* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Organic Chemistry Fifth Edition
Survey
Document related concepts
Aromaticity wikipedia , lookup
Asymmetric induction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Discodermolide wikipedia , lookup
George S. Hammond wikipedia , lookup
Ene reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Aromatization wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Hydroformylation wikipedia , lookup
Stille reaction wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Transcript
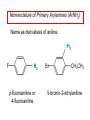
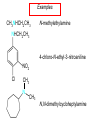
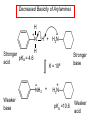
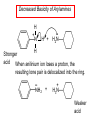
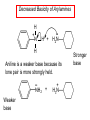
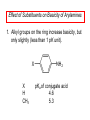
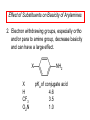
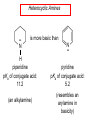
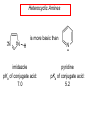
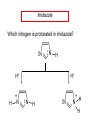
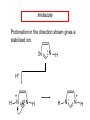
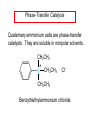
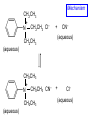
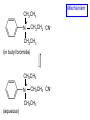
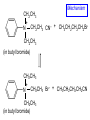
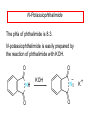
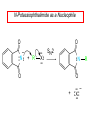
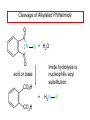
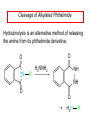
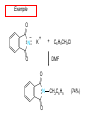
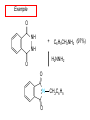
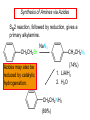
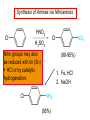
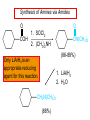
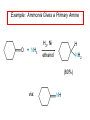
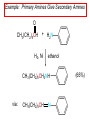
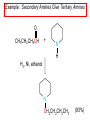
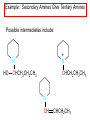
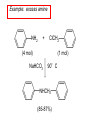
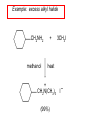
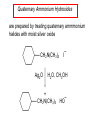
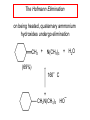
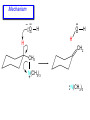
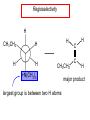
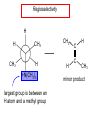
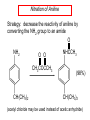
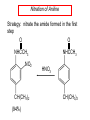
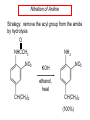
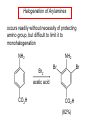
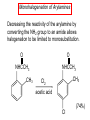
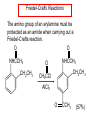
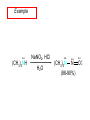
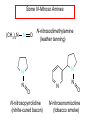
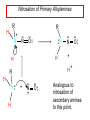
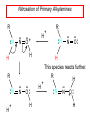
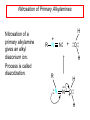
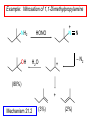
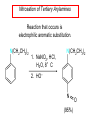
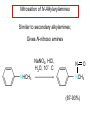
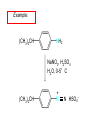
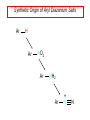
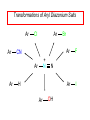
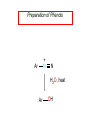
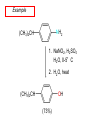
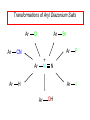
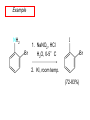
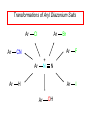
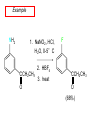
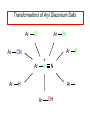
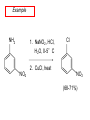
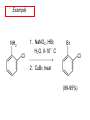
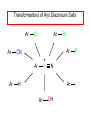
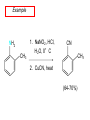
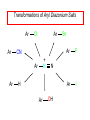
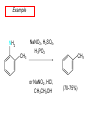
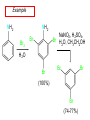
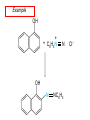
