* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chemical Equations & Reactions
Destruction of Syria's chemical weapons wikipedia , lookup
Double layer forces wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Asymmetric induction wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Isotopic labeling wikipedia , lookup
Computational chemistry wikipedia , lookup
Chemical bond wikipedia , lookup
Drug discovery wikipedia , lookup
Fine chemical wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
California Green Chemistry Initiative wikipedia , lookup
Electrochemistry wikipedia , lookup
Atomic theory wikipedia , lookup
Al-Shifa pharmaceutical factory wikipedia , lookup
Process chemistry wikipedia , lookup
History of chemistry wikipedia , lookup
Chemical weapon proliferation wikipedia , lookup
Safety data sheet wikipedia , lookup
Chemical weapon wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Chemical potential wikipedia , lookup
Chemical Corps wikipedia , lookup
Click chemistry wikipedia , lookup
Chemical plant wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
Rate equation wikipedia , lookup
Chemical reaction wikipedia , lookup
Chemical industry wikipedia , lookup
George S. Hammond wikipedia , lookup
Chemical equilibrium wikipedia , lookup
History of molecular theory wikipedia , lookup
Transition state theory wikipedia , lookup
VX (nerve agent) wikipedia , lookup
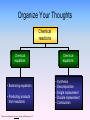
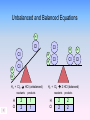
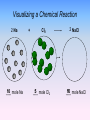
Chemical Equations & Reactions Chapter 8 Lesson 1 Chemical Reactions You should be able to Classify reactions by type. Write a balanced molecular equation, complete ionic equation, and a net ionic equation. Balance oxidation-reduction reactions. Predict if a precipitate will form using the solubility rules. Predict products of reactions given the chemical names of the reactants. Organize Your Thoughts Chemical reactions Chemical equations • Balancing equations • Predicting products from reactants Packard, Jacobs, Marshall, Chemistry Pearson AGS Globe, page 175 Chemical equations • Synthesis • Decomposition • Single replacement • Double replacement • Combustion Describing a Chemical Reaction Indications of a Chemical Reaction – Evolution of heat, light, and/or sound – Production of a gas – Formation of a precipitate – Color change Signs of Chemical Reactions There are five main signs that indicate a chemical reaction has taken place: release input change in color change in odor production of new gases or vapor input or release of energy difficult to reverse Chemical Equations aluminum oxide Depict the kind of reactants and product products and their relative amounts in a reaction. 4 Al(s) + 3 O2(g) 2 Al2O3(s) The letters (s), (g), and (l) are the physical states of compounds. The numbers in the front are called stoichiometric coefficients. Chemical Equations 4 Al(s) + 3 O2(g) 2 Al2O3(s) aluminum oxide sandpaper 4 g Al + 3 g O2 yield 2 g Al2O3 This equation means: 4 Al atoms + 3 O2 molecules yield 2 molecules of Al2O3 or 4 Al moles + 3 O2 moles yield 2 moles of Al2O3 4 mol Al@27g/mol 108 g 3 mol O2@32g/mol + 96 g 2 mol Al2O3@102g/mol = 204 g Chemical Equations Because the same atoms are present in a reaction at the beginning (reactants) and at the end (products), the amount of matter in a system does not change. The Law of Conservation of Matter 100% Kotz web Chemical Factory 100% 20% 80% Chemical Equations Because of the principle of the conservation of matter, An equation must be balanced. It must have the same number of atoms of the same kind on both sides. Lavoisier, 1788 Characteristics of Chemical Equations • The equation must represent known facts. • The equation must contain the correct formulas for the reactants and products. • The law of conservation of mass must be satisfied. Chemical Equations • Reactants – the substances that