File
... continues. These 2 processes continue at the same rate (speed), therefore no net change in concentration occurs and the solution is in “dynamic equilibrium”. The rate of dissolving = the rate of recrystallization ...
... continues. These 2 processes continue at the same rate (speed), therefore no net change in concentration occurs and the solution is in “dynamic equilibrium”. The rate of dissolving = the rate of recrystallization ...
Document
... When lumps of calcium carbonate react with hydrochloric acid, carbon dioxide gas is released. CaCO3(s) ...
... When lumps of calcium carbonate react with hydrochloric acid, carbon dioxide gas is released. CaCO3(s) ...
AP Chemistry Summer Packet ANSWERS
... 4.0 mL 10.25 mL 15 mL 2. A student performed an analysis of a sample for its calcium content and got the following results: 14.92%, 14.91%, 14.88%, and 14.91% The actual amount of calcium in the sample is 15.70%. What conclusion can you draw about the accuracy and precision of these results? ...
... 4.0 mL 10.25 mL 15 mL 2. A student performed an analysis of a sample for its calcium content and got the following results: 14.92%, 14.91%, 14.88%, and 14.91% The actual amount of calcium in the sample is 15.70%. What conclusion can you draw about the accuracy and precision of these results? ...
Here are the answers and work for your summer packet.
... 4.0 mL 10.25 mL 15 mL 2. A student performed an analysis of a sample for its calcium content and got the following results: 14.92%, 14.91%, 14.88%, and 14.91% The actual amount of calcium in the sample is 15.70%. What conclusion can you draw about the accuracy and precision of these results? ...
... 4.0 mL 10.25 mL 15 mL 2. A student performed an analysis of a sample for its calcium content and got the following results: 14.92%, 14.91%, 14.88%, and 14.91% The actual amount of calcium in the sample is 15.70%. What conclusion can you draw about the accuracy and precision of these results? ...
GHW - Louisiana Tech University
... Why do we need the concept of isotopes describing an element? How average atomic masse is calculated from isotopic masses? In chemistry, why is the concept of mole central to standard measurement of amount of substance? Why are amount of substances measured in moles when they react according to the ...
... Why do we need the concept of isotopes describing an element? How average atomic masse is calculated from isotopic masses? In chemistry, why is the concept of mole central to standard measurement of amount of substance? Why are amount of substances measured in moles when they react according to the ...
1. Review (MC problems, due Monday) 2. - mvhs
... 3. A solution of barium hydroxide is titrated with 0.1-M sulfuric acid and the electrical conductivity of the solution is measured as the titration proceeds. a) For the reaction that occurs during the titration described above, write a balanced net ionic equation. (b) Explain why the conductivity de ...
... 3. A solution of barium hydroxide is titrated with 0.1-M sulfuric acid and the electrical conductivity of the solution is measured as the titration proceeds. a) For the reaction that occurs during the titration described above, write a balanced net ionic equation. (b) Explain why the conductivity de ...
Chapter 1 Chemistry: the study of the composition of matter and the
... Conversion questions. For example, how many grams in 27.4 kilograms? Significant figures a. Count them, round to a specific significant figure, add/subtract/multiply/divide Write in scientific notation Is it accurate? Precise? What is the percent error? Be able to solve for the missing value in a de ...
... Conversion questions. For example, how many grams in 27.4 kilograms? Significant figures a. Count them, round to a specific significant figure, add/subtract/multiply/divide Write in scientific notation Is it accurate? Precise? What is the percent error? Be able to solve for the missing value in a de ...
Reactions Balancing Chemical Equations uses Law of conservation
... Gas formation is a driving force. • Direct production of a gas CO2, H2S, NO2, SO2 • Production of weak acid which decomposes. ...
... Gas formation is a driving force. • Direct production of a gas CO2, H2S, NO2, SO2 • Production of weak acid which decomposes. ...
1. You should review balancing equations and identifying types of
... b. the reaction of sodium bicarbonate with sulfuric acid to produce sodium sulfate, water, and carbon dioxide 2NaHCO3 +2H2SO4 Na2SO4 + 2H2O+ 2CO2 c. the treatment of phosphorus pentachloride with water to produce phosphoric acid and hydrogen chloride PCl5 + 4H2O H3PO4 +5HCl ...
... b. the reaction of sodium bicarbonate with sulfuric acid to produce sodium sulfate, water, and carbon dioxide 2NaHCO3 +2H2SO4 Na2SO4 + 2H2O+ 2CO2 c. the treatment of phosphorus pentachloride with water to produce phosphoric acid and hydrogen chloride PCl5 + 4H2O H3PO4 +5HCl ...
FREE Sample Here
... C) molecules move from reactants to products and back. D) molecules are rearranged to form new molecules. E) small molecules are assembled into larger ones. 71) In an equilibrium reaction A) increasing the amount of one of the products will increase the amount of reactants available. B) decreasing t ...
... C) molecules move from reactants to products and back. D) molecules are rearranged to form new molecules. E) small molecules are assembled into larger ones. 71) In an equilibrium reaction A) increasing the amount of one of the products will increase the amount of reactants available. B) decreasing t ...
