A Motivation of Quantum Mechanics
... This is most easily understood as follows: The time evolution of a single wave is Re(A1/2 (x)eiωt ) with some complex amplitude A1/2 (x). The intensity for this wave is given by I1/2 (x) = |A1/2 (x)eiωt |2 . But for waves, it’s not the intensities that add but the (complex) amplitudes: A12 (x) = A1 ...
... This is most easily understood as follows: The time evolution of a single wave is Re(A1/2 (x)eiωt ) with some complex amplitude A1/2 (x). The intensity for this wave is given by I1/2 (x) = |A1/2 (x)eiωt |2 . But for waves, it’s not the intensities that add but the (complex) amplitudes: A12 (x) = A1 ...
X PS EM - deo kadapa
... Units: C.G.S cal / g°c; M.K.S units – J / Kg – °k (2) Why does ice float on water? Ans:- The density of ice is less than that of water so ice floats on water. (3) Convert 35°c into Kelvin scale? Ans:- Temperature of Kelvin = 273 + Temperature in Celsius .. . Temperature in kelvin scale = 273 + 35 = ...
... Units: C.G.S cal / g°c; M.K.S units – J / Kg – °k (2) Why does ice float on water? Ans:- The density of ice is less than that of water so ice floats on water. (3) Convert 35°c into Kelvin scale? Ans:- Temperature of Kelvin = 273 + Temperature in Celsius .. . Temperature in kelvin scale = 273 + 35 = ...
Chapter 2. Electronic, Vibrational and Spin
... on the wavefunction Ψ (e.g., operation of the Hamiltonian operator, H, on Ψ produces the allowed energies). The operator is related to some measurable property of a system (energy, dipole moment, bond angle, angular momentum, transition probability, etc.) and generally has a form similar to the math ...
... on the wavefunction Ψ (e.g., operation of the Hamiltonian operator, H, on Ψ produces the allowed energies). The operator is related to some measurable property of a system (energy, dipole moment, bond angle, angular momentum, transition probability, etc.) and generally has a form similar to the math ...
Chapter 8 and 9
... Lysine is an amino acid which has the following elemental composition: C, H, O, N. In one experiment, 2.175 g of lysine was combusted to produce 3.94 g of CO2 and 1.89 g H2O. In a separate experiment, 1.873 g of lysine was burned to produce 0.436 g of NH2. The molar mass of lysine is 150 g/mol. Dete ...
... Lysine is an amino acid which has the following elemental composition: C, H, O, N. In one experiment, 2.175 g of lysine was combusted to produce 3.94 g of CO2 and 1.89 g H2O. In a separate experiment, 1.873 g of lysine was burned to produce 0.436 g of NH2. The molar mass of lysine is 150 g/mol. Dete ...
Quantum defect theory description of weakly bound levels and Feshbach...
... representation, a full N × N smooth reaction matrix is obtained for the system, and after closed channel elimination, Fano–Feshbach resonances emerge at various energies and magnetic field strengths B. (The closedchannel elimination step simply imposes correct exponentially decaying boundary conditio ...
... representation, a full N × N smooth reaction matrix is obtained for the system, and after closed channel elimination, Fano–Feshbach resonances emerge at various energies and magnetic field strengths B. (The closedchannel elimination step simply imposes correct exponentially decaying boundary conditio ...
Time reversal and the symplectic symmetry of the electron spin.
... superconductors, allowing a description of the development of valence bonds in a spin fluid, and the transmission of their pair correlations to the electron sea[20, 21]. For the first time, this physics can now be explored within a controlled large N approach. As an example of this physics in action ...
... superconductors, allowing a description of the development of valence bonds in a spin fluid, and the transmission of their pair correlations to the electron sea[20, 21]. For the first time, this physics can now be explored within a controlled large N approach. As an example of this physics in action ...
Revision IB2 Topic 1
... Explain why the stream of hydrogen gas was maintained until the apparatus cooled. ...
... Explain why the stream of hydrogen gas was maintained until the apparatus cooled. ...
Quantum Mechanics and Solid State Physics for Electric
... electrical engineers: the differential and integral calculus. Although during the semester we introduce the basics of operator calculus, to understand that part only elementary algebra is required. The material in this book is organized in three distinct parts: Quantum Mechanics, Solid State Physics ...
... electrical engineers: the differential and integral calculus. Although during the semester we introduce the basics of operator calculus, to understand that part only elementary algebra is required. The material in this book is organized in three distinct parts: Quantum Mechanics, Solid State Physics ...
Chemistry Olympiad Support Booklet
... use their calculator properly to determine the H+ concentration as 3.7 x 10–3 mol dm–3. (e) The percentage of HCOOH molecules ionising is therefore: ...
... use their calculator properly to determine the H+ concentration as 3.7 x 10–3 mol dm–3. (e) The percentage of HCOOH molecules ionising is therefore: ...
DEPARTMENT OF CHEMISTRY, CFS, IIUM
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
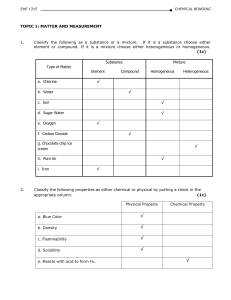
STUDY MATERIAL 2015-16 CHEMISTRY CLASS XI
... This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical endeavour to help students learn more effectively and efficiently. It is designed to give proper platform to students for better practice and understanding of the chapters. This can suitably be used during revisio ...
... This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical endeavour to help students learn more effectively and efficiently. It is designed to give proper platform to students for better practice and understanding of the chapters. This can suitably be used during revisio ...
A Formal Cause Beyond Space and Time
... Hence, the spectral lines of a hydrogen atom are manifestations of the gaps between two possible orbits of the electron. We can also conclude from the equation that h is the cause of such gap, for when h approaches 0 the numerical difference between the two energy states vanishes and they become a c ...
... Hence, the spectral lines of a hydrogen atom are manifestations of the gaps between two possible orbits of the electron. We can also conclude from the equation that h is the cause of such gap, for when h approaches 0 the numerical difference between the two energy states vanishes and they become a c ...
Measuring Quantum Entanglement
... it increases under Local Operations and Classical Communication ...
... it increases under Local Operations and Classical Communication ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.