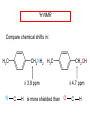
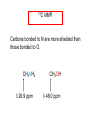
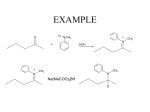
Chapter 21 Amines 21.1 Amine Nomenclature Classification of Amines Alkylamine N attached to alkyl group Arylamine N attached to aryl group Primary, secondary, or tertiary determined by number of carbon atoms directly attached to nitrogen Nomenclature of Primary Alkylamines (RNH2) Two IUPAC styles 1) Analogous to alcohols: replace -e ending with -amine 2) Name alkyl group and attach -amine as a suffix Examples: some primary alkylamines (RNH2: one carbon directly attached to N) ethylamine or ethanamine CH3CH2NH2 NH2 CH3CHCH2CH2CH3 NH2 cyclohexylamine or cyclohexanamine 1-methylbutylamine or 2-pentanamine or pentan-2-amine Nomenclature of Primary Arylamines (ArNH2) Name as derivatives of aniline. NH2 F NH2 p-fluoroaniline or 4-fluoroaniline Br CH2CH3 5-bromo-2-ethylaniline Amino Groups as Substituents Amino groups rank below OH groups and higher oxidation states of carbon. In such cases name the amino group as a substituent. O HOCH2CH2NH2 HC 2-aminoethanol p-aminobenzaldehyde NH2 Secondary and Tertiary Amines Name as N-substituted derivatives of parent primary amine. (N is a locant-it is not alphabetized, but is treated the same way as a numerical locant) Parent amine is one with longest carbon chain. Examples CH3NHCH2CH3 N-methylethylamine NHCH2CH3 4-chloro-N-ethyl-3-nitroaniline NO2 Cl CH3 N CH3 N,N-dimethylcycloheptylamine Ammonium Salts A nitrogen with four substituents is positively charged and is named as a derivative of ammonium ion (NH4+). + – CH3NH3 Cl methylammonium chloride CH3 + – N CH2CH3 CF3CO2 H N-ethyl-N-methylcyclopentylammonium trifluoroacetate Ammonium Salts When all four atoms attached to N are carbon, the ion is called a quaternary ammonium ion and salts that contain it are called quaternary ammonium salts. CH3 CH2 + N CH3 I – CH3 benzyltrimethylammonium iodide 21.2 Structure and Bonding Alkylamines 147 pm 112° 106° Alkylamines Most prominent feature is high electrostatic potential at nitrogen. Reactivity of nitrogen lone pair dominates properties of amines. Geometry at N Compare geometry at N of methylamine, aniline, and formamide. H H H sp3 sp2 NH2 C NH C 2 O H Pyramidal geometry at sp3-hybridized N in methylamine. Planar geometry at sp2-hybridized N in formamide. Geometry at N Compare geometry at N of methylamine, aniline, and formamide. sp3 sp2 Pyramidal geometry at sp3-hybridized N in methylamine. Planar geometry at sp2-hybridized N in formamide. Geometry at N Angle that the C—N bond makes with bisector of H—N—H angle is a measure of geometry at N. sp3 sp2 180° ~125° Note: This is not the same as the H—N—H bond angle. Geometry at N Angle that the C—N bond makes with bisector of H—N—H angle is a measure of geometry at N. sp2 sp3 180° ~125° 142.5° Geometry at N Geometry at N in aniline is pyramidal; closer to methylamine than to formamide. 