* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Pauli Exclusion Principle
Matter wave wikipedia , lookup
Density functional theory wikipedia , lookup
James Franck wikipedia , lookup
Nitrogen-vacancy center wikipedia , lookup
Renormalization group wikipedia , lookup
Dirac equation wikipedia , lookup
Franck–Condon principle wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Wave–particle duality wikipedia , lookup
Ferromagnetism wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Chemical bond wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Tight binding wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Auger electron spectroscopy wikipedia , lookup
Atomic orbital wikipedia , lookup
Electron-beam lithography wikipedia , lookup
Hydrogen atom wikipedia , lookup
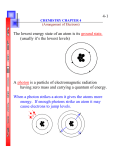
Pauli Exclusion Principle No more than one electron may occupy a given quantum state specified by a particular set of singleparticle quantum numbers n, l, ml ms. ground state Ground State of Atoms H : 1s1 He: 1s2 (filled shell n=1) Na: 1s2 2s2 2p6 3s1 Mg: 1s2 2s2 2p6 3s2 Li: 1s2 2s1 Al: 1s2 2s2 2p6 3s2 3p1 Be: 1s2 2s2 Si : 1s2 2s2 2p6 3s2 3p2 B : 1s2 2s2 2p1 P: 1s2 2s2 2p6 3s2 3p3 C : 1s2 2s2 2p2 S: 1s2 2s2 2p6 3s2 3p4 N: 1s2 2s2 2p3 O: 1s2 2s2 2p4 F: 1s2 2s2 2p5 Ne: 1s2 2s2 2p6 (filled shell n=2) Cl: 1s2 2s2 2p6 3s2 3p5 Ar: 1s2 2s2 2p6 3s2 3p6 K: 1s2 2s2 2p6 3s2 3p6 4s1 7-46. Write the ground-state electron configuration of (a) carbon, (b) oxygen, and (c) argon. C : 1s2 2s2 2p2 O: 1s2 2s2 2p4 Ar: 1s2 2s2 2p6 3s2 3p6 7-51. What elements have these ground-state electron configurations? (a) 1s22s22p63s23p2 and (b) 1s22s22p63s23p64s2? a) 7-49. If the 3s electron in sodium did not penetrate the inner core, its energy would be -13.6 eV/32 = -1.51 eV. Because it does penetrate, it sees a higher effective Z and its energy is lower. Use the measured ionization potential of 5.14 V to calculate Zeff for the 3s electron in sodium. 7-72 (a) Show that the function is a solution of Equation 7-9, where A is a constant and a0 is the Bohr radius. (b) Find the constant A. 7-72 (a) Show that the function is a solution of Equation 7-9, where A is a constant and a0 is the Bohr radius. (b) Find the constant A. 7-72 (a) Show that the function is a solution of Equation 7-9, where A is a constant and a0 is the Bohr radius. (b) Find the constant A. 7-72 (a) Show that the function is a solution of Equation 7-9, where A is a constant and a0 is the Bohr radius. (b) Find the constant A. 7-65 Show that the expectation value of r for the electron in the ground state of a one electron atom is <r > = 3/2 <a0>/Z. Change of variable Excited States and Spectra of Alkali Atoms - An excited state of the atom usually involves a change in the state of one of the electrons or, more rarely, two or even more electrons. -Even in the case of the excitation of only one electron, the change in state of this electron changes the energies of the others. -This effect is negligible, and the energy levels can be calculated accurately from a relatively simple model of one electron plus a stable core for the alkali metals: Li, Na, K, Rb, and Cs -Similar to H Na: Ne 3s1 Na: 1s2 2s2 2p6 3s1 outermost electron 3s 2p electrons 1s electrons Transitions between occupied levels are prohibited by Pauli principle Only outermost electrons are involved Excited States and Spectra of Alkali Atoms Na: 1s2 2s2 2p6 3s1 Ground state Excited states Na: Ne 3s1 7-31. The optical spectra of atoms with two electrons in the same outer shell are similar, but they are quite different from the spectra of atoms with just one outer electron because of the interaction of the two electrons. Separate the following elements into two groups such that those in each group have similar spectra: lithium, beryllium, sodium, magnesium, potassium, calcium, chromium, nickel, cesium, and barium. lithium, sodium, potassium, chromium, cesium, (one electron in the outermost shell) 7-55. Which of the following elements should have optical spectra similar to that of hydrogen and which should have optical spectra similar to that of helium: Li, Ca, Ti, Rb, Ag, Cd, Ba, Hg, Fr, Ra? Li, Rb, Ag, Fr (similar to H) Spin-orbit coupling Atomic states with the same n and l ,but different j have slightly different energies 7-49. Which of the following atoms would you expect to have its ground state split by the spin-orbit interaction: Li, B, Na, Al, K, Ag, Cu, Ga? (Hint: Use Appendix C to see which elements have / = 0 in their ground state and which do not.) Li: 1s2 2s1 Na: 1s2 2s2 2p6 3s1 B : 1s2 2s2 2p1 Al: 1s2 2s2 2p6 3s2 3p1 Ga: 1s2 2s2 2p6 3s2 3p6 3d104s2 4p1 K: 1s2 2s2 2p6 3s2 3p6 4s1 Ag: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s1 Cu: 1s2 2s2 2p6 3s2 3p6 3d10 4s1 7-59. Since the P states and the D states of sodium are all doublets, there are four possible energies for transitions between these states. Indicate which three transitions are allowed and which one is not allowed by the selection rule of Equation 7-65. 7-62 Show that the change in wavelength Dl of a transition due to a small change in energy is (Hint: Differentiate E = hc/λ) 7-69 The wavelengths of the photons emitted by potassium corresponding to transitions from the 4P3/2 and 4P1/2 states to the ground state are 766.41 nm and 769.90 nm. (a) Calculate the energies of these photons in electron volts. (b) The difference in energies of these photons equals the difference in energy ΔE between the 4P3/2 and 4P1/2 states in potassium. Calculate ΔE . (c) Estimate the magnetic field that the 4p electron in potassium experiences. a) b) c) 7-74 If relativistic effects are ignored, the n = 3 level for one-electron atoms consists of the 32S1/2, 32P1/2, 32P3/2, 32D3/2, and 32D5/2 states. Compute the spin-orbit-effect splittings of 3P and 3D states for hydrogen. Thomas precession 7-74 If relativistic effects are ignored, the n = 3 level for one-electron atoms consists of the 32S1/2, 32P1/2, 32P3/2, 32D3/2, and 32D5/2 states. Compute the spin-orbit-effect splittings of 3P and 3D states for hydrogen. 7-74 If relativistic effects are ignored, the n = 3 level for one-electron atoms consists of the 32S1/2, 32P1/2, 32P3/2, 32D3/2, and 32D5/2 states. Compute the spin-orbit-effect splittings of 3P and 3D states for hydrogen. Zeeman effect When an atom is placed in an external magnetic field B, the total angular momentum J is quantized in space relative to the direction of B and the energy of the atomic state characterized by the angular momentum quantum number j is split into 2j + 1 energy levels corresponding to the 2j +1 possible values of the z component of J and therefore to the 2j +1 possible values of the z component of the total magnetic moment. Lamb shift energy level fluctuations of the vacuum 7-41. The Lamb shift energy difference between the 22S1/2 and 22P1/2 levels in atomic hydrogen is 4.372 x 10-6 eV. (a) What is the frequency of the photon emitted in this transition? (b) What is the photon’s wavelength? (c) In what part of the electromagnetic spectrum does this transition lie? a) b)