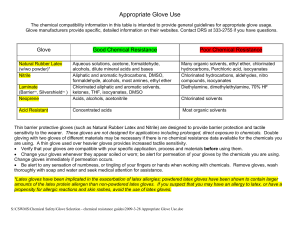
Appropriate Glove Use
... sensitivity to the wearer. These gloves are not designed for applications including prolonged, direct exposure to chemicals. Double gloving with two gloves of different materials may be necessary if there is no chemical resistance data available for the chemicals you are using. A thin glove used ove ...
... sensitivity to the wearer. These gloves are not designed for applications including prolonged, direct exposure to chemicals. Double gloving with two gloves of different materials may be necessary if there is no chemical resistance data available for the chemicals you are using. A thin glove used ove ...
3.4 Laboratory Chemical Storage and Inventory Proper storage of
... Proper storage of chemicals in laboratories is always a major concern. Chemicals that have been stored improperly could react, forming hazardous products. Sometimes improper storage results only in disorder, but at other times, it has resulted in loss of life and property. Each department is respons ...
... Proper storage of chemicals in laboratories is always a major concern. Chemicals that have been stored improperly could react, forming hazardous products. Sometimes improper storage results only in disorder, but at other times, it has resulted in loss of life and property. Each department is respons ...
chemical reaction
... Chemical Equations, continued • How to Balance an Equation To balance an equation, you must use coefficients. A coefficient is a number that is placed in front of a chemical symbol or formula. • For an equation to be balanced, all atoms must be counted. So, you multiply the subscript of each element ...
... Chemical Equations, continued • How to Balance an Equation To balance an equation, you must use coefficients. A coefficient is a number that is placed in front of a chemical symbol or formula. • For an equation to be balanced, all atoms must be counted. So, you multiply the subscript of each element ...
Describing Chemical Reactions
... Chemical equations use chemical formulas and other symbols instead of words to summarize a reaction. All chemical equations have a common structure. A chemical equation tells you the substances you start with in a reaction and the substances you get at the end. The substances you have at the beginni ...
... Chemical equations use chemical formulas and other symbols instead of words to summarize a reaction. All chemical equations have a common structure. A chemical equation tells you the substances you start with in a reaction and the substances you get at the end. The substances you have at the beginni ...
21:3 Classifying Chemical Reactions
... 21:1 Chemical Changes Writing Equations: chemical equations use chemical formulas and symbols to describe a chemical reaction and the products it produces. Chemical formulas express the relationship between elements in the compound and ...
... 21:1 Chemical Changes Writing Equations: chemical equations use chemical formulas and symbols to describe a chemical reaction and the products it produces. Chemical formulas express the relationship between elements in the compound and ...
How to balance chemical equations.
... •The law of conservation of matter states that matter can not be created or destroyed. •In any reaction, you must have the same amount of each part before and after the reaction happens. •We show how reactions become balanced by using chemical equations. ...
... •The law of conservation of matter states that matter can not be created or destroyed. •In any reaction, you must have the same amount of each part before and after the reaction happens. •We show how reactions become balanced by using chemical equations. ...
Chemical Properties - Michigan State University
... or when you observe rusting or rotting. The process that produces a chemical change is known as a chemical reaction. A chemical reaction is composed of reactants and products. Many chemical reactions are irreversible. The chemical reaction only goes one way. There are many ways to recognize a chemic ...
... or when you observe rusting or rotting. The process that produces a chemical change is known as a chemical reaction. A chemical reaction is composed of reactants and products. Many chemical reactions are irreversible. The chemical reaction only goes one way. There are many ways to recognize a chemic ...
Chemistry
... 19. Score at least 75% on a standard ACS exam for a one semester biochemistry course. 20. Correctly use and care for all lab-ware and instruments common to an undergraduate biochemistry laboratory. 21. Understand an MSDS for any compound. 22. Follow all safety rules, actions, protocols with respect ...
... 19. Score at least 75% on a standard ACS exam for a one semester biochemistry course. 20. Correctly use and care for all lab-ware and instruments common to an undergraduate biochemistry laboratory. 21. Understand an MSDS for any compound. 22. Follow all safety rules, actions, protocols with respect ...
Ductility-the ability to be stretched into wires
... Does the paper change its chemistry (chemical identity) and form a new substance with different properties when it is burned? Is the ability to burn a physical or chemical property? Chemical Property: A property that can only be tested/observed by changing the chemical identity of a substance; ...
... Does the paper change its chemistry (chemical identity) and form a new substance with different properties when it is burned? Is the ability to burn a physical or chemical property? Chemical Property: A property that can only be tested/observed by changing the chemical identity of a substance; ...
