FINAL EXAM REVIEW
... 1. What is the molar mass of the following compounds? a. Pb(C2O4)2 b. Ni(OH)2 c. Tin (IV) acetate pentahydrate d. CH3COOH 2. Calculate the mass of the following: a. 7.01 mol of SiF4 b. 6.59 x 10-4 mol H3PO4 c. 0.0765 mol Li2HSO4 d. 6.85 mol CH3CH2 CH2 CH2CH3 3. Calculate the number of moles of the f ...
... 1. What is the molar mass of the following compounds? a. Pb(C2O4)2 b. Ni(OH)2 c. Tin (IV) acetate pentahydrate d. CH3COOH 2. Calculate the mass of the following: a. 7.01 mol of SiF4 b. 6.59 x 10-4 mol H3PO4 c. 0.0765 mol Li2HSO4 d. 6.85 mol CH3CH2 CH2 CH2CH3 3. Calculate the number of moles of the f ...
SpectraPart2
... Describe or sketch a simple model of the atom (the Bohr model) including permitted orbits. Given a model of the atom, show which jumps correspond to emission spectra and which to absorption spectra. Given a model of the atom showing several energy levels, identify which photon comes from which elect ...
... Describe or sketch a simple model of the atom (the Bohr model) including permitted orbits. Given a model of the atom, show which jumps correspond to emission spectra and which to absorption spectra. Given a model of the atom showing several energy levels, identify which photon comes from which elect ...
PPT format
... about the same distance from the nucleus as the other electrons in the shell. The electrons in a shell with the same n are spread out and do not shield each other from the positive charge of the nucleus very well. Thus, the effective nuclear charge, Zeff, increases as Z increases across the periodic ...
... about the same distance from the nucleus as the other electrons in the shell. The electrons in a shell with the same n are spread out and do not shield each other from the positive charge of the nucleus very well. Thus, the effective nuclear charge, Zeff, increases as Z increases across the periodic ...
acceleration: change in an object`s speed or direction (velocity) over
... parasite: organism that feeds on the body another living organism periodic table of elements: a organized chart of all the elements permeability: description of how well a material allows water to pass through phenotype: the physical appearance of a trait of an organism photosynthesis: chemical proc ...
... parasite: organism that feeds on the body another living organism periodic table of elements: a organized chart of all the elements permeability: description of how well a material allows water to pass through phenotype: the physical appearance of a trait of an organism photosynthesis: chemical proc ...
Physics 124 : Particles and Waves
... of electron orbits and characterized by the quantum number n = 1, 2, 3, … 3. Each state has a discrete, well-defined energy En, with E1
... of electron orbits and characterized by the quantum number n = 1, 2, 3, … 3. Each state has a discrete, well-defined energy En, with E1
Quantum Theory and Atomic Structure
... the exact position and momentum (velocity) of a particle can not be known simultaneously ∆x⋅∆p ≥ h/4π ∆x and ∆p = m∆u – uncertainty in position and momentum, respectively – A consequence of the wave-particle duality of matter – The exact location of very small particles is not well known due to thei ...
... the exact position and momentum (velocity) of a particle can not be known simultaneously ∆x⋅∆p ≥ h/4π ∆x and ∆p = m∆u – uncertainty in position and momentum, respectively – A consequence of the wave-particle duality of matter – The exact location of very small particles is not well known due to thei ...
The Bohr atom and the Uncertainty Principle
... The minimum energy (binding energy) we need to provide to ionize an H atom in its ground state (n=1) is 13.6 eV (it is lower if the atom is in some excited state and the electron is less bound to the nucleus). ...
... The minimum energy (binding energy) we need to provide to ionize an H atom in its ground state (n=1) is 13.6 eV (it is lower if the atom is in some excited state and the electron is less bound to the nucleus). ...
End Show
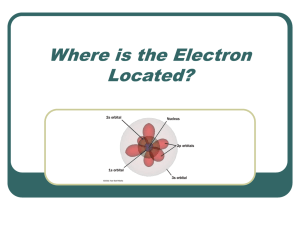
... What does the quantum mechanical model determine about the electrons in an atom? The quantum mechanical model determines the allowed energies an electron can have and how likely it is to find the electron in various locations Slide around the nucleus. 6 of 26 © Copyright Pearson Prentice Hall ...
... What does the quantum mechanical model determine about the electrons in an atom? The quantum mechanical model determines the allowed energies an electron can have and how likely it is to find the electron in various locations Slide around the nucleus. 6 of 26 © Copyright Pearson Prentice Hall ...
1.5.16(Chem) - mrcarlsonschemistryclass
... Cations and Anions • Cations are ions with a POSITIVE charge. • Anions are ions with a NEGATIVE charge. • Draw the funny way to remember cations and anions: ...
... Cations and Anions • Cations are ions with a POSITIVE charge. • Anions are ions with a NEGATIVE charge. • Draw the funny way to remember cations and anions: ...
3.3 The Quantum Mechanical Model of the Atom
... • Heisenberg’s Uncertainty Principle is the idea that it is impossible to know the exact position and speed of an electron at the same time – The method used to determine the speed of an electron changes its position – The method used to determine the position of an electron changes its speed ...
... • Heisenberg’s Uncertainty Principle is the idea that it is impossible to know the exact position and speed of an electron at the same time – The method used to determine the speed of an electron changes its position – The method used to determine the position of an electron changes its speed ...
Slide 1
... Not only is energy & momentum QUANTIZED (energy levels/orbitals) but like photons are quanta of electromagnetic energy, all particle states are the physical manifestation of quantum mechanical wave functions (fields). Not only does each atomic electron exist trapped within quantized energy levels or ...
... Not only is energy & momentum QUANTIZED (energy levels/orbitals) but like photons are quanta of electromagnetic energy, all particle states are the physical manifestation of quantum mechanical wave functions (fields). Not only does each atomic electron exist trapped within quantized energy levels or ...
Chemistry Module 1- Basic Revision Notes 1.1a Atomic Structure 1.1
... 1.1.3 Elements (H, He, Li, Be,…..) are the basic building blocks of all matter, and cannot be broken down into simpler parts by chemical means. 1.1.4 There is a clear relationship between an elements electronic structure and its position in the periodic table. P E r i o d ...
... 1.1.3 Elements (H, He, Li, Be,…..) are the basic building blocks of all matter, and cannot be broken down into simpler parts by chemical means. 1.1.4 There is a clear relationship between an elements electronic structure and its position in the periodic table. P E r i o d ...
Answer key
... Protons and neutrons are found in the center of the atom, called the nucleus. The electrons move about in the electron cloud that surrounds the nucleus. 46. Which subatomic particle(s) defines the identity of the atom? Protons 47. Which subatomic particle(s) determines chemical properties? electrons ...
... Protons and neutrons are found in the center of the atom, called the nucleus. The electrons move about in the electron cloud that surrounds the nucleus. 46. Which subatomic particle(s) defines the identity of the atom? Protons 47. Which subatomic particle(s) determines chemical properties? electrons ...
File
... Protons and neutrons are found in the center of the atom, called the nucleus. The electrons move about in the electron cloud that surrounds the nucleus. 46. Which subatomic particle(s) defines the identity of the atom? Protons 47. Which subatomic particle(s) determines chemical properties? electrons ...
... Protons and neutrons are found in the center of the atom, called the nucleus. The electrons move about in the electron cloud that surrounds the nucleus. 46. Which subatomic particle(s) defines the identity of the atom? Protons 47. Which subatomic particle(s) determines chemical properties? electrons ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.