Question 2 - The King`s School, Canterbury
... 1. (a) A student investigated the effect of light intensity on leaf size. The student collected 25 leaves from bramble plants at two different sites. One of the sites was a woodland with low light levels and the other, a woodland with high light levels. The student found the average surface area of ...
... 1. (a) A student investigated the effect of light intensity on leaf size. The student collected 25 leaves from bramble plants at two different sites. One of the sites was a woodland with low light levels and the other, a woodland with high light levels. The student found the average surface area of ...
Core Lab 4 Newton`s Second Law of Motion - eLearning
... Student B “ The bigger the pull or the push, the bigger the change in motion experienced by an object. There is a linear relationship between the size of the exerted force and the acceleration experienced by an object.” Student C “The amount of mass to be moved is also important. If the mass is incr ...
... Student B “ The bigger the pull or the push, the bigger the change in motion experienced by an object. There is a linear relationship between the size of the exerted force and the acceleration experienced by an object.” Student C “The amount of mass to be moved is also important. If the mass is incr ...
UNIVERSITY OF CALICUT Scheme and Syllabus for 2010 M.Sc. (Physics) Programme(CSS)
... CGPA – between 0 to 4 and in two decimal points. An overall letter grade (Cumulative Grade) for the whole programme shall be awarded to the student based on the value of CGPA using a 7-point scale given below. Overall Grade in a Programme CGPA 3.80 to 4.00 3.50 to 3.79 3.00 to 3.49 2.50 to 2.99 2.00 ...
... CGPA – between 0 to 4 and in two decimal points. An overall letter grade (Cumulative Grade) for the whole programme shall be awarded to the student based on the value of CGPA using a 7-point scale given below. Overall Grade in a Programme CGPA 3.80 to 4.00 3.50 to 3.79 3.00 to 3.49 2.50 to 2.99 2.00 ...
Stoichiometry
... b. Suppose that x moles of water form in the above reaction. The other three mole quantities (not in order) are 2x, 1.5x, and 0.5x. Match these quantities to their respective components in the ...
... b. Suppose that x moles of water form in the above reaction. The other three mole quantities (not in order) are 2x, 1.5x, and 0.5x. Match these quantities to their respective components in the ...
msc_f_p1b2 - Bhoj University
... is a universal phenomenon independent of the nature of the reference frame that allows calculation of energy values of the system under different conditions. In relativistic domain also it holds good. Rate of change of angular momentum gives torque on the system of particles. In the following discus ...
... is a universal phenomenon independent of the nature of the reference frame that allows calculation of energy values of the system under different conditions. In relativistic domain also it holds good. Rate of change of angular momentum gives torque on the system of particles. In the following discus ...
The d- and f- Block Element Block Elements The d- and f
... The irregular trend in the first ionisation enthalpy of the 3d metals, though of little chemical significance, can be accounted for by considering that the removal of one electron alters the relative n energies of 4s and 3d orbitals. So the unipositive ions have d configurations with no 4s electrons ...
... The irregular trend in the first ionisation enthalpy of the 3d metals, though of little chemical significance, can be accounted for by considering that the removal of one electron alters the relative n energies of 4s and 3d orbitals. So the unipositive ions have d configurations with no 4s electrons ...
29 Sept 08 - Seattle Central
... • Chemistry is the study of the rearrangement of matter due to the flow of energy. • In a chemical reaction, some bonds are broken and others are formed, resulting in a reorganization of the atoms. • Atoms are neither created or destroyed in a chemical reaction! When methane (CH4) reacts with oxygen ...
... • Chemistry is the study of the rearrangement of matter due to the flow of energy. • In a chemical reaction, some bonds are broken and others are formed, resulting in a reorganization of the atoms. • Atoms are neither created or destroyed in a chemical reaction! When methane (CH4) reacts with oxygen ...
Physical Science - SC1114 Unit Topic Lesson Objectives Define
... Elements, and the Periodic Table Atomic Structure Explain Dalton's atomic theory, and describe why it was more successful than Democritus's theory. State the charge, mass, and location of each part of an atom according to the modern model of the atom. Compare and contrast Bohr's model with the moder ...
... Elements, and the Periodic Table Atomic Structure Explain Dalton's atomic theory, and describe why it was more successful than Democritus's theory. State the charge, mass, and location of each part of an atom according to the modern model of the atom. Compare and contrast Bohr's model with the moder ...
Chemical Reactions
... this problem, chemists long ago defined a unit called the mole (mol). A mole is the amount of substance that contains as many atoms, molecules, or ions as there are atoms in exactly 12 g of carbon-12. The important point here is that whether we are dealing with a mole of iron atoms, a mole of methan ...
... this problem, chemists long ago defined a unit called the mole (mol). A mole is the amount of substance that contains as many atoms, molecules, or ions as there are atoms in exactly 12 g of carbon-12. The important point here is that whether we are dealing with a mole of iron atoms, a mole of methan ...
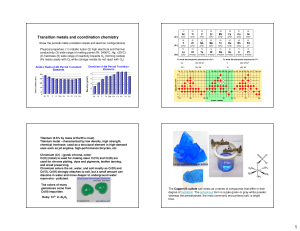
Transition metals and coordination chemistry
... molecule of the chemical formula [Pt(NH3)2Cl2] were connected. The theories at the time predicted such molecules to be connected as linear chains: [Pt-NH3-NH3-Cl]Cl or Cl-NH3-Pt-NH3-Cl ...
... molecule of the chemical formula [Pt(NH3)2Cl2] were connected. The theories at the time predicted such molecules to be connected as linear chains: [Pt-NH3-NH3-Cl]Cl or Cl-NH3-Pt-NH3-Cl ...
