EVS - RSC - Developments in Microwave Chemistry
... chemical synthesis, whereas multi-mode microwave equipment is mainly used for chemical analysis. This equipment has been limited to laboratories due to lack of scalability of the technology. Presently, the manufacturers are directing their research to develop products that can increase the yield vol ...
... chemical synthesis, whereas multi-mode microwave equipment is mainly used for chemical analysis. This equipment has been limited to laboratories due to lack of scalability of the technology. Presently, the manufacturers are directing their research to develop products that can increase the yield vol ...
Chemical Transport Model - Technical Description
... comprises general statements based on scientific research. The reader is advised and needs to be aware that such information may be incomplete or unable to be used in any specific situation. No reliance or actions must therefore be made on that information without seeking prior expert professional, ...
... comprises general statements based on scientific research. The reader is advised and needs to be aware that such information may be incomplete or unable to be used in any specific situation. No reliance or actions must therefore be made on that information without seeking prior expert professional, ...
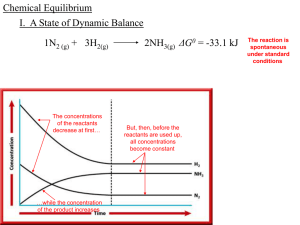
Chemical Equilibrium - local.brookings.k12.sd.us
... A. Safety: The surfaces of the hot plates and the water will be hot enough to cause burns. Use caution. Cobalt(II) chloride hexahydrate is toxic, with an LD50 = 80mg/kg Avoid ingestion (don’t eat or drink it). Wash hands thoroughly with soap and water before leaving lab. Ethanol is extremely flammab ...
... A. Safety: The surfaces of the hot plates and the water will be hot enough to cause burns. Use caution. Cobalt(II) chloride hexahydrate is toxic, with an LD50 = 80mg/kg Avoid ingestion (don’t eat or drink it). Wash hands thoroughly with soap and water before leaving lab. Ethanol is extremely flammab ...
High Performance Plastics for the Chemical Processing Industry
... Depending on the polymerization process however, polyethylene can be manufactured with different densities. The different densities results from the number of cross branches between the main molecular chains. Further, the greater the number and length of the branches between these chains the lower t ...
... Depending on the polymerization process however, polyethylene can be manufactured with different densities. The different densities results from the number of cross branches between the main molecular chains. Further, the greater the number and length of the branches between these chains the lower t ...
Chemical Equilibrium
... completion. Indeed, our stoichiometric calculations were based on this; when we asked how much of a product is produced when so much of a reactant reacts, we are assuming that all of a reactant reacts. However, this is usually not the case; many reactions do not go to completion, and many chemists h ...
... completion. Indeed, our stoichiometric calculations were based on this; when we asked how much of a product is produced when so much of a reactant reacts, we are assuming that all of a reactant reacts. However, this is usually not the case; many reactions do not go to completion, and many chemists h ...
Chemical Equilibrium - 2012 Book Archive
... where [A] is the molar concentration of species A at equilibrium, and so forth. The coefficients a, b, c, and d in the chemical equation become exponents in the expression for Keq. The Keq is a characteristic numerical value for a given reaction at a given temperature; that is, each chemical reactio ...
... where [A] is the molar concentration of species A at equilibrium, and so forth. The coefficients a, b, c, and d in the chemical equation become exponents in the expression for Keq. The Keq is a characteristic numerical value for a given reaction at a given temperature; that is, each chemical reactio ...
Chemical Reactions - 2012 Book Archive
... antibiotics such as amoxicillin, were unknown only a few years ago. Their development required that chemists understand how substances combine in certain ratios and under specific conditions to produce a new substance with particular properties. ...
... antibiotics such as amoxicillin, were unknown only a few years ago. Their development required that chemists understand how substances combine in certain ratios and under specific conditions to produce a new substance with particular properties. ...
DEPARTMENT OF CHEMISTRY Requirements For Chemistry Major
... The course for Bachelor of Science (Industrial Chemistry) will run for three academic years (6 semesters) and will follow the format offered by the Department of Chemistry. Year I: Semester I (Core Courses) ...
... The course for Bachelor of Science (Industrial Chemistry) will run for three academic years (6 semesters) and will follow the format offered by the Department of Chemistry. Year I: Semester I (Core Courses) ...