Section 2 Chemical Formulas and Equations
... Figure 4 Like chemical symbols, the symbols on this musical score are understood around the world! Describing Reactions by Using Equations In the same way that composers use musical symbols, chemists around the world use chemical symbols and chemical formulas. Chemists use chemical equations to des ...
... Figure 4 Like chemical symbols, the symbols on this musical score are understood around the world! Describing Reactions by Using Equations In the same way that composers use musical symbols, chemists around the world use chemical symbols and chemical formulas. Chemists use chemical equations to des ...
Section 1 Forming New Substances Chapter 9
... Reactants and products are separated by putting an arrow between them to show the direction of the reaction. Reactions at equilibrium will have arrows facing both directions. Apply the Law of Conservation of Mass to get the same number of atoms of every element on each side of the equation. Tip: Sta ...
... Reactants and products are separated by putting an arrow between them to show the direction of the reaction. Reactions at equilibrium will have arrows facing both directions. Apply the Law of Conservation of Mass to get the same number of atoms of every element on each side of the equation. Tip: Sta ...
What is a property?
... If the substance is still the same substance after it has been changed… It’s a Physical Change. Example… Boiling water Boiling Water becomes Water vapor Water = water ...
... If the substance is still the same substance after it has been changed… It’s a Physical Change. Example… Boiling water Boiling Water becomes Water vapor Water = water ...
Experiment #5 WHERE`S THE EVIDENCE
... A physical property is a characteristic of a substance that can be observed without changing the substance into another substance. The temperature at which a solid melts is a physical property. Color, hardness, and texture are other physical properties. A chemical property is a characteristic of a s ...
... A physical property is a characteristic of a substance that can be observed without changing the substance into another substance. The temperature at which a solid melts is a physical property. Color, hardness, and texture are other physical properties. A chemical property is a characteristic of a s ...
What are Physical Properties and Changes? - Mamanakis
... The formation of a gas is a clue to chemical changes. The bubbles of gas that you observed form when an antacid is dropped into water is an example of change. Another clue that a chemical change has occurred is the formation of a solid. A solid that separates out of solution during a chemical change ...
... The formation of a gas is a clue to chemical changes. The bubbles of gas that you observed form when an antacid is dropped into water is an example of change. Another clue that a chemical change has occurred is the formation of a solid. A solid that separates out of solution during a chemical change ...
Physical and Chemical Properties worksheet
... Your team will investigate the properties of 4 different unknowns. Record observations in the columns, be as specific as possible. When testing with the four liquids, use your straw-scoop to place a small amount in the well, and add a few mL of liquid with the pipette and stir with a toothpick. When ...
... Your team will investigate the properties of 4 different unknowns. Record observations in the columns, be as specific as possible. When testing with the four liquids, use your straw-scoop to place a small amount in the well, and add a few mL of liquid with the pipette and stir with a toothpick. When ...
Holt Chemistry – Guided Notes, Chapter 1
... A _______________ is any substance that has a definite composition. A ___________ _______________ is the process by which one or more substances change to produce one or more new substances. The ___________ of ______________ are the physical forms of matter which are solid, liquid, gas, and plasma. ...
... A _______________ is any substance that has a definite composition. A ___________ _______________ is the process by which one or more substances change to produce one or more new substances. The ___________ of ______________ are the physical forms of matter which are solid, liquid, gas, and plasma. ...
Physical properties
... • Distillation is the process of heating a liquid until it boils, capturing and cooling the resultant hot vapors, and collecting the condensed vapors. • Mankind has applied the principles of distillation for thousands of years. Distillation was probably first used by ancient Arab chemists to isolate ...
... • Distillation is the process of heating a liquid until it boils, capturing and cooling the resultant hot vapors, and collecting the condensed vapors. • Mankind has applied the principles of distillation for thousands of years. Distillation was probably first used by ancient Arab chemists to isolate ...
(the products). Mass is conserved in a chemical reaction
... and obey the Law of Conservation of Mass. • Chemical equations are used to describe chemical reactions. • There are six common types of chemical reactions. ...
... and obey the Law of Conservation of Mass. • Chemical equations are used to describe chemical reactions. • There are six common types of chemical reactions. ...
Chemical Equations
... Chemical Equations In this lesson, you will go from chemical formulas to chemical equations, a required step in stoichiometry. If you have trouble writing chemical formulas, please review that concept before continuing here. ...
... Chemical Equations In this lesson, you will go from chemical formulas to chemical equations, a required step in stoichiometry. If you have trouble writing chemical formulas, please review that concept before continuing here. ...
balanced - drseemaljelani
... Balancing Chemical Equations The numbers multiplying chemical formulas in a chemical equation are called: Stoichiometric Coefficients (S.C.) 2H2 + O2 → 2H2O Balanced ...