142.5° Geometry at N Hybridization of N in aniline lies between sp3 and sp2. Lone pair of N can be delocalized into ring best if N is sp2 and lone pair is in a p orbital. Lone pair bound most strongly by N if pair is in an sp3 orbital of N, rather than p. Actual hybridization is a compromise that maximizes binding of lone pair. 142.5° Electrostatic Potential Maps of Aniline Nonplanar geometry at N. Region of highest negative potential is at N. Planar geometry at N. High negative potential shared by N and ring. Figure 21.2 (page 934) 21.3 Physical Properties Physical Properties Amines are more polar and have higher boiling points than alkanes; but are less polar and have lower boiling points than alcohols. CH3CH2CH3 CH3CH2NH2 CH3CH2OH dipole moment (): 0D 1.2 D 1.7 D boiling point: -42°C 17°C 78°C Physical Properties CH3CH2CH2NH2 CH3CH2NHCH3 boiling point: 50°C 34°C (CH3)3N 3°C Boiling points of isomeric amines decrease in going from primary to secondary to tertiary amines. Primary amines have two hydrogens on N capable of being involved in intermolecular hydrogen bonding. Secondary amines have one. Tertiary amines cannot be involved in intermolecular hydrogen bonds. 21.4 Basicity of Amines Effect of Structure on Basicity 1. Alkylamines are slightly stronger bases than ammonia. Table 21.1 Basicity of Amines in Aqueous Solution Amine Conj. Acid pKa NH3 NH4+ 9.3 CH3CH2NH2 CH3CH2NH3+ 10.8 CH3CH2NH3+ is a weaker acid than NH4+; therefore, CH3CH2NH2 is a stronger base than NH3. Effect of Structure on Basicity 1. Alkylamines are slightly stronger bases than ammonia. 2. Alkylamines differ very little in basicity. Table 21.1 Basicity of Amines in Aqueous Solution Amine Conj. Acid pKa NH3 NH4+ 9.3 CH3CH2NH2 CH3CH2NH3+ 10.8 (CH3CH2)2NH (CH3CH2)2NH2+ 11.1 (CH3CH2)3N (CH3CH2)3NH+ 10.8 Notice that the difference separating a primary, secondary, and tertiary amine is only 0.3 pK units. Effect of Structure on Basicity 1. Alkylamines are slightly stronger bases than ammonia. 2. Alkylamines differ very little in basicity. 3. Arylamines are much weaker bases than ammonia. Table 21.1 Basicity of Amines in Aqueous Solution Amine Conj. Acid pKa NH3 NH4+ 9.3 CH3CH2NH2 CH3CH2NH3+ 10.8 (CH3CH2)2NH (CH3CH2)2NH2+ 11.1 (CH3CH2)3N (CH3CH2)3NH+ 10.8 C6H5NH2 C6H5NH3+ 4.6 Decreased Basicity of Arylamines H + N H + H Stronger pKa = 4.6 acid •• NH2 + Weaker base •• H2N K = 106 Stronger base + H3N pKa =10.6 Weaker acid Decreased Basicity of Arylamines H + N H + •• H2N H Stronger acid When anilinium ion loses a proton, the resulting lone pair is delocalized into the ring. •• NH2 + + H3N Weaker acid Decreased Basicity of Arylamines H + N H + •• H2N H Aniline is a weaker base because its lone pair is more strongly held. •• NH2 + Weaker base + H3N Stronger base Decreased Basicity of Arylamines Increasing delocalization makes diphenylamine a weaker base than aniline, and triphenylamine a weaker base than diphenylamine. C6H5NH2 pKa of conjugate acid: 4.6 (C6H5)2NH (C6H5)3N 0.8 ~-5 Effect of Substituents on Basicity of Arylamines 1. Alkyl groups on the ring increase basicity, but only slightly (less than 1 pK unit). X X H CH3 NH2 pKa of conjugate acid 4.6 5.3 Effect of Substituents on Basicity of Arylamines 2. Electron withdrawing groups, especially ortho and/or para to amine group, decrease basicity and can have a large effect. X X H CF3 O2N NH2 pKa of conjugate acid 4.6 3.5 1.0 p-Nitroaniline – •• •• O •• •• O •• + N •• O •• – •• •• NH2 + N •• O •• – •• Lone pair on amine nitrogen is conjugated with p-nitro group—more delocalized than in aniline itself. Delocalization is lost on protonation. + NH2 Effect is Cumulative Aniline