Quantitative Comparison of the Hydrogen Bond
... FIGURE 1: Hydrogen bond topologies as derived by detected h3JNC′ H-bond correlations for ubiquitin in the native state (A) and in the A-state (B). Top: two-dimensional maps representing the detected h3JNC′ scalar correlations (filled diamonds). Secondary structure elements are indicated as filled ar ...
... FIGURE 1: Hydrogen bond topologies as derived by detected h3JNC′ H-bond correlations for ubiquitin in the native state (A) and in the A-state (B). Top: two-dimensional maps representing the detected h3JNC′ scalar correlations (filled diamonds). Secondary structure elements are indicated as filled ar ...
Chapter 5: Calculations and the Chemical Equation
... conservation of matter -- in a chemical reaction matter can neither be created nor destroyed. From Dalton's atomic theory we know that all substances are composed of atoms. During a chemical reaction atoms may be combined, separated, or rearranged, but not created or destroyed. Equations Must Be Bal ...
... conservation of matter -- in a chemical reaction matter can neither be created nor destroyed. From Dalton's atomic theory we know that all substances are composed of atoms. During a chemical reaction atoms may be combined, separated, or rearranged, but not created or destroyed. Equations Must Be Bal ...
Chemical Reaction Equations
... zinc with an excess of lead(II) nitrate solution? Design: A known mass of zinc is place in a beaker with an excess of lead(II) nitrate solution. The lead is produced in the reaction is separated by filtration and dried. The mass of the lead is determined ...
... zinc with an excess of lead(II) nitrate solution? Design: A known mass of zinc is place in a beaker with an excess of lead(II) nitrate solution. The lead is produced in the reaction is separated by filtration and dried. The mass of the lead is determined ...
this page - Course Catalogs
... Department of Physics and Astronomy section of this catalog. A program of study emphasizing a strong background in chemistry is available for students planning a future in medicine. The Department of Chemistry is on the approved list of the American Chemical Society (ACS). Approval is received after ...
... Department of Physics and Astronomy section of this catalog. A program of study emphasizing a strong background in chemistry is available for students planning a future in medicine. The Department of Chemistry is on the approved list of the American Chemical Society (ACS). Approval is received after ...
Stoichiometry and the Mole
... involve relating a quantity of one substance to a quantity of another substance or substances. The relating of one chemical substance to another using a balanced chemical reaction is called stoichiometry. Using stoichiometry is a fundamental skill in chemistry; it greatly broadens your ability to pr ...
... involve relating a quantity of one substance to a quantity of another substance or substances. The relating of one chemical substance to another using a balanced chemical reaction is called stoichiometry. Using stoichiometry is a fundamental skill in chemistry; it greatly broadens your ability to pr ...
Subject Area Standard Area Organizing Category Grade Level
... CHEM.A.2.2.4: Relate the existence of quantized energy levels to atomic emission spectra. ...
... CHEM.A.2.2.4: Relate the existence of quantized energy levels to atomic emission spectra. ...
Slide 1
... • At any given temperature, density does not change. • No matter how much or how little C2H5OH is present, its concentration remains constant. • Therefore, the term in the denominator is a constant and can be combined with K. ...
... • At any given temperature, density does not change. • No matter how much or how little C2H5OH is present, its concentration remains constant. • Therefore, the term in the denominator is a constant and can be combined with K. ...
Stoichiometry and the Mole - 2012 Book Archive
... molar masses3 to emphasize the fact that they are the mass for 1 mol of things. (The term molar is the adjective form of mole and has nothing to do with teeth.) Here are some examples. The mass of a hydrogen atom is 1.0079 u; the mass of 1 mol of hydrogen atoms is 1.0079 g. Elemental hydrogen exists ...
... molar masses3 to emphasize the fact that they are the mass for 1 mol of things. (The term molar is the adjective form of mole and has nothing to do with teeth.) Here are some examples. The mass of a hydrogen atom is 1.0079 u; the mass of 1 mol of hydrogen atoms is 1.0079 g. Elemental hydrogen exists ...
Chapter 3 Stoichiometry STOICHIOMETRY: The chemical arithmetic
... • For NUMBER of Atoms • Or NUMBER of Molecules • 32 Grams of Oxygen = ? Molecules • 32 Grams of Oxygen = ? Atoms • 1 Molecule of Oxygen = ? Grams ...
