IN-SITU CHARACTERIZATION OF SPUTTERED PD THIN-FILMS UNDERGOING ELECTROLYSIS
... Figure 1. (a) Schematic for the resistance measurement. (b) A Pd/Ni electrode with electrical leads attached through indium contacts The immediate problem encountered in measuring resistance during electrolysis is the bypass (short-circuit) effect of electrolyte. For a wire experiment [2], this is n ...
... Figure 1. (a) Schematic for the resistance measurement. (b) A Pd/Ni electrode with electrical leads attached through indium contacts The immediate problem encountered in measuring resistance during electrolysis is the bypass (short-circuit) effect of electrolyte. For a wire experiment [2], this is n ...
Precipitation Reactions
... The rules you just learned assume that the redox reaction is taking place under acidic conditions. (You are, after all, either producing or consuming H+ ions.) There are slightly different rules for basic conditions: 1. Balance the reaction (using your method of choice) as if it were under acidic co ...
... The rules you just learned assume that the redox reaction is taking place under acidic conditions. (You are, after all, either producing or consuming H+ ions.) There are slightly different rules for basic conditions: 1. Balance the reaction (using your method of choice) as if it were under acidic co ...
(NH 3 ) 2 - GZ @ Science Class Online
... Ionic Bonding is where one atom completely takes valence electrons from another to form ions and the resulting negative and positive ions hold together with electrostatic attraction. This type of bonding occurs when a metal and non-metal react and there is a transfer of electrons to form ions. The i ...
... Ionic Bonding is where one atom completely takes valence electrons from another to form ions and the resulting negative and positive ions hold together with electrostatic attraction. This type of bonding occurs when a metal and non-metal react and there is a transfer of electrons to form ions. The i ...
Lecture 4-2 - Purdue Physics
... outside a large spherical surface. One is at the center while the other is outside the surface. Which statement about the net electric flux through the surfaces of the sphere is true? a) The flux is larger when the charge is outside the surface. b) The magnitude of the flux is the same. c) The flux ...
... outside a large spherical surface. One is at the center while the other is outside the surface. Which statement about the net electric flux through the surfaces of the sphere is true? a) The flux is larger when the charge is outside the surface. b) The magnitude of the flux is the same. c) The flux ...
TEMPUS: PROTECTION AGAINST OVERVOLTAGES 1: The use of
... Overvoltage protection is not always obtained by using arcing horns in the free air. An encapsulated or gas filled arcing horn has the same working principle but the horns or electrodes are encapsulated (sometimes they contain a noble gas). 2: Voltage Dependent Resistors Instead of using arcing hor ...
... Overvoltage protection is not always obtained by using arcing horns in the free air. An encapsulated or gas filled arcing horn has the same working principle but the horns or electrodes are encapsulated (sometimes they contain a noble gas). 2: Voltage Dependent Resistors Instead of using arcing hor ...
Dilutions Worksheet
... How much water would I need to add to 500 mL of a 2.4 M KCl solution to make a 1.0 M solution? M1V1 = M2V2 (2.4 M)(500 mL) = (1.0 M) x x = 1200 mL 1200 mL will be the final volume of the solution. However, since there’s already 500 mL of solution present, you only need to add 700 mL of water to get ...
... How much water would I need to add to 500 mL of a 2.4 M KCl solution to make a 1.0 M solution? M1V1 = M2V2 (2.4 M)(500 mL) = (1.0 M) x x = 1200 mL 1200 mL will be the final volume of the solution. However, since there’s already 500 mL of solution present, you only need to add 700 mL of water to get ...
Name
... Potential Drop Around The Circuit Lab Purpose In this lab, you will investigate the potential drop around a circuit. Background Kirchhoff's Loop Rule (also called Kirchhoff's Voltage Law or just the Loop Rule): The sum of the potential differences across all circuit elements (following the loop rule ...
... Potential Drop Around The Circuit Lab Purpose In this lab, you will investigate the potential drop around a circuit. Background Kirchhoff's Loop Rule (also called Kirchhoff's Voltage Law or just the Loop Rule): The sum of the potential differences across all circuit elements (following the loop rule ...
Electrostatics/Electric Circuits/Magnetic Fields
... 3.1 Direct Current Current and Electromotive Force A battery is a device that stores electrical energy. Most batteries have two terminals (conducting surfaces), one positive (high potential), the other negative (low potential). When a conducting loop of wire is placed on each of a battery’s terminal ...
... 3.1 Direct Current Current and Electromotive Force A battery is a device that stores electrical energy. Most batteries have two terminals (conducting surfaces), one positive (high potential), the other negative (low potential). When a conducting loop of wire is placed on each of a battery’s terminal ...
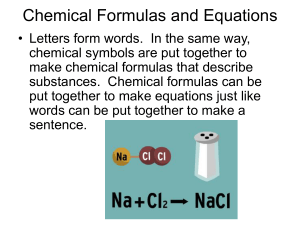
Chemical Formulas and Equations
... chemical symbols are put together to make chemical formulas that describe substances. Chemical formulas can be put together to make equations just like words can be put together to make a sentence. ...
... chemical symbols are put together to make chemical formulas that describe substances. Chemical formulas can be put together to make equations just like words can be put together to make a sentence. ...
Chemistry - SchoolNotes.com
... 47) An open manometer is filled with mercury. The difference in the mercury level in the arms is 81.2 mm. The mercury level is higher in the gas sample arm. What is the pressure, in kilopascals, of the gas in the container if the air pressure is 95.6 kPa? 84.7 kPa ...
... 47) An open manometer is filled with mercury. The difference in the mercury level in the arms is 81.2 mm. The mercury level is higher in the gas sample arm. What is the pressure, in kilopascals, of the gas in the container if the air pressure is 95.6 kPa? 84.7 kPa ...
Welcome to AP Chemistry! AP Chemistry is
... which contains a monatomic anion, drop the start the name with hydro, drop the -ide ending of the anion and add the suffix, -ic acid. For example, HBr is hydrobromic acid. 2. Oxyacids contain polyatomic anions such as nitrite, carbonate, etc. To name an acid with an anion ending with the -ite suffix ...
... which contains a monatomic anion, drop the start the name with hydro, drop the -ide ending of the anion and add the suffix, -ic acid. For example, HBr is hydrobromic acid. 2. Oxyacids contain polyatomic anions such as nitrite, carbonate, etc. To name an acid with an anion ending with the -ite suffix ...
Nanofluidic circuitry
Nanofluidic circuitry is a nanotechnology aiming for control of fluids in nanometer scale. Due to the effect of an electrical double layer within the fluid channel, the behavior of nanofluid is observed to be significantly different compared with its microfluidic counterparts. Its typical characteristic dimensions fall within the range of 1–100 nm. At least one dimension of the structure is in nanoscopic scale. Phenomena of fluids in nano-scale structure are discovered to be of different properties in electrochemistry and fluid dynamics.