is 3800 times more basic than p-nitroaniline. Aniline is ~1,000,000,000 times more basic than 2,4-dinitroaniline. Heterocyclic Amines •• is more basic than N N H piperidine pyridine pKa of conjugate acid: 11.2 pKa of conjugate acid: 5.2 (an alkylamine) (resembles an arylamine in basicity) •• Heterocyclic Amines •• N •• N is more basic than H N •• imidazole pyridine pKa of conjugate acid: 7.0 pKa of conjugate acid: 5.2 Imidazole Which nitrogen is protonated in imidazole? •• N •• N H H+ + H N H+ •N • H •• N + H N H Imidazole Protonation in the direction shown gives a stabilized ion. •• N •• N H H+ + H N •N • H H N •• + N H 21.5 Tetraalkylammonium Salts as Phase-Transfer Catalysts Phase-Transfer Catalysis Phase-transfer agents promote the solubility of ionic substances in nonpolar solvents. They transfer the ionic substance from an aqueous phase to a non-aqueous one. Phase-transfer agents increase the rates of reactions involving anions. The anion is relatively unsolvated and very reactive in nonpolar media compared to water or alcohols. Phase-Transfer Catalysis Quaternary ammonium salts are phase-transfer catalysts. They are soluble in nonpolar solvents. H3C CH2CH2CH2CH2CH2CH2CH2CH3 + N CH2CH2CH2CH2CH2CH2CH2CH3 CH2CH2CH2CH2CH2CH2CH2CH3 Methyltrioctylammonium chloride Cl– Phase-Transfer Catalysis Quaternary ammonium salts are phase-transfer catalysts. They are soluble in nonpolar solvents. CH2CH3 + N CH2CH3 Cl– CH2CH3 Benzyltriethylammonium chloride Example The SN2 reaction of sodium cyanide with butyl bromide occurs much faster when benzyltriethylammonium chloride is present than when it is not. CH3CH2CH2CH2Br + NaCN benzyltriethylammonium chloride CH3CH2CH2CH2CN + NaBr Mechanism CH2CH3 + N CH2CH3 Cl– CH2CH3 CN– + (aqueous) (aqueous) CH2CH3 + N CH2CH3 CN– CH2CH3 (aqueous) + Cl– (aqueous) Mechanism CH2CH3 + N CH2CH3 CN– CH2CH3 (in butyl bromide) CH2CH3 + N CH2CH3 CN– CH2CH3 (aqueous) Mechanism CH2CH3 + N CH2CH3 CN– + CH3CH2CH2CH2Br CH2CH3 (in butyl bromide) CH2CH3 + N CH2CH3 Br– + CH3CH2CH2CH2CN CH2CH3 (in butyl bromide) 21.6 Reactions that Lead to Amines: A Review and a Preview Preparation of Amines Two questions to answer: 1) How is the C—N bond to be formed? 2) How do we obtain the correct oxidation state of nitrogen (and carbon)? Methods for C—N Bond Formation Nucleophilic substitution by azide ion (N3–) (Section 8.1, 8.11) Nitration of arenes (Section 12.3) Nucleophilic ring opening of epoxides by ammonia (Section 16.12) Nucleophilic addition of amines to aldehydes and ketones (Sections 17.10, 17.11) Nucleophilic substitution by ammonia on a-halo acids (Section 20.15) Nucleophilic acyl substitution (Sections 19.4, 19.5, and 19.11) 21.7 Preparation of Amines by Alkylation of Ammonia Alkylation of Ammonia Desired reaction is: 2 NH3 + R—X R—NH2 + NH4X + H3N •• – •• X •• •• via: H3N •• + R then: H3N •• + H •• X •• •• H + N R H R + H + H3N H + •• N H R Alkylation of Ammonia But the method doesn't work well in practice. Usually gives a mixture of primary, secondary, and tertiary amines, plus the quaternary salt. NH3 RX RNH2 RX R2NH RX + R4N X – RX R3 N Example CH3(CH2)6CH2Br NH3 CH3(CH2)6CH2NH2 (45%) + CH3(CH2)6CH2NHCH2(CH2)6CH3 (43%) As octylamine is formed, it competes with ammonia for the remaining 1-bromooctane. Reaction of octylamine with 1-bromooctane gives N,N-dioctylamine. 21.8 The Gabriel Synthesis of Primary Alkylamines Gabriel Synthesis Gives primary amines without formation of secondary, etc. amines as byproducts. Uses an SN2 reaction on an alkyl halide to form the C—N bond. The nitrogen-containing nucleophile is N-potassiophthalimide. Gabriel Synthesis Gives primary amines without formation of secondary, etc. amines as byproducts. Uses an SN2 reaction on an alkyl halide to form the C—N bond. The nitrogen-containing nucleophile is N-potassiophthalimide. O – •• N • • O K + N-Potassiophthalimide The pKa of phthalimide is 8.3. N-potassiophthalimide is easily prepared by the reaction of phthalimide with KOH. O O •• NH O KOH – •• N • • O K + N-Potassiophthalimide as a Nucleophile O O – •• N • + R • •• X •• SN2 •• N •• O O + •• – •• X •• •• R Cleavage of Alkylated Phthalimide O •• N R + H2O O Imide hydrolysis is nucleophilic acyl substitution. acid or base CO2H + CO2H H2N R Cleavage of Alkylated Phthalimide Hydrazinolysis is an alternative method of releasing the amine from its phthalimide derivative. O O •• N R H2NNH2 NH NH O O + H2N R Example O – •N• • • K + + C6H5CH2Cl DMF O O •• N O CH2C6H5 (74%) Example O NH + C6H5CH2NH2 (97%) NH H2NNH2 O O •• N O CH2C6H5 21.9 Preparation of Amines by Reduction Preparation of Amines by Reduction Almost any nitrogen-containing compound can be reduced to an amine, including: azides nitriles nitro-substituted benzene derivatives amides Synthesis of Amines via Azides SN2 reaction, followed by reduction, gives a primary alkylamine. CH2CH2Br NaN3 CH2CH2N3 (74%) 1. LiAlH4 2. H2O Azides may also be reduced by catalytic hydrogenation. CH2CH2NH2 (89%) Synthesis of Amines via Nitriles SN2 reaction, followed by reduction, gives a primary alkylamine. NaCN CH3CH2CH2CH2Br Nitriles may also be reduced by lithium aluminum hydride. CH3CH2CH2CH2CN (69%) H2 (100 atm), Ni CH3CH2CH2CH2CH2NH2 (56%) Synthesis of Amines via Nitriles SN2 reaction, followed by reduction, gives a primary alkylamine. NaCN CH3CH2CH2CH2Br CH3CH2CH2CH2CN The reduction also works with cyanohydrins. (69%) H2 (100 atm), Ni CH3CH2CH2CH2CH2NH2 (56%) Synthesis of Amines via Nitroarenes HNO3 Cl H2SO4 Nitro groups may also be reduced with tin (Sn) + HCl or by catalytic hydrogenation. Cl NO2 Cl (88-95%) 1. Fe, HCl 2. NaOH NH2 (95%) Synthesis of Amines via Amides O COH O 1. SOCl2 CN(CH3)2 2. (CH3)2NH (86-89%) Only LiAlH4 is an appropriate reducing agent for this reaction. 1. LiAlH4 2. H2O CH2N(CH3)2 (88%) 21.10 Reductive Amination Synthesis of Amines via Reductive Amination In reductive amination, an aldehyde or ketone is subjected to catalytic hydrogenation in the presence of ammonia or an amine. R fast C R' R O + NH3 C NH + R' The aldehyde or ketone equilibrates with the imine faster than hydrogenation occurs. H2O Synthesis of Amines via Reductive Amination The imine undergoes hydrogenation faster than the aldehyde or ketone. An amine is the product. R fast C R O + NH3 R' C H NH + R' R R' C H2, Ni NH2 H2O Example: Ammonia Gives a Primary Amine O + NH3 H2, Ni H ethanol NH2 (80%) via: NH Example: Primary Amines Give Secondary Amines O CH3(CH2)5CH + H2N H2, Ni ethanol CH3(CH2)5CH2NH via: CH3(CH2)5CH N (65%) Example: Secondary Amines Give Tertiary Amines O CH3CH2CH2CH + N H