... • For NUMBER of Atoms • Or NUMBER of Molecules • 32 Grams of Oxygen = ? Molecules • 32 Grams of Oxygen = ? Atoms • 1 Molecule of Oxygen = ? Grams ...
word - My eCoach
... C. It contains 6 oxygen atoms. D. It contains 1.204 x 1024 nitrate (NO3-) ions. 10) 9.03 x 1023 atoms of silver are placed on a balance. The balance should read A. 53.96 g B. 107.87g C. 161.81g D. 215.74g Part 2. Free Response. Answer the questions as comprehensively as you can. Make sure that calcu ...
... C. It contains 6 oxygen atoms. D. It contains 1.204 x 1024 nitrate (NO3-) ions. 10) 9.03 x 1023 atoms of silver are placed on a balance. The balance should read A. 53.96 g B. 107.87g C. 161.81g D. 215.74g Part 2. Free Response. Answer the questions as comprehensively as you can. Make sure that calcu ...
Topic 3: Chemical Kinetics - Manitoba Education and Training
... Demonstrate appropriate scientific skills when seeking answers to questions. Demonstrate curiosity, skepticism, creativity, open-mindedness, accuracy, precision, honesty, and persistence, and appreciate their importance as scientific and technological habits of mind. Understand the properties and st ...
... Demonstrate appropriate scientific skills when seeking answers to questions. Demonstrate curiosity, skepticism, creativity, open-mindedness, accuracy, precision, honesty, and persistence, and appreciate their importance as scientific and technological habits of mind. Understand the properties and st ...
U3 Student Workbook - The Connected Chemistry Curriculum
... of Mass to chemical equations by learning how to balance them. Following a teacher demonstration of the simulation and procedures, students will use the simulations to look at ten different reactions. In each of the reactions, students will create submicroscopic sketches and balance the chemical for ...
... of Mass to chemical equations by learning how to balance them. Following a teacher demonstration of the simulation and procedures, students will use the simulations to look at ten different reactions. In each of the reactions, students will create submicroscopic sketches and balance the chemical for ...
Chapter 8 - Chemical Equations and Reactions
... Sample Problem A Write word and formula equations for the chemical reaction that occurs when solid sodium oxide is added to water at room temperature and forms sodium hydroxide (dissolved in the water). Include symbols for physical states in the formula equation. Then balance the formula equation to ...
... Sample Problem A Write word and formula equations for the chemical reaction that occurs when solid sodium oxide is added to water at room temperature and forms sodium hydroxide (dissolved in the water). Include symbols for physical states in the formula equation. Then balance the formula equation to ...
Chapter 8 Notes
... Sample Problem A Write word and formula equations for the chemical reaction that occurs when solid sodium oxide is added to water at room temperature and forms sodium hydroxide (dissolved in the water). Include symbols for physical states in the formula equation. Then balance the formula equation to ...
... Sample Problem A Write word and formula equations for the chemical reaction that occurs when solid sodium oxide is added to water at room temperature and forms sodium hydroxide (dissolved in the water). Include symbols for physical states in the formula equation. Then balance the formula equation to ...
Chemistry: Percent Yield
... 17: 3.4e Equal volumes of gases at the same temperature and pressure contain an equal number of particles. 33: 3.2b Types of chemical reactions include synthesis, decomposition, single replacement, and double replacement 36: M1.1C – Use algebraic and geometric representations to describe and compare ...
... 17: 3.4e Equal volumes of gases at the same temperature and pressure contain an equal number of particles. 33: 3.2b Types of chemical reactions include synthesis, decomposition, single replacement, and double replacement 36: M1.1C – Use algebraic and geometric representations to describe and compare ...
Chemical Vapor Deposition (CVD)
... • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total pressure gas flows, etc.— materials with different properties can be grown. ...
... • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total pressure gas flows, etc.— materials with different properties can be grown. ...
Wilhelm Ostwald, the Father of Physical Chemistry
... The NO that is produced in this step can be recycled through the reaction. This process was patented by Ostwald in the year 1902 and it proved very useful for the Germans during the first World War in the production of explosives. Ostwald did not support the idea of Berzelius that there exists a cat ...
... The NO that is produced in this step can be recycled through the reaction. This process was patented by Ostwald in the year 1902 and it proved very useful for the Germans during the first World War in the production of explosives. Ostwald did not support the idea of Berzelius that there exists a cat ...
Chemical Corps

The Chemical Corps is the branch of the United States Army tasked with defending against chemical, biological, radiological, and nuclear (CBRN) weapons. The corps was founded as the Chemical Warfare Service (CWS) during World War I. Its name was changed to the Chemical Corps in 1946.