exist before a chemical change (or reaction) takes place. • Products – the new substance(s) that are formed during the chemical changes. • CHEMICAL EQUATION indicates the reactants and products of a reaction. REACTANTS PRODUCTS Word Equations • A WORD EQUATION describes chemical change using the names of the reactants and products. Write the word equation for the reaction of methane gas with oxygen gas to form carbon dioxide and water. methane + oxygen Reactant CH4 + 2 O2 carbon dioxide + water Product CO2 + 2 H2O Unbalanced and Balanced Equations H Cl Cl H H H H Cl H2 + Cl2 HCl (unbalanced) reactants H Cl 2 2 H H Cl Cl Cl H2 + Cl2 2 HCl (balanced) reactants products 1 1 Cl H Cl 2 2 products 2 2 Visualizing a Chemical Reaction 2 Na 10 mole Na ___ + Cl2 5 mole Cl2 ___ 2 NaCl 10 ? mole NaCl ___ Visualizing a Chemical Reaction 2 Na + Cl2 2 NaCl Meaning of Chemical Formula Chemical Symbol Meaning Composition H2O One molecule of water: Two H atoms and one O atom 2 H2O Two molecules of water: Four H atoms and two O atoms H 2 O2 One molecule of hydrogen peroxide: Two H atoms and two O atoms Balancing Chemical Equations Balanced Equation – one in which the number of atoms of each element as a reactant is equal to the number of atoms of that element as a product What is the relationship between conservation of mass and the fact that a balanced equation will always have the same number of atoms of each element on both sides of an equation? Determine whether the following equation is balanced. 2 Na + H2O 2 NaOH + H2 2 Na + 2 H2O 2 NaOH + H2 Balancing Chemical Equations • Write a word equation for the reaction. • Write the correct formulas for all reactants and products. • Determine the coefficients that make the equation balance. Balancing Chemical Equations An important point to remember 2 NO(g) + O2(g) 2NO2(g) The 2 to the left of NO(g) and NO2(g) refers to the number of molecules present in the balanced equation. It is a “multiplier” for every atom in the molecule. The subscript 2 in O2 (g) and NO2(g) refers to the number of atoms of this type that are present in each molecules (or ionic compound). Showing Phases in Chemical Equations H2O(s) H2O(l) H2O(g) Solid Phase – the substance is relatively rigid and has a definite volume and shape. NaCl(s) Liquid Phase – the substance has a definite volume, but is able to change shape by flowing. H2O(l) Gaseous Phase – the substance has no definite volume or shape, and it shows little response to gravity. Cl2(g) Additional Symbols Used in Chemical Equations “Yields”; indicates result of reaction Used to indicate a reversible reaction (s) A reactant or product in the solid state; also used to indicate a precipitate Alternative to (s), but used only to indicate a precipitate (l) A reactant or product in the liquid state (aq) A reactant or product in an aqueous solution (dissolved in water) (g) A reactant or product in the gaseous state Additional Symbols Used in Chemical Equations Alternative to (g), but used only to indicate a gaseous product D 2 atm pressure Reactants are heated Pressure at which reaction is carried out, in this case 2 atm Pressure at which reaction is carried out exceeds normal atmospheric pressure 0 oC Temperature at which reaction is carried out, in this case 0 oC MnO2 Formula of catalyst, in this case manganese (IV) oxide, used to alter the rate of the reaction Solubility Ionic Equations Cover the answers, work the problem, then check the answer. 1. Dissolve ammonium nitrate: NH4NO3 (s) ---> NH4+1 (aq) + NO3-1 (aq) 2. Precipitate cupric hydroxide: Cu+2 (aq) + 2OH-1 (aq) ---> Cu(OH)2 (s) 3. Dissolve chromium thiocyanate: 4. Precipitate lead arsenate: Cr(SCN)3 (s) ---> Cr+3 (aq) + 3SCN-1 (aq) 3Pb+2 (aq) + 2AsO4-3 (aq) ---> Pb3(AsO4)2 (s) 5. Dissolve silicon permanganate: Si(MnO4)4 (s) ---> Si+4 (aq) + 4MnO4-1 (aq) 6. Precipitate zinc phosphate: 3Zn+2 (aq) + 2PO4-3 (aq) ---> Zn3(PO4)2 (s)