* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Protein Therapeutics
No-SCAR (Scarless Cas9 Assisted Recombineering) Genome Editing wikipedia , lookup
Epigenetics of diabetes Type 2 wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Point mutation wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Genetic engineering wikipedia , lookup
Molecular cloning wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Gene therapy of the human retina wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
History of genetic engineering wikipedia , lookup
Protein moonlighting wikipedia , lookup
Mir-92 microRNA precursor family wikipedia , lookup
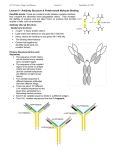
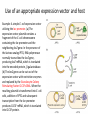
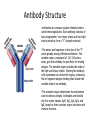
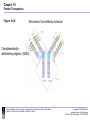
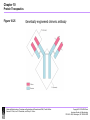
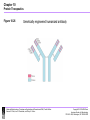
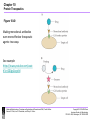
Chapter 10-Protein Therapeutics •Pharmaceutical proteins and enzymes •Monoclonal antibodies and recombinant antibodies Table 10.1 Some recombinant proteins approved for human use ($190 billion-2015) Protein Company Disorder Factor VIII Baxter, Bayer Hemophilia A Factor IX Genetics Institute Hemophilia B Tissue plasminogen Genetech activator (TPA) Acute myocardial infarction Insulin Eli Lilly, Novo Nordisk Diabetes mellitus Human growth hormone Eli Lilly, Genetech, Upjohn, Novo Nordisk GH deficiency in children (dwarfism) Erythropoietin Amgen, Ortho Biotech Anemia DNase I Genetech Cystic fibrosis Various interferons (IFN) Schering, Biogen, Chiron, Hepatitis B and C, multiple sclerosis Genetech Chapter 10 Protein Therapeutics Table 10.1 Molecular Biotechnology: Principles and Applications of Recombinant DNA, Fourth Edition Bernard R. Glick, Jack J. Pasternak, and Cheryl L. Patten Copyright © 2010 ASM Press American Society for Microbiology 1752 N St. NW, Washington, DC 20036-2904 Recombinant proteins-from http://en.wikipedia.org/wiki/List_of_recombinant_proteins -2/6/16 • • • • • • • • • • • • • • • • • • • • • • • Human recombinants that largely replaced animal or harvested from human types Human growth hormone (rHGH): Humatrope from Lilly and Serostim from Serono replaced cadaver harvested human growth hormone human insulin (BHI): Humulin from Lilly and Novolin from Novo Nordisk among others largely replaced bovine and porcine insulin for human therapy. Some prefer to continue using the animal-sourced preparations, as there is some evidence that synthetic insulin varieties are more likely to induce hypoglycemia unawareness. Remaining manufacturers of highly purified animal-sourced insulin include the U.K.'s Wockhardt Ltd. (headquartered in India), Argentina's Laboratorios Beta S.A., and China's Wanbang Biopharma Co. Follicle-stimulating hormone (FSH) as a recombinant gonadotropin preparation replaced Serono's Pergonal which was previously isolated from post-menopausal female urine Factor VIII: Kogenate from Bayer replaced blood harvested factor VIII Human recombinants with recombination as only source Erythropoietin (EPO): Epogen from Amgen Granulocyte colony-stimulating factor (G-CSF): filgrastim sold as Neupogen from Amgen; pegfilgrastim sold as Neulasta alpha-glactosidase A: Fabrazyme by Genzyme alpha-L-iduronidase: (rhIDU; laronidase) Aldurazyme by BioMarin Pharmaceutical and Genzyme N-acetylgalactosamine-4-sulfatase (rhASB; galsulfase): Naglazyme by BioMarin Pharmaceutical Dornase alfa, a DNase sold under the trade name Pulmozyme by Genentech Tissue plasminogen activator (TPA) Activase by Genentech Glucocerebrosidase: Ceredase by Genzyme Interferon (IF) Interferon-beta-1a: Avonex from Biogen Idec; Rebif from Serono; Interferon beta-1b as Betaseron from Schering Insulin-like growth factor 1 (IGF-1) Animal recombinants Bovine somatotropin (bST) Porcine somatotropin (pST) Bovine Chymosin Viral recombinants Envelope protein of the hepatitis B virus marketed as Engerix-B by SmithKline Beecham HPV Vaccine proteins Cloning and expression of a foreign protein in a suitable host Expression systems are based on the insertion of a gene into a host cell for its translation and expression into protein. Host cells include : Bacteria - e.g. Escherichia coli (E.coli), Bacillus subtilis (B. subtilis) Yeast Cultured insect cells Cultured mammalian cells The choice of cell type used depends upon the protein to be expressed. All require DNA to be cloned into the an appropriate vector. Advantages of bacterial cells simple physiology short generation times, as bacteria grow and multiply rapidly large yields of product - up to 10 % of mass (low cost) With B. subtilis and some others, it is possible to induce secretion of a gene product into the surrounding medium. This method is in use in the pharmaceutical industry in the production of hormones such as insulin and human growth hormone. Disadvantages of bacterial cells The expressed proteins often do not fold properly and so are biologically inactive. The synthesised protein is often toxic to bacteria preventing the cell cultures from reaching high densities. A solution to this problem is to incorporate an inducible promoter, which may be turned on to transcribe the inserted gene after the culture has been grown Lack of enzymes responsible for post-translational modifications (effect on function of proteins), eg if the protein to be expressed is a glycoprotein, there is not apparatus in the bacterium to 'stick on' the necessary sugar residues. Advantages of yeast cells Yeast is a simple eukaryote and performs many of the post-translational modifications required for human proteins Can be induced to