* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download ENZYMES
Discovery and development of tubulin inhibitors wikipedia , lookup
Discovery and development of direct thrombin inhibitors wikipedia , lookup
Orphan drug wikipedia , lookup
Metalloprotease inhibitor wikipedia , lookup
Discovery and development of direct Xa inhibitors wikipedia , lookup
Discovery and development of non-nucleoside reverse-transcriptase inhibitors wikipedia , lookup
Drug design wikipedia , lookup
Discovery and development of HIV-protease inhibitors wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Drug discovery wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Prescription costs wikipedia , lookup
Neuropharmacology wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Pharmacognosy wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
Psychopharmacology wikipedia , lookup
Discovery and development of integrase inhibitors wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Discovery and development of ACE inhibitors wikipedia , lookup
Drug interaction wikipedia , lookup
Discovery and development of cyclooxygenase 2 inhibitors wikipedia , lookup
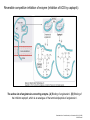
Charles University in Prague, Third Faculty of Medicine GENERAL MEDICINE 6-YEAR MASTER‘S STUDY PROGRAMME Subject: General Pharmacology Drug – enzymes interactions Prof. M. Kršiak Department of Pharmacology, Third Faculty of Medicine Ruská 87, Prague 10, Academic year 2013-2014 http://vyuka.lf3.cuni.cz CVSE3P0012 ID9226 Four major targets for drug action: ENZYMES Figure 3.1 Types of target for drug action. Downloaded from: StudentConsult (on 5 November 2013 05:28 PM) © 2005 Elsevier Enzyme inhibition by drugs Other drug-enzymes interactions Many drugs are targeted on enzymes and mostly act by inhibiting them: Therapeutic groups, indications Enzymes Inhibitors Cyclo-oxygenase aspirin, ibuprofen, diclofenac Monoamine oxidase moclobemide Acetylcholinesterase neostigmine, rivastigmin Parasympathomimetics, Anti-dementiadrugs Angiotensin-converting enzyme enalapril, ramipril Antihypertensives HMG-CoA reductase simvastatin, atorvastatin Lipid modifying agents; (hypercholesterolaemia) Xanthinoxidase allopurinol Drugs inhibiting uric acid production Phosphodiesterase type V sildenafil Drugs used in erectile dysfunction Dihydrofolate reductase trimethoprim Antiinflammatory and antirheumatic agents, analgesics Antidepressants Antimicrobial agents methotrexate Antimetabolites, folic acid analogues Neuroamidase oseltamivir Antivirals ( influenza virus) Thymidine kinase aciclovir Antivirals (Herpes virus) HIV protease saquinavir Antivirals (HIV), protease inhibitors An enzyme inhibitor is a molecule which binds to enzymes and decreases their activity Drugs can inhibit enzymes reversibly (usually a competitive inhibition by non-covalent binding) or irreversibly (enzyme is usually changed chemically by covalent binding) Competitive inhibition is a form of enzyme inhibition where binding of the inhibitor to the active site on the enzyme prevents binding of the substrate and vice versa. Often, the drug molecule is a substrate analogue (e.g. captopril, acting on angiotensin-converting enzyme) Irreversible inhibitors usually react with the enzyme and change it chemically (e.g. via covalent bond formation). These inhibitors modify key amino acid residues needed for enzymatic activity (e.g. aspirin, acting on cyclo-oxygenase) Reversible competitive inhibition of enzyme (inhibition of ACE by captopril):: The active site of angiotensin-converting enzyme. [A] Binding of angiotensin I. [B] Binding of the inhibitor captopril, which is an analogue of the terminal dipeptide of angiotensin I. Downloaded from: StudentConsult (on 6 November 2013 02:30 PM) © 2005 Elsevier Irreversible non-competitive inhibition of enzyme (inhibition of COX-1 or COX-2 by aspirin): Aspirin acetylates serine residue in the active site of the COX enzyme This makes aspirin different from other NSAIDs (such as diclofenac and ibuprofen, which are reversible inhibitors). Irreversible inhibition of enzyme: Recovery is possible only by synthesis of a new enzyme Irreversible inhibition of COX in thrombocytes and in endothelium • As thrombocytes (platelets) do not have nucleus (adequate DNA), they are unable to synthesize new COX once aspirin has irreversibly inhibited the enzyme • Endothelial cells have nucleus and are able to recover synthesis of COX MECHANISM OF ACTION OF NON-OPIOID ANALGESICS CYKLO-OXYGENASE COX CONSTITUTIVE ISOENZYME COX-1 PHYSIOLOGICAL FUNCTIONS PROTECTION OF GASTRIC MUCOUS MEMBR. INCREASE OF BLOOD FLOW AND SODIUM EXCRETION IN THE KIDNEY COX-1 inhibitors: ibuprofen, diclofenac and other risk of gastropathy INDUCIBLE ISOENZYME COX-2 INFLAMMATORY RESPONSE INFLAMMATION FEVER PAIN Selective COX-2 inhibitors: COXIBS lower risk of gastropathy Selective COX-2 inhibitors (Coxibs) have lower gastropathy but a higher risk for heart attack and stroke promotes clotting has protective anti-coagulative effect Arachidonic acid COX-1 COX-2 coxibs aspirin tromboxan A2 promotes clotting increases platelet + vasoconstriction aggregation aspirin prevents platelet aggregation prostacyclin PGI2 inhibits platelet aggregation + vasodilatation coxibs = selective COX-2 inhibitors : higher trombotic risk Selective COX-2 inhibitors (coxibs) increase in the risk for heart attack and stroke through an increase of thromboxane unbalanced by prostacyclin (which is reduced by COX-2 inhibition) Non-steroidal Anti-inflammatory Drugs (NSAIDs) Major required effects: Analgesic + Antipyretic +Anti-inflammatory Classification of NSAIDs (by selectivity of inhibition of COX-1 and COX -2): • Nonselective (COX-1 and COX-2) ibuprofen, diklofenac … • Preferential (COX-2 > COX-1) nimesulide, meloxicam • Selective (coxibs) (COX-2 only) celecoxib … Acetylcholinesterase inhibitors inhibit the acetylcholinesterase from breaking down acetylcholine, thereby increasing both the level and duration of action of the neurotransmitter acetylcholine. REVERSIBLE physostigmine, neostigmine, rivastigmine Are used medicinally: • antidote to anticholinergic poisoning • to treat glaucoma • to treat myasthenia gravis • to treat Alzheimer disease • to reverse the effect of non-depolarising muscle relaxants IRREVERSIBLE • Are used as weapons in the form of nerve agents • Are used as insecticides Monoamine oxidase inhibitors (MAOIs) a long history originally irreