Chemistry - Set as Home Page
... 23. The symbolic representation of a molecule of a compound is called __________. 24. Molecular formula of CHCl3 and its Empirical formula is __________. 25. Molecular formula of benzene is C6H6 and its empirical formula is __________. 26. 58.5 is the __________ of NaCl. 27. 4.5 gms of nitrogen wil ...
... 23. The symbolic representation of a molecule of a compound is called __________. 24. Molecular formula of CHCl3 and its Empirical formula is __________. 25. Molecular formula of benzene is C6H6 and its empirical formula is __________. 26. 58.5 is the __________ of NaCl. 27. 4.5 gms of nitrogen wil ...
Your views are welcomed upon the theme of
... Teaching chemistry - but ignoring the ‘why’ question? One way of avoiding the question of how to teach about ‘why reactions occur’ to relatively unsophisticated school pupils, is not to teach about this at all. Indeed some teachers have expressed the view that a good deal of the theoretical material ...
... Teaching chemistry - but ignoring the ‘why’ question? One way of avoiding the question of how to teach about ‘why reactions occur’ to relatively unsophisticated school pupils, is not to teach about this at all. Indeed some teachers have expressed the view that a good deal of the theoretical material ...
AP Chem Equations - Speedway High School
... Remember which acids are strong (ionize completely) and which are weak (write as molecule). ...
... Remember which acids are strong (ionize completely) and which are weak (write as molecule). ...
WRITING AP EQUATIONS AP equation sets are found in the free
... Remember which acids are strong (ionize completely) and which are weak (write as molecule). ...
... Remember which acids are strong (ionize completely) and which are weak (write as molecule). ...
Reductive decomposition of calcium sulfate with carbon monoxide
... potential must be greater than that indicated by Figure 1. It can be seen that if the sulfur dioxide concentration of the gas phase is limited to 10 mol '30, the equilibrium reducing potential for reaction 7 will be less than that for reaction 4 as long as the temperature of the system is greater th ...
... potential must be greater than that indicated by Figure 1. It can be seen that if the sulfur dioxide concentration of the gas phase is limited to 10 mol '30, the equilibrium reducing potential for reaction 7 will be less than that for reaction 4 as long as the temperature of the system is greater th ...
JSUNIL TUTORIAL , SAMASTIPUR, BIHAR
... Here, 12 gm of carbon combine with 16g and 32gm of Oxygen to form Carbon monoxide and Carbon dioxide respectively. The ratio of oxygen combining with 12 gm of Carbon is 16: 32 or, 1:2 which is in a simple ratio Dalton’s explanation for the law of conservation of mass and the law of definite proporti ...
... Here, 12 gm of carbon combine with 16g and 32gm of Oxygen to form Carbon monoxide and Carbon dioxide respectively. The ratio of oxygen combining with 12 gm of Carbon is 16: 32 or, 1:2 which is in a simple ratio Dalton’s explanation for the law of conservation of mass and the law of definite proporti ...
www.fahadsacademy.com
... achieve an inert gas configuration, forming ions. Ionic bonds are formed between METALLIC and NON- METALLIC ATOMS ONLY. - Metals lose electrons to form positive ions (cations) - Non-metals gain electrons to form negative ions (anions) The formation of ions is resulted from transfer of atoms from one ...
... achieve an inert gas configuration, forming ions. Ionic bonds are formed between METALLIC and NON- METALLIC ATOMS ONLY. - Metals lose electrons to form positive ions (cations) - Non-metals gain electrons to form negative ions (anions) The formation of ions is resulted from transfer of atoms from one ...
Chapter 12
... ratio, but now we also use molar mass to get to grams Example: How many grams of chlorine are required to react completely with 5.00 moles of sodium to produce sodium chloride? 2 Na + Cl2 2 NaCl 5.00 moles Na 1 mol Cl2 70.90g Cl2 = 177g Cl2 2 mol Na 1 mol Cl2 ...
... ratio, but now we also use molar mass to get to grams Example: How many grams of chlorine are required to react completely with 5.00 moles of sodium to produce sodium chloride? 2 Na + Cl2 2 NaCl 5.00 moles Na 1 mol Cl2 70.90g Cl2 = 177g Cl2 2 mol Na 1 mol Cl2 ...
The structure and mass of atoms - Brentwood Ursuline Convent
... 2 Sometimes other reactions can take place as well the reaction you want. For example, when methane is burned not all of it reacts to form carbon dioxide because other reactions make carbon monoxide or carbon (soot) instead. When you bake a cake, some of the ingredients get left in the bowl or on th ...
... 2 Sometimes other reactions can take place as well the reaction you want. For example, when methane is burned not all of it reacts to form carbon dioxide because other reactions make carbon monoxide or carbon (soot) instead. When you bake a cake, some of the ingredients get left in the bowl or on th ...
Unit 3: Bonding and Nomenclature Content Outline: Chemical
... b. This is the amount of energy released when one mole of an ionic crystalline compound is formed from gaseous ions. B. The electrical charges are balanced, so as to be neutral. C. Ionic compounds are hard but brittle (crumbly). D. In the solid state, they cannot conduct electricity (as the atoms ca ...