secrete certain proteins into the growth medium for harvesting - e.g. Hepatitis B virus (HBV) vaccine. Disadvantages of yeast cells Presence of active proteases that degrade foreign (expressed) proteins, thereby reducing their yield (a solution to this problem is the construction of yeast strains from which the protease genes have been deleted). Insect Cells-Expression of foreign proteins in insect cells through incorporation of their genes into baculovirus vectors Advantages of insect cells High level of expression Correct folding Post-translational modifications similar to those in mammalian cells Cost, though more than for culturing bacteria and yeast, less than for mammalian cells e.g. potential vaccine for AIDS virus produced by expression of one of the HIV glycoproteins with this system Disadvantages of insect cells More difficult to work with Expensive Slow generation time Not suitable for proteins with repetitive sequences Comparison of Recombinant Protein Expression Use of an appropriate expression vector and host Example: A simple E. coli expression vector utilizing the lac promoter. (a) The expression vector plasmid contains a fragment of the E. coli chromosome containing the lac promoter and the neighboring lacZ gene. In the presence of the lactose analog IPTG, RNA polymerase normally transcribes the lacZ gene, producing lacZ mRNA, which is translated into the encoded protein, b-galactosidase. (b) The lacZ gene can be cut out of the expression vector with restriction enzymes and replaced by the Granulocyte-Colony Stimulating Factor G-CSF cDNA. When the resulting plasmid is transformed into E. coli cells, addition of IPTG and subsequent transcription from the lac promoter produces G-CSF mRNA, which is translated into G-CSF protein. Therapeutic monoclonal antibodies approved for human use • See https://en.wikipedia.org/wiki/Monoclonal_an tibody_therapy • Also see https://en.wikipedia.org/wiki/List_of_therape utic_monoclonal_antibodies Chapter 10 Protein Therapeutics Table 10.3 mAB Mode of Action: https://www.youtube.com/watch?v=48VSU4AZ-L0 or https://www.youtube.com/watch?v=wdTejNk8ato Molecular Biotechnology: Principles and Applications of Recombinant DNA, Fourth Edition Bernard R. Glick, Jack J. Pasternak, and Cheryl L. Patten Copyright © 2010 ASM Press American Society for Microbiology 1752 N St. NW, Washington, DC 20036-2904 Therapeutic monoclonal antibodies approved for human use: A multi-billion dollar industry “Since the commercialization of the first therapeutic monoclonal antibody product in 1986, this class of biopharmaceutical products has grown significantly so that, as of November 10, 2014, forty-seven monoclonal antibody products have been approved in the US or Europe for the treatment of a variety of diseases, and many of these products have also been approved for other global markets. At the current approval rate of ~ four new products per year, ~70 monoclonal antibody products will be on the market by 2020, and combined world-wide sales will be nearly $125 billion.” -Dawn M Ecker, Susan Dana Jones, and Howard L Levine (2015) The therapeutic monoclonal antibody market. mAbs 7(1), 9-14. Antibody Structure •Antibodies are immune system-related proteins called immunoglobulins. Each antibody consists of four polypeptides– two heavy chains and two light chains joined to form a "Y" shaped molecule. •The amino acid sequence in the tips of the "Y" varies greatly among different antibodies. This variable region, composed of 110-130 amino acids, give the antibody its specificity for binding antigen. The variable region includes the ends of the light and heavy chains. Treating the antibody with a protease can cleave this region, producing Fab or fragment antigen binding that include the variable ends of an antibody. •The constant region determines the mechanism used to destroy antigen. Antibodies are divided into five major classes, IgM, IgG, IgA, IgD, and IgE, based on their constant region structure and immune function. Chapter 10 Protein Therapeutics Figure 10.24 Structure of an antibody molecule Complementaritydetermining regions (CDRs) Molecular Biotechnology: Principles and Applications of Recombinant DNA, Fourth Edition Bernard R. Glick, Jack J. Pasternak, and Cheryl L. Patten Copyright © 2010 ASM Press American Society for Microbiology 1752 N St. NW, Washington, DC 20036-2904 Chapter 10 Protein Therapeutics Figure 10.25 Genetically engineered chimeric antibody Molecular Biotechnology: Principles and Applications of Recombinant DNA, Fourth Edition Bernard R. Glick, Jack J. Pasternak, and Cheryl L. Patten Copyright © 2010 ASM Press American Society for Microbiology 1752 N St. NW, Washington, DC 20036-2904 Chapter 10 Protein Therapeutics Figure 10.26 Genetically engineered humanized antibody Molecular Biotechnology: Principles and Applications of Recombinant DNA, Fourth Edition Bernard R. Glick, Jack J. Pasternak, and Cheryl L. Patten Copyright © 2010 ASM Press American Society for Microbiology 1752 N St. NW, Washington, DC 20036-2904 Chapter 10 Protein Therapeutics Figure 10.40 Making monoclonal antibodies even more effective therapeutic agents: two ways See example: https://www.youtube.com/watc h?v=GD0gcZoqtcM Molecular Biotechnology: Principles and Applications of Recombinant DNA, Fourth Edition Bernard R. Glick, Jack J. Pasternak, and Cheryl L. Patten Copyright © 2010 ASM Press American Society for Microbiology 1752 N St. NW, Washington, DC 20036-2904




















![Anti-PCB antibody [3H2AD9] ab110314 Product datasheet 3 Images Overview](http://s1.studyres.com/store/data/000076345_1-acbfa58e194757c519d151062b812354-150x150.png)