Homework,1 Atoms, molecules, and ions
... 6- Calculate the mass percent of nitrogen in HNO3. a) 45.2% b) 25.0% c) 22.2% d) 20.0% e) none of these 7- You are setting up a reaction between two chemicals that react according to the equation 3 A + 4 B products. If you start with 1.00 mole each of both A and B, which chemical will be in exces ...
... 6- Calculate the mass percent of nitrogen in HNO3. a) 45.2% b) 25.0% c) 22.2% d) 20.0% e) none of these 7- You are setting up a reaction between two chemicals that react according to the equation 3 A + 4 B products. If you start with 1.00 mole each of both A and B, which chemical will be in exces ...
Chapter One
... each element in the compound (percent composition), and vice versa. When solving this kind of problems, we can use masses expressed in a.m.u. or in g/mol ...
... each element in the compound (percent composition), and vice versa. When solving this kind of problems, we can use masses expressed in a.m.u. or in g/mol ...
in a Chemical Reactor - Max-Planck
... particularly mutually soluble. However, what they do form under certain conditions is an azeotrope – a mixture in which the two components evaporate at the same temperature. For this reason, they cannot be separated through the process of distillation. We need to control the process correctly to ena ...
... particularly mutually soluble. However, what they do form under certain conditions is an azeotrope – a mixture in which the two components evaporate at the same temperature. For this reason, they cannot be separated through the process of distillation. We need to control the process correctly to ena ...
Review - cloudfront.net
... d. 653 g When two substances react to form products, the reactant which is used up is called the ____. a. determining reagent c. excess reagent b. limiting reagent d. catalytic reagent Why does the pressure inside a container of gas increase if more gas is added to the container? a. There is an incr ...
... d. 653 g When two substances react to form products, the reactant which is used up is called the ____. a. determining reagent c. excess reagent b. limiting reagent d. catalytic reagent Why does the pressure inside a container of gas increase if more gas is added to the container? a. There is an incr ...
College Chemistry I PHS 1025 Fall 2012 Practice Exam 3A
... containing 500. g of water, the temperature of the water increases by 8.63°C. Assuming that the specific heat of water is 4.18 J/(g ∙ °C), and that the heat absorption by the calorimeter is negligible, estimate the enthalpy of combustion per mole of anthracene. A) -7070 kJ/mol B) +39.7 kJ/mol C) -81 ...
... containing 500. g of water, the temperature of the water increases by 8.63°C. Assuming that the specific heat of water is 4.18 J/(g ∙ °C), and that the heat absorption by the calorimeter is negligible, estimate the enthalpy of combustion per mole of anthracene. A) -7070 kJ/mol B) +39.7 kJ/mol C) -81 ...
Unit 4 - Calculations and Chemical Reactions
... Notice that the K+ and NO3- and ions don’t undergo chemical changes. They are in the exact same form on both sides of the equation. Ions that don’t undergo a chemical change during a chemical reaction are called spectator ions. If we omit the spectator ions, we will have the net ionic equation: Ag+( ...
... Notice that the K+ and NO3- and ions don’t undergo chemical changes. They are in the exact same form on both sides of the equation. Ions that don’t undergo a chemical change during a chemical reaction are called spectator ions. If we omit the spectator ions, we will have the net ionic equation: Ag+( ...
Summer Work
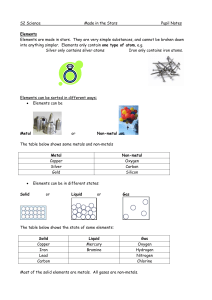
... that element. The atomic number gives the “identity “ of an element as well as its location on the Periodic Table. 5. No two different elements will have the ______________________ atomic number. 6. The ______________________ of an element is the average mass of an element’s naturally occurring atom ...
... that element. The atomic number gives the “identity “ of an element as well as its location on the Periodic Table. 5. No two different elements will have the ______________________ atomic number. 6. The ______________________ of an element is the average mass of an element’s naturally occurring atom ...
enjoy learning - System Dynamics Society
... The volume of the produced gas should be as large as possible. How can the unit mass of the explosive produce as large volume of gas as possible? First, change the reactants into gases as much as possible; second, the amount of the gas(es) production material should be as large as possible, that is, ...
... The volume of the produced gas should be as large as possible. How can the unit mass of the explosive produce as large volume of gas as possible? First, change the reactants into gases as much as possible; second, the amount of the gas(es) production material should be as large as possible, that is, ...
Final Exam Practice-2017
... 92. What is the element that is reduced in the following reaction? Br2 (g) + 2HI (aq) 2HBr (aq) + I2 (l) a) Br b) H c) I 93. Which of the following is the correct balanced half reaction for I2O5 I2 in a basic solution? a) 10H+ + I2O5 + 5e- I2 + 5H2O c) 5H2O + I2O5 + 5e- I2 + 10 OHb) 10H+ + I ...
... 92. What is the element that is reduced in the following reaction? Br2 (g) + 2HI (aq) 2HBr (aq) + I2 (l) a) Br b) H c) I 93. Which of the following is the correct balanced half reaction for I2O5 I2 in a basic solution? a) 10H+ + I2O5 + 5e- I2 + 5H2O c) 5H2O + I2O5 + 5e- I2 + 10 OHb) 10H+ + I ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.