... b. This is the amount of energy released when one mole of an ionic crystalline compound is formed from gaseous ions. B. The electrical charges are balanced, so as to be neutral. C. Ionic compounds are hard but brittle (crumbly). D. In the solid state, they cannot conduct electricity (as the atoms ca ...
Ch 3
... A fuel mixture used in the early days of rocketry is composed of two liquids, hydrazine(N2H4) and dinitrogen tetraoxide(N2O4), which ignite on contact to form nitrogen gas and water vapor. How many grams of nitrogen gas form when 1.00x102g of N2H4 and 2.00x102g of N2O4 are mixed? ...
... A fuel mixture used in the early days of rocketry is composed of two liquids, hydrazine(N2H4) and dinitrogen tetraoxide(N2O4), which ignite on contact to form nitrogen gas and water vapor. How many grams of nitrogen gas form when 1.00x102g of N2H4 and 2.00x102g of N2O4 are mixed? ...
Introduction to Computational Chemistry
... from the province of a small nucleus of theoretical work to a large, significant component of scientific research. By virtue of the great flexibility and power of electronic computers, basic principles of classical and quantum mechanics are now implemented in a form which can handle the many-body pr ...
... from the province of a small nucleus of theoretical work to a large, significant component of scientific research. By virtue of the great flexibility and power of electronic computers, basic principles of classical and quantum mechanics are now implemented in a form which can handle the many-body pr ...
Chemistry(I) Final Exam 1/11/2008
... (b) According to Charles’s law, lower temperature causes smaller volume of a gas. The smallest volume of an ideal gas can have is 0. So the temperature at which an ideal gas reach zero volume is the absolute zero. ...
... (b) According to Charles’s law, lower temperature causes smaller volume of a gas. The smallest volume of an ideal gas can have is 0. So the temperature at which an ideal gas reach zero volume is the absolute zero. ...
Chapter 4 2013
... total solution volume (not 1 L of liquid!). (3) Note: It does not mean 3.5 moles of FeCl3 is dissolved in 1.00 liter of water! (4) [Fe3+] = 3.5M and [Cl-] = 3 x 3.5 M (5) It can be used as a conversion factor ...
... total solution volume (not 1 L of liquid!). (3) Note: It does not mean 3.5 moles of FeCl3 is dissolved in 1.00 liter of water! (4) [Fe3+] = 3.5M and [Cl-] = 3 x 3.5 M (5) It can be used as a conversion factor ...
SAT - mvhs-fuhsd.org
... separate from each other to form a liquid. The ions are loosely held together by the oppositely charged ions, but the ions are moving too fast for the crystal lattice to stay together. ...
... separate from each other to form a liquid. The ions are loosely held together by the oppositely charged ions, but the ions are moving too fast for the crystal lattice to stay together. ...
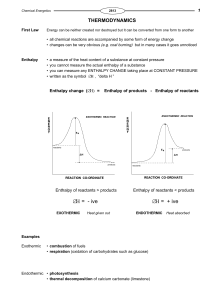
Thermodynamics - Shailendra Kumar Chemistry
... Which of the following statements concerning the change in ∆G° and ∆G during a chemical reaction is most correct? a. ∆G° remains constant while ∆G changes and becomes equal to ∆G° at equilibrium. b. Both ∆G° and ∆G remain constant during a chemical reaction. c. Initially both ∆G and ∆G° are equal to ...
... Which of the following statements concerning the change in ∆G° and ∆G during a chemical reaction is most correct? a. ∆G° remains constant while ∆G changes and becomes equal to ∆G° at equilibrium. b. Both ∆G° and ∆G remain constant during a chemical reaction. c. Initially both ∆G and ∆G° are equal to ...
Ratio and Proportion
... one ratio is equal to another ratio. Ex: a ratio of 4:8 = a ratio of 3:6 ...
... one ratio is equal to another ratio. Ex: a ratio of 4:8 = a ratio of 3:6 ...
Chapter 04
... electrons are transferred from one reactant to another. Oxidation is the loss of electrons. Reduction is the gain of electrons. Zn metal loses 2 electrons and is oxidized to Zn2+ Zn2+ is called the reducing agent ...
... electrons are transferred from one reactant to another. Oxidation is the loss of electrons. Reduction is the gain of electrons. Zn metal loses 2 electrons and is oxidized to Zn2+ Zn2+ is called the reducing agent ...
Deans Community High School Intermediate 2 Revision Notes www
... Uses of catalyst. As we have seen, reactions are more likely to take place when high concentrations, large surface areas and high temperatures are used. These factors increase the likelihood of collisions of the reactants, and the more energy that these collision have, the more likely it will be tha ...
... Uses of catalyst. As we have seen, reactions are more likely to take place when high concentrations, large surface areas and high temperatures are used. These factors increase the likelihood of collisions of the reactants, and the more energy that these collision have, the more likely it will be tha ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.