* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chapter 15
William Flynn Martin wikipedia , lookup
Potential energy wikipedia , lookup
Open energy system models wikipedia , lookup
Kinetic energy wikipedia , lookup
Energy storage wikipedia , lookup
Energy subsidies wikipedia , lookup
Low-Income Home Energy Assistance Program wikipedia , lookup
Public schemes for energy efficient refurbishment wikipedia , lookup
Regenerative brake wikipedia , lookup
100% renewable energy wikipedia , lookup
Zero-energy building wikipedia , lookup
Energy Charter Treaty wikipedia , lookup
Low-carbon economy wikipedia , lookup
World energy consumption wikipedia , lookup
Internal energy wikipedia , lookup
Energy policy of Australia wikipedia , lookup
Alternative energy wikipedia , lookup
Energy harvesting wikipedia , lookup
International Energy Agency wikipedia , lookup
Energy returned on energy invested wikipedia , lookup
Energy policy of the United Kingdom wikipedia , lookup
Energy efficiency in transport wikipedia , lookup
Life-cycle greenhouse-gas emissions of energy sources wikipedia , lookup
Distributed generation wikipedia , lookup
Energy policy of Finland wikipedia , lookup
Negawatt power wikipedia , lookup
Conservation of energy wikipedia , lookup
Energy in the United Kingdom wikipedia , lookup
Energy policy of the European Union wikipedia , lookup
United States energy law wikipedia , lookup
Energy efficiency in British housing wikipedia , lookup
Energy applications of nanotechnology wikipedia , lookup
Energy Independence and Security Act of 2007 wikipedia , lookup
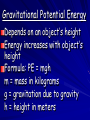
Chapter 15: Energy Energy • The ability to do work •Work is a transfer of energy •Metric unit Types of energy Joule (j) •Kinetic: moving •Potential: not moving Kinetic Energy The energy of motion 2 mv Formula: KE = ½ m = mass in kilograms v = speed in m/s Potential Energy The energy that is stored as a result of position or shape (not moving) 2 forms: 1. gravitational potential 2. elastic potential Gravitational Potential Energy Depends on an object’s height Energy increases with object’s height Formula: PE = mgh m = mass in kilograms g = gravitation due to gravity h = height in meters Elastic Potential Energy The stored energy of an object that is stretched or compressed Examples: spring rubber band Forms of Energy Mechanical Energy Thermal Energy Chemical Energy Electrical Energy Electromagnetic Energy Nuclear Energy Mechanical Energy Energy associated with motion Sum of an object’s potential energy and kinetic energy Examples: bouncing ball, speeding train, athletes Thermal Energy The energy of all of the microscopic particles in an object Sum of an atoms potential and kinetic energy Chemical Energy The energy that is stored in chemical bonds When the bonds are broken, the released energy can do work Examples: Burning wood Electrical Energy The energy associated with electrical charges Examples: flashlights, calculator, lightning Electromagnetic Energy The energy that travels through space in waves Examples: Visible light, x-rays Nuclear Energy The energy stored in an atom’s nucleus Most powerful energy Compare the KE of an 80kg man and a 30kg boy both moving at 4m/s. 2 KE=1/2 mv KE = ½ 80Kg(4m/s)2 = 80kg(16m2/s2) = 1280Kgm2/s2 = ½(1280Kgm2/s2 = 640J KE = ½ 30Kg(4m/s)2 = 30kg(16m2/s2) = 480Kgm2/s2 = ½(480Kgm2/s2 = 240J The man is producing the most KE. ex. How much PE does a 10kg ball have if its sitting on a 2m high shelf? PE=mgh Energy Conservation Energy converted from one form into another Examples: wind-up toy, match Conservation of Energy Law states: “Energy cannot be created or destroyed When energy changes forms, the total energy remains unchanged Energy Conversions Most common conversion is between PE to KE Examples: Pendulums, Pole Vaults Calculating Energy Conversion: (KE + PE)beginning = (KE + PE)end Practice on Page 458 Energy and Mass E = mc2 Energy and mass can be converted into each other Energy is released as matter is destroyed, and matter can be created from energy Nonrenewable Energy Resources • Resources that cannot be replaced as quickly as used. • AKA Fossil Fuels Once living organisms • ex. Coal, Petroleum (oil), natural gas, nuclear, etc. • usually produce pollution Renewable Energy Resources • Resources that are replaced as quickly as used. • usually environment safe •Examples include: hydroelectric, solar, geothermal, wind, and biomass Hydroelectric Energy Energy obtained from flowing water Kinetic energy is used to turn turbines and supply electricity Advantage: low cost and lack of pollution Solar Energy Sunlight that is converted into usable energy Solar energy depends on the climate Geothermal Energy Thermal energy beneath the Earth’s surface Not widely available Other Renewable Resources Biomass energy – chemical energy stored in living things Hydrogen fuel cell – generates electricity by reacting hydrogen with oxygen Conserving Energy Resources Reduce energy needs and increase the efficiency of energy use Energy Conservation – find ways to use less energy or to use energy more efficiently Conserving Energy • More energy is used in the U.S. than any other country! • Why? Money & Lifestyle • Where does it come from? 93% Nonrenewable resources 7% Renewable resources Practice test on-line http://www.proprofs.com/quizschool/quizshow.php?title=EnergyPractice-Test&quesnum=1 http://www.cstephenmurray.com/onlinequizes/physics/workandenergy /potentialenergyproblems.htm