* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chemistry -- Acids and Bases
Electrochemistry wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Electrolysis of water wikipedia , lookup
Peptide synthesis wikipedia , lookup
Liquid–liquid extraction wikipedia , lookup
Biological aspects of fluorine wikipedia , lookup
Citric acid cycle wikipedia , lookup
Metalloprotein wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Acid throwing wikipedia , lookup
Sulfuric acid wikipedia , lookup
Nitric acid wikipedia , lookup
Nitrocellulose wikipedia , lookup
Biosynthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Butyric acid wikipedia , lookup
Biochemistry wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
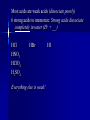
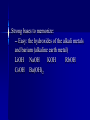
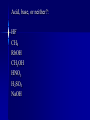
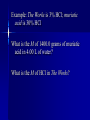
Chemistry Acid//Base Chemistry I. Introduction We’ve learned about: - formation - decomposition - combustion Now, we will learn about acid/base reactions II. Acids & Bases A. What is an acid? 1. Release a hydrogen ion (H+) in water (conduct electricity when added H2O) 2. Neutralize bases in a neutralization rxn 3. Corrode active metals (releasing H2 gas) 4. Turn blue litmus paper red 5. Taste sour Notes on the properties of acids… Don’t taste! Conduct electricity – ionic compounds conduct electricity -- Acids are special covalent compounds that conduct electricity when added to H2O Litmus --an indicator Indicator: a substance that turns one color in the presence of acids and another color in the presence of bases Most acids are weak acids (dissociate poorly) 6 strong acids to memorize: Strong acids dissociate completely in water (H+ + __-) HCl HNO3 HClO4 H2SO4 HBr Everything else is weak! HI Acetic v. HCl B. What is a base? 1. Release hydroxide ion in water 2. Neutralize acids in neutralization rxns 3. Denature proteins (feel slippery to the touch) 4. Bases turn red litmus paper blue 5. Taste bitter (Do NOT taste!) Strong bases to memorize: -- Easy: the hydroxides of the alkali metals and barium (alkaline earth metal) LiOH NaOH KOH RbOH CsOH Ba(OH)2 III. The Chemical Definition of Acids and Bases Acid: a molecule that donates H+ ion(s) Bases: a molecule that accepts H+ ion(s) For example: HCl + H2O → H3O+ + ClWhich is the acid and which is the base? Important note about H3O+ -- Called the hydronium ion -- Most often written as H+ -- As H+, just referred to as H+, hydrogen ion, or a proton -- H3O+ and H+ are the really the same thing and are used interchangeably Another acid/base reaction: H2O(l) + NH3(aq) → NH4+(aq) + OHWhich is the acid and which is the base? Water can be both an acid or a base – depending on what it reacts with Amphiprotic compounds: Compounds that can act as either an acid or a base Examples: Which is acid and which is base? H2SO4(aq) + CaCO3(aq) → Ca2+(aq) + HSO4-(aq) + HCO3-(aq) HCl(aq) + NaOH(aq)→H2O(l) + NaCl(aq) 2HNO3(aq) + Mg(OH)2(aq) → Mg(NO3)2(aq) + 2H2O(l) OYO’s Determine the acid and base in the following reactions: 10.1 HBr(aq) +NH3(aq)→NH4+(aq) + Br -(aq) 10.2 3C2H4O2(aq) +Al(OH)3 → Al(C2H3O2)3(aq) + 3H2O(l) 10.3 H3PO4(aq) + 3KOH(aq) → 3H2O(l) + K3PO4(aq) IV. Recognizing and Naming Acids and Bases from their Formulas A. Recognizing - General Trends (not strictly true): -- Acids start with “H” in the formula except C2H4O2 (aka CH3COOH and HC2H3O2) -- Bases that are ionic compounds and have “OH” in formula (2 notable exceptions: H2O and NH3) Acid, base, or neither?: HF CH4 RbOH CH3OH HNO3 H2SO4 NaOH B. Naming acids and bases 1. Acids -- ions that end in “-ide” hydro______ic acid (e.g., HCl) -- ions that end in “-ate” __________ic acid (e.g., H2SO4) -- ions that end in “-ite” __________ous acid (e.g., HNO2) 2. Bases -- Normal ionic naming rules (e.g., Sr(OH)2 – strontium hydroxide) V. Predicting the Reactions that Occur between Acids and Bases A. How many protons can an acid donate H3PO4 can donate up to 3 H+ -- “more than one” – “proton” Polyprotic acid: An acid that can donate more than one proton Triprotic acid: An acid that can donate up to 3 