... Balancing Chemical Equations The numbers multiplying chemical formulas in a chemical equation are called: Stoichiometric Coefficients (S.C.) 2H2 + O2 → 2H2O Balanced ...
Unit 1 Matter Day 32 2016 Counting Atoms
... same mass after the reaction as you do before the reaction. This means… *The # and type of atoms are the same in the reactants and products (just in different ...
... same mass after the reaction as you do before the reaction. This means… *The # and type of atoms are the same in the reactants and products (just in different ...
chemical reaction?
... • What is an exothermic reaction? – A chemical reaction in which energy is released to the surroundings – Exothermic reactions often feel __________ because energy is released as heat – An example of an exothermic reaction is _______________ ...
... • What is an exothermic reaction? – A chemical reaction in which energy is released to the surroundings – Exothermic reactions often feel __________ because energy is released as heat – An example of an exothermic reaction is _______________ ...
Destruction of Syria's chemical weapons

The destruction of Syria's chemical weapons began with several international agreements that were arrived at with Syria, with an initial destruction deadline of 30 June 2014. United Nations Security Council Resolution 2118 imposed on Syria responsibilities and a timeline for the destruction of its chemical weapons and chemical weapons production facilities. The Security Council resolution incorporated and bound Syria to an implementation plan enacted in an Organisation for the Prohibition of Chemical Weapons (OPCW) Executive Council Decision. On 23 June 2014, the last declared chemical weapons were shipped out of Syria for destruction. The destruction of the most dangerous chemical weapons began at sea aboard the Maritime Administration Ready Reserve Force vessel CAPE RAY crewed with U.S. civilian merchant mariners. It took 42 days aboard ship to destroy 600 metric tons of chemical agents that would have been used to make deadly Sarin and Mustard Gas.The impetus toward destroying Syria's chemical weapons began with a 9 September 2013 rhetorical suggestion by U.S. Secretary of State John Kerry that Syria turn over all of its chemical weapons within a week. At the time, the U.S. and France headed a coalition of countries on the verge of carrying out air strikes on Syria in response to the 21 August 2013 Ghouta attacks. The suggestion received a positive response from Russia and Syria, and U.S.–Russian negotiations led to the 14 September 2013 ""Framework for Elimination of Syrian Chemical Weapons,"" which calls for the elimination of Syria's chemical weapon stockpiles by mid-2014. Following the agreement, Syria acceded to the Chemical Weapons Convention and agreed to apply that convention provisionally until its entry into force on 14 October 2013. On 21 September, Syria ostensibly provided a list of its chemical weapons to the OPCW, before the deadline set by the framework.On 27 September, the Executive Council of the OPCW adopted a decision, ""Destruction of Syrian Chemical Weapons,"" a detailed implementation plan based on the U.S./Russian agreement. Later on 27 September, the UN Security Council unanimously passed United Nations Security Council Resolution 2118, incorporating the OPCW plan and making it binding on Syria. A joint OPCW-UN mission will supervise the destruction or removal of Syria's chemical arms, while its Director-General is charged with notifying the Executive Council regarding any delay in implementation. The Executive Council would decide whether the non-compliance should be reported to the Security Council, which is responsible for making certain Syria fulfills its commitments under Resolution 2118.OPCW began preliminary inspections of Syria's chemical weapons arsenal on 1 October 2013, and actual destruction began on 6 October. Under OPCW supervision Syrian military personnel began ""destroying munitions such as missile warheads and aerial bombs and disabling mobile and static mixing and filling units."" The destruction of Syria's declared chemical weapons production, mixing, and filling equipment was successfully completed by 31 October deadline. The destruction of the chemical weapons fell well behind schedule. The entire chemical weapons stockpile had been scheduled to be completely removed from the country by 6 February 2014. Only on 23 June 2014, had Syria finished shipping the remaining declared chemicals. On 18 August 2014, all of the most toxic chemicals had been destroyed offshore. Western officials such as British Ambassador Mark Lyall Grant have expressed concerns about the completeness of Syria's disclosures, and believe the OPCW mission should remain in place following the removal of chemical weapons until verification tasks can be completed.Chlorine, a common industrial chemical, is outside the scope of the disarmament agreement; however, its use as a poison gas would violate the Chemical Weapons Convention, which Syria joined in 2013. Various parties, including Western governments, have accused Assad of conducting illegal chlorine attacks in 2014 and 2015. A late disclosure in 2014 regarding Syria's ricin program raised doubts about completeness of the government's declaration of its chemical weapons stockpile, and in early May 2015, OPCW announced that inspectors had found traces of sarin and VX nerve agent at a military research site in Syria that had not been declared previously by the Assad regime.