H2, Ni, ethanol N CH2CH2CH2CH3 (93%) Example: Secondary Amines Give Tertiary Amines Possible intermediates include: HO N + N CHCH2CH2CH3 CHCH2CH2CH3 N CH CHCH2CH3 21.11 Reactions of Amines: A Review and a Preview Reactions of Amines Reactions of amines almost always involve the nitrogen lone pair. as a base: N •• H X as a nucleophile: N •• C O Reactions of Amines Reactions already discussed basicity (Section 21.4) reaction with aldehydes and ketones (Sections 17.10, 17.11) reaction with acyl chlorides (Section 19.4), anhydrides (Section 19.5), and esters (Section 19.11) 21.12 Reactions of Amines with Alkyl Halides Reaction with Alkyl Halides Amines act as nucleophiles toward alkyl halides. N •• + R + •• – N R + •• X •• •• X •• •• •• H H N •• R + H + Example: excess amine NH2 + ClCH2 (4 mol) (1 mol) NaHCO3 90°C NHCH2 (85-87%) Example: excess alkyl halide CH2NH2 methanol + 3CH3I heat + CH2N(CH3)3 (99%) I – 21.13 The Hofmann Elimination The Hofmann Elimination A quaternary ammonium hydroxide is the reactant and an alkene is the product. It is an anti elimination. The leaving group is a trialkylamine. The regioselectivity is opposite to the Zaitsev rule. Quaternary Ammonium Hydroxides are prepared by treating quaternary ammmonium halides with moist silver oxide CH2N(CH3)3 Ag2O I – H2O, CH3OH + CH2N(CH3)3 – HO The Hofmann Elimination on being heated, quaternary ammonium hydroxides undergo elimination CH2 + N(CH3)3 + (69%) 160°C + CH2N(CH3)3 – HO H2O Mechanism – •• •• O •• •• O •• H H H H CH2 CH2 N(CH3)3 + •• N(CH3)3 Regioselectivity Elimination occurs in the direction that gives the less-substituted double bond. This is called the Hofmann rule. H2C CH3CHCH2CH3 CHCH2CH3 (95%) heat + N(CH3)3 HO – + CH3CH CHCH3 (5%) Regioselectivity Steric factors seem to control the regioselectivity. The transition state that leads to 1-butene is less crowded than the one leading to cis or trans-2-butene. Regioselectivity H CH3CH2 H H H H CH3CH2 +N(CH3)3 largest group is between two H atoms H C C H major product Regioselectivity H H CH3 CH3 H +N(CH3)3 largest group is between an H atom and a methyl group CH3 H H C C CH3 minor product 21.14 Electrophilic Aromatic Substitution in Arylamines Nitration of Aniline NH2 is a very strongly activating group. NH2 not only activates the ring toward electrophilic aromatic substitution, it also makes it more easily oxidized. Attemped nitration of aniline fails because nitric acid oxidizes aniline to a black tar. Nitration of Aniline Strategy: decrease the reactivity of aniline by converting the NH2 group to an amide O NH2 O O CH3COCCH3 CH(CH3)2 NHCCH3 (98%) CH(CH3)2 (acetyl chloride may be used instead of acetic anhydride) Nitration of Aniline Strategy: nitrate the amide formed in the first step O O NHCCH3 NO2 CH(CH3)2 (94%) NHCCH3 HNO3 CH(CH3)2 Nitration of Aniline Strategy: remove the acyl group from the amide by hydrolysis O NHCCH3 NO2 NH2 NO2 KOH ethanol, heat CH(CH3)2 CH(CH3)2 (100%) Halogenation of Arylamines occurs readily without necessity of protecting amino group, but difficult to limit it to monohalogenation NH2 NH2 Br2 Br Br acetic acid CO2H CO2H (82%) Monohalogenation of Arylamines Decreasing the reactivity of the arylamine by converting the NH2 group to an amide allows halogenation to be limited to monosubstitution. O O NHCCH3 NHCCH3 CH3 CH3 Cl2 acetic acid Cl (74%) Friedel-Crafts Reactions The amino group of an arylamine must be protected as an amide when carrying out a Friedel-Crafts reaction. O O NHCCH3 CH2CH3 NHCCH3 O CH2CH3 CH3CCl AlCl3 O CCH3 (57%) 21.15 Nitrosation of Alkylamines Nitrite Ion, Nitrous Acid, and Nitrosyl Cation – •• •• O •• •• O •• N •• H + •• O H •• + H • O •• • H O •• N H H •• •• + •• N + + •• O •• O •• H •• N •• O •• Nitrosyl Cation and Nitrosation •• N + •• O •• Nitrosyl Cation and Nitrosation + N N •• + •• •• O •• N •• N + •• O •• Nitrosation of Secondary Alkylamines + N •• •• O •• N •• N H H O •• N + H N •• •• •• + •• N + •• O •• + Nitrosation of secondary amines gives an N-nitroso amine. Example •• (CH3)2NH NaNO2, HCl H2O •• (CH3)2N •• N (88-90%) •• O •• Some N-Nitroso Amines (CH3)2N N O N-nitrosodimethylamine (leather tanning) N N N N O N-nitrosopyrrolidine (nitrite-cured bacon) N O N-nitrosonornicotine (tobacco smoke) Nitrosation of Primary Alkylamines R H + N R •• N •• O •• •• N H H H N •• H + •• N + •• O •• O •• N + H R •• •• + Analogous to nitrosation of secondary amines to this point. Nitrosation of Primary Alkylamines R •• N •• N H •• + H •• N H + N O •• H O •• N H R •• •• •• •• N O H •• + R H + This species reacts further. R H •• N •• N O •• + H Nitrosation of Primary Alkylamines Nitrosation of a primary alkylamine gives an alkyl diazonium ion. Process is called diazotization. H R + N N •• + •• O •• H R •• N H •• N O •• + H Alkyl Diazonium Ions + + •N R • N •• Alkyl diazonium ions readily lose N2 to give carbocations. R + N N •• Example: Nitrosation of 1,1-Dimethylpropylamine HONO NH2 OH + N H2O – N2 + (80%) + Mechanism 21.2 (3%) N (2%) Nitrosation of Tertiary Alkylamines There is no useful chemistry associated with the nitrosation of tertiary alkylamines. R R R R + N N •• R R •• N •• O •• 21.16 Nitrosation of Arylamines Nitrosation of Tertiary Arylamines Reaction that occurs is electrophilic aromatic substitution. N(CH2CH3)2 1. NaNO2, HCl, H2O, 8°C N(CH2CH3)2 2. HO– N (95%) O Nitrosation of N-Alkylarylamines Similar to secondary alkylamines; Gives N-nitroso amines NaNO2, HCl, H2O, 10°C NHCH3 N O NCH3 (87-93%) Nitrosation of Primary Arylamines Gives aryl diazonium ions. Aryl diazonium ions are much more stable than alkyl diazonium ions. Most aryl diazonium ions are stable under the conditions of their formation (0-10°C). + RN + ArN N N fast + R + N2 slow + Ar + N2 Example: (CH3)2CH NH2 NaNO2, H2SO4 H2O, 0-5°C (CH3)2CH + N N HSO4– Synthetic Origin of Aryl Diazonium Salts Ar H Ar NO2 Ar NH2 Ar + N N 21.17 Synthetic Transformations of Aryl Diazonium Salts Transformations of Aryl Diazonium Salts Ar Ar Cl Ar CN + N Ar Ar Ar F Ar I N H Ar Br OH Preparation of Phenols + N Ar N H2O, heat Ar OH Example NH2 (CH3)2CH 1. NaNO2, H2SO4 H2O, 0-5°C 2. H2O, heat OH (CH3)2CH (73%) Transformations of Aryl Diazonium Salts Ar Ar Cl Ar CN + N Ar Ar Ar F Ar I N H Ar Br OH Preparation of Aryl Iodides Reaction of an aryl diazonium salt with potassium iodide: Ar + N N KI Ar I Example NH2 Br 1. NaNO2, HCl H2O, 0-5°C I Br 2. KI, room temp. (72-83%) Transformations of Aryl Diazonium Salts Ar Ar Cl Ar CN + N Ar Ar Ar F Ar I N H Ar Br OH Preparation of Aryl Fluorides Ar Ar + N F N Heat the tetrafluoroborate salt of a diazonium