protons Diprotic acid: An acid that can donate up to 2 protons B. Acids and Ionic Bases Hydrofluoric acid and potassium hydroxide HF + KOH → ? Steps to solving: 1. Write acid and base as individual ions 2. Combine the negative acid ion with the positive base ion, put the H+ with the negative base ion 3. Balance (if necessary) Acid + Ionic base → Salt + water HCl + NaOH → NaCl + H2O (You think of “salt” and NaCl – but a salt is any ionic compound that formed because an ionic base reacted with an acid!) Examples: Hydrochloric acid and rubidium hydroxide Sulfuric acid and sodium hydroxide Nitric acid and aluminum hydroxide OYO’s 10.4 Give the chemical equation that represents the reaction between each of the following: a. HClO3 and KOH b. HBr and Ca(OH)2 c. H3PO4 and Mg(OH)2 Everyday example of acids and bases: Drain uncloggers contain base ; -- proteins contain amino acids (hair, food) Acid + Ionic base→ Salt + water All-purpose Cleaners contain bases -- most spills are acidic Toilet bowl cleaners (contain acid) --lime stains (CaO) are a common problem -- CaO reacts with water in toilet to become CaOH -- acid neutralizes B. Acids and Covalent Bases So…Acid + Ionic Base → Salt + water …but when an acid is reacts with a covalent base (and it always does – because it is in aqueous solution) – ions result H2SO4 + H2O → 2H3O+ + SO42Acid + Covalent Base → ions Examples: H3PO4 + 3NH3 → 3NH4+ + PO43HOCl + H2O → H3O+ + ClO- OYO’s 10.5 What is the chemical equation that describes the reaction between H2SO4 and ammonia (NH3)? 10.6 What reaction occurs between HI and water? V. Molarity (a.k.a. concentration) Density = g/mL Concentration in Chemistry: moles/L = Molarity (M) Example: The Works is 3% HCl; muriatic acid is 30% HCl What is the M of 1400.0 grams of muriatic acid in 4.00 L of water? What is the M of HCl in The Works? More Examples: What is the M of NaOH when 2.6 moles of NaOH are dissolved in 534 mL? 124.3 g of HNO3 are dissolved in 250.0mL. What is the M? OYO’s 10.7 Determine the concentration for the following acids and bases: A. 2.1 moles HCl in 346 mL solution B. 6.78 g KOH in 150.0 mL C. 20.1 g H2SO4 in 3.4 L VI. Dilution Chemists don’t usually measure grams of acid or base solution to make certain concentrations… They dilute! (Always add acid TO water – if you add water to acid, the acid may splash on you) Here’s the formula (memorize): C1V1 = C2V2 C1 = M of thing you are diluting V1 = Volume of initial thing (how much of it you will use (can be in mL or L – as long as V2 is measured in the same units) C2 = M you want in the end V2 = Final volume of new concentration (important: this is V1 + water to get to V2) water you add = V2 – V1 Examples: Chemist wants to dilute 12.0M HCl to get 500 mL of 3.5M HCl solution Chemist makes 2.4L of 5.6M NaOH and adds water to 10.0 L. New concentration? Dilution equation not only applies to acids and bases, but to all chemicals! OYO 10.8 Chemist needs 750.0 mL of 3.5M H2SO4. Has 10.0M H2SO4. Write out how you would do the dilution. VII. Using Concentration in Stoichiometry You can use the M of an acid or base to find out how many grams of product 2HCl + 2H2O 100 mL 6.18M Ba(OH)2 → excess BaCl2 + ?g Example: Sodium sulfate can be made by reacting H2SO4 with sodium hydroxide. How many grams of sodium sulfate can be produced if 500.0 mL of 1.6M sodium hydroxide is reacted with excess sulfuric acid? OYO 10.9 Barium hydroxide is often used to clean up toxic acid spills in industrial settings. If 10.0 L container of 12.0 M nitric acid is broken, how many grams of barium hydroxide will be needed to clean up the spill? VIII. Acid/Base Titrations Titration: The process of slowly reacting a base of unknown concentration with an acid of known concentration (or vice versa) until just enough acid has been added to react with all of the base (the endpoint has been reached). Just stoichiometry with acids and bases! OYO 10.10 125mL of nitric acid with unknown M is titrated against magnesium hydroxide with a concentration of 2.3 M. If 35.4 mL of base are required in order to reach the endpoint, what was the concentration of the acid?