* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Carbohydrates & Lipids - mvhs
Survey
Document related concepts
Evolution of metal ions in biological systems wikipedia , lookup
Metalloprotein wikipedia , lookup
Citric acid cycle wikipedia , lookup
Butyric acid wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Biosynthesis wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Transcript
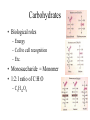
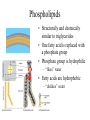
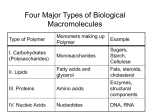
Biomolecules: Carbohydrates & Lipids Images taken without permission from http://www.poster.net/marciali/marciali-fresh-pasta-2404234.jpg and http://www.sciencemuseum.org.uk/exhibitions/lifecycle/images/1-2-6-2-1-0-0-0-0-0-0.jpg and http://www.worldroom.com/pages/health/healthyoil_feature.jpg Monomers and Polymers • Monomer = single unit – Examples: amino acids, monosaccharides • Polymer = made up of monomers – Examples: DNA, protein Images taken without permission from http://www.greenspirit.org.uk/resources/glucose.gi and http://www.nmslab.com/images/dna.jpg Dehydration Synthesis • Building reaction • H2O is removed in order to form a new bond Hydrolysis Reaction • Breaking reaction • H2O is required to break a bond “Structure-Function” Theme • Recurring theme on the AP exam • We will look at how the physical structures in biology support their specific function – Ex. How does the structure of the cell membrane support its function? Carbohydrates • Biological roles – Energy – Cell to cell recognition – Etc. • Monosaccharide = Monomer • 1:2:1 ratio of C:H:O – C6H12O6 Disaccharides • 2 monosaccharides linked together • Key disaccharides you need to know: – Fructose + glucose sucrose + H2O – Glucose + galactose lactose + H2O – Glucose + glucose maltose + H2O Image taken without permission from http://www.pp3moo.com/ and http://www.iktmc.edu.hk/subjects/hecon/milk.jpg Glycosidic linkage • The bond between monosaccharides • What type of reaction would form this bond? – Dehydration reaction • What kind of bond is a glycosidic linkage? – Polar covalent OH OH H2 C H2C O OH O C HO OH OH OH HO HO HO OH OH H2 C H2 C O OH HO O C OH O HO HO + H2O OH Polysaccharides: Starch & Glycogen • Both are used for energy storage – Starch = energy storage in plants – Glycogen = energy storage in animals • When energy is needed, enzymes break the bonds between monosaccharides Image taken without permission from http://www.dpiw.tas.gov.au/inter.nsf/Images/TTAR-5P549Z/$File/faf_potato_variety.jpg Structure of Starch & Glycogen • Branched structure – How does the branched structure support the function of these molecules? – branching allows for more points of access for enzymes to act (greater surface area) Image taken without permission from: http://wps.prenhall.com/wps/media/objects/724/741576/Instructor_Resources/Chapter_22/Text_Images/FG22_02.JPG Polysaccharides: Cellulose + Chitin • For structural support – Cellulose = structural support in plants – Chitin = makes up the hard exoskeleton of insects Structure of Cellulose & Chitin • Long, unbranched chains – Fibers form parallel chains – How would the structure of cellulose & chitin support their functions? – Long, unbranched chains provide greater strength Lipids • Biological roles: energy (storage), structural, protection, insulation, pigments • Mostly hydrocarbons (nonpolar, hydrophobic) – Lots of C-C and C-H bonds – How would this property support its function of energy storage? – Nonpolar quality allows for them to be stored without water– more compact http://www.matthewklein.com/adm/photo/9_Bread_PeanutButter.jpg Triglycerides: Fats & Oils glycerol • Made up of 1 glycerol and 3 fatty acids • What kind of reaction forms a triglyceride? – Dehydration reaction H O H C O C O H C O C O H C O C H Fatty acids Types of Fatty Acids • Fatty Acids can be saturated or unsaturated • Saturated Fatty Acid OH H2C – No double C-C bonds – Molecules are more straight – Fats: solid at room temperature • Unsaturated Fatty Acid H2C H2C H2C H2C H2C H2C CH2 H2C CH2 H2C CH2 H2C CH2 HC CH2 CH2 HC H2C H2C H2C H2C CH2 CH2 CH2 CH2 OH CH3 O C – Contain at least 1 C-C double bond – Causes “kinks” in molecule – Liquid at room temperature H2C CH2 CH2 H2C CH2 H2C CH2 OH H2C CH2 H2C CH2 CH3 O C H2C OH O C O C CH2 H2C CH2 H2C CH2 H2C CH2 CH2 CH2 CH2 CH3 HC HC CH2 CH2 CH2 CH2 CH2 CH2 H2C CH2 H2C CH2 H2C CH2 CH3 Fatty Acid Structure • Why would the type of fatty acid determine its state at room temperature? – Double bonds create the kinks in the structure can’t be packed as closely together less Van der waals forces – This makes them more fluid at room temperature lower melting temperature The peanut butter puzzle… • Fats are usually found in animals • Oils are usually found in plants • So why is peanut butter solid? – hydrogenation Images taken without permission from http://www.theunderweardrawer.homestead.com/files/peanut_butter.JPG and http://chinaproducts.supplierlist.com/manufacturers/36428/en_madeinchina.htm Phospholipids • Structurally and chemically similar to triglycerides • One fatty acid is replaced with a phosphate group • Phosphate group is hydrophilic – “likes” water • Fatty acids are hydrophobic – “dislikes” water Steroids • Chemically and structurally different from other lipid categories • Classified as a lipid because of their nonpolar/hydrophobic nature • Can be recognized by 6 carbon rings and 5 carbon rings • Chemical messengers (hormones) • Structural components in cell membranes cholesterol