ion; process is called the Schiemann reaction. Example NH2 1. NaNO2, HCl, H2O, 0-5°C CCH2CH3 O 2. HBF4 3. heat F CCH2CH3 O (68%) Transformations of Aryl Diazonium Salts Ar Ar Cl Ar CN + N Ar Ar Ar F Ar I N H Ar Br OH Preparation of Aryl Chlorides and Bromides Ar Cl Ar Ar + N Br N Aryl chlorides and aryl bromides are prepared by heating a diazonium salt with copper(I) chloride or bromide. Substitutions of diazonium salts that use copper(I) halides are called Sandmeyer reactions. Example NH2 1. NaNO2, HCl, H2O, 0-5°C NO2 2. CuCl, heat Cl NO2 (68-71%) Example NH2 Cl 1. NaNO2, HBr, H2O, 0-10°C Br Cl 2. CuBr, heat (89-95%) Transformations of Aryl Diazonium Salts Ar Ar Cl Ar CN + N Ar Ar Ar F Ar I N H Ar Br OH Preparation of Aryl Nitriles Ar CN Ar + N N Aryl nitriles are prepared by heating a diazonium salt with copper(I) cyanide. This is another type of Sandmeyer reaction. Example NH2 CH3 1. NaNO2, HCl, H2O, 0°C CN CH3 2. CuCN, heat (64-70%) Transformations of Aryl Diazonium Salts Ar Ar Cl Ar CN + N Ar Ar Ar F Ar I N H Ar Br OH Transformations of Aryl Diazonium Salts Hypophosphorous acid (H3PO2) reduces diazonium salts; ethanol does the same thing. This is called reductive deamination. Ar Ar H + N N Example NH2 CH3 NaNO2, H2SO4, H3PO2 or NaNO2, HCl, CH3CH2OH CH3 (70-75%) Value of Diazonium Salts 1) Allows introduction of substituents such as OH, F, I, and CN on the ring. 2) Allows preparation of otherwise difficultly accessible substitution patterns. Example NH2 NH2 Br2 NaNO2, H2SO4, Br H O, CH CH OH 2 3 2 Br H2O Br Br Br (100%) Br (74-77%) 21.18 Azo Coupling Azo Coupling Diazonium salts are weak electrophiles. React with strongly activated aromatic compounds by electrophilic aromatic substitution. Ar + N N + Ar' H Ar N N Ar' an azo compound Ar' must bear a strongly electron-releasing group such as OH, OR, or NR2. Example OH + + C6H5N N OH N NC6H5 Cl– Section 21.19 Spectroscopic Analysis of Amines Infrared Spectroscopy The N—H stretching band appears in the range 3000-3500 cm-1. Primary amines give two peaks in this region, one for a symmetrical stretching vibration, the other for an antisymmetrical stretch. H R N H R H symmetric N H antisymmetric Infrared Spectroscopy Primary amines give two N—H stretching peaks, secondary amines give one (Figure 21.8). 1H NMR Compare chemical shifts in: H3C CH2NH2 H3C 3.9 ppm N C H is more shielded than O CH2OH 4.7 ppm C H 13C NMR Carbons bonded to N are more shielded than those bonded to O. CH3NH2 26.9 ppm CH3OH 48.0 ppm UV-VIS An amino group on a benzene ring shifts max to longer wavelength. Protonation of N causes UV spectrum to resemble that of benzene. + NH3 NH2 max 204 nm 256 nm max 230 nm 280 nm max 203 nm 254 nm Mass Spectrometry Compounds that contain only C, H, and O have even molecular weights. If an odd number of N atoms is present, the molecular weight is odd. A molecular-ion peak with an odd m/z value suggests that the sample being analyzed contains N. Mass Spectrometry Nitrogen stabilizes carbocations, which drives the fragmentation pathways. •• (CH3)2NCH2CH2CH2CH3 e– •+ (CH3)2NCH2CH2CH2CH3 + (CH3)2N CH2 + •CH2CH2CH3 Mass Spectrometry Nitrogen stabilizes carbocations, which drives the fragmentation pathways. •• CH3NHCH2CH2CH(CH3)2 e– •+ CH3NHCH2CH2CH(CH3)2 + CH3NH CH2 + •CH2CH(CH3)2