior of nanofluid is observed to be significantly different compared with its microfluidic counterparts. Its typical characteristic dimensions fall within the range of 1–100 nm. At least one dimension of the structure is in nanoscopic scale. Phenomena of fluids in nano-scale structure are discovered to be of different properties in electrochemistry and fluid dynamics.