AP Chemistry - Freehold Regional High School District
... All compounds are molecules but not all molecules are compounds. 1. Students will understand that subatomic particles define an atom. 2. The periodic table is an important tool in chemistry due to its organized arrangement. 3. All molecules can be represented by a formula that can be written using a ...
... All compounds are molecules but not all molecules are compounds. 1. Students will understand that subatomic particles define an atom. 2. The periodic table is an important tool in chemistry due to its organized arrangement. 3. All molecules can be represented by a formula that can be written using a ...
Coupling Quantum Tunneling with Cavity Photons
... thus possessing an additional static dipole moment aligned perpendicularly to the plane—and has a very small overlap of electron and hole wave functions, hence low oscillator strength. When a bias voltage is applied to bring the electron levels into resonance, the electron states in the two QWs mix ...
... thus possessing an additional static dipole moment aligned perpendicularly to the plane—and has a very small overlap of electron and hole wave functions, hence low oscillator strength. When a bias voltage is applied to bring the electron levels into resonance, the electron states in the two QWs mix ...
quantum field theory in curved spacetime
... All this is just as in conventional particle physics. The only trouble with it is: it's wrong_. It is not wrong in a technical mathematical sense. It simply provides a grossly inadequate founqation for the theory. Here are just some of the situations in which it fails: 1. There may be no Killing vec ...
... All this is just as in conventional particle physics. The only trouble with it is: it's wrong_. It is not wrong in a technical mathematical sense. It simply provides a grossly inadequate founqation for the theory. Here are just some of the situations in which it fails: 1. There may be no Killing vec ...
Chapter 1 Introduction: Matter and Measurement
... Atomic Theory of Matter The theory that atoms are the fundamental building blocks of matter reemerged in the early 19th century, championed by John Dalton. ...
... Atomic Theory of Matter The theory that atoms are the fundamental building blocks of matter reemerged in the early 19th century, championed by John Dalton. ...
第三次工業革命
... However, in the 190 mass region, the decay from the SD to the normal states is spread over many different available paths. This means the SD state are coupled to many ND states. Therefore it is difficult to understand how the single ND state model is able to account for the data in the 190 mass regi ...
... However, in the 190 mass region, the decay from the SD to the normal states is spread over many different available paths. This means the SD state are coupled to many ND states. Therefore it is difficult to understand how the single ND state model is able to account for the data in the 190 mass regi ...
Chapter 7 -- Removal of Particles from Gas Streams
... object in its path, suspended particles tend to keep moving in their original direction due to their inertia. Particulate collection devices based on this principle include cyclones, scrubbers, and filters. 4. Brownian diffusion. Particles suspended in a gas are always in Brownian motion. When the g ...
... object in its path, suspended particles tend to keep moving in their original direction due to their inertia. Particulate collection devices based on this principle include cyclones, scrubbers, and filters. 4. Brownian diffusion. Particles suspended in a gas are always in Brownian motion. When the g ...
Universal diffraction of atoms and molecules from a quantum
... could be that some dimers start to break up as they approach closer to the surface with increasing incidence angle. Furthermore, we note that universal behavior was also found for the –3rd-order diffraction efficiency curves of He, He2, and D2. In conclusion, we have observed emerging beam resonance ...
... could be that some dimers start to break up as they approach closer to the surface with increasing incidence angle. Furthermore, we note that universal behavior was also found for the –3rd-order diffraction efficiency curves of He, He2, and D2. In conclusion, we have observed emerging beam resonance ...
The Vacuum-Lattice model – a new route to longitudinal
... the vacuum lattice comprises real electrons and positrons it has an enormous mass density, estimated as, me/δ3, of about 1013kg/m3, or roughly 109 times more dense than iron! Simhony argued that although such a high density may be psychologically difficult for us to accept, it is not really a proble ...
... the vacuum lattice comprises real electrons and positrons it has an enormous mass density, estimated as, me/δ3, of about 1013kg/m3, or roughly 109 times more dense than iron! Simhony argued that although such a high density may be psychologically difficult for us to accept, it is not really a proble ...
Chapter 12: Symmetries in Physics: Isospin and the Eightfold Way
... M eV /c2 ’s which is minuscule compared to the typical energy scale of hadrons (i.e. strongly interacting particles) which is about a GeV /c2 . This is why isospin is such a good symmetry and why isomultiplets have nearly identical masses. As it later turned out, the up and down quarks are not the o ...
... M eV /c2 ’s which is minuscule compared to the typical energy scale of hadrons (i.e. strongly interacting particles) which is about a GeV /c2 . This is why isospin is such a good symmetry and why isomultiplets have nearly identical masses. As it later turned out, the up and down quarks are not the o ...
Chapter 3
... 4. Balance those elements that appear in two or more reactants or products. 4. Remove all fractions (generally by multiplying everything by 2) and reduce all stoichiometric coefficients to try to get one equal to 1 (generally by dividing everything by 2). 5. Check to make sure that you have the same ...
... 4. Balance those elements that appear in two or more reactants or products. 4. Remove all fractions (generally by multiplying everything by 2) and reduce all stoichiometric coefficients to try to get one equal to 1 (generally by dividing everything by 2). 5. Check to make sure that you have the same ...
Stoichiometric Conversions
... gas, the two will combust and form carbon dioxide and water CH4 + 2O2 CO2 + 2H2O How many moles of H2O will be formed when 28.0 g of methane combusts? ...
... gas, the two will combust and form carbon dioxide and water CH4 + 2O2 CO2 + 2H2O How many moles of H2O will be formed when 28.0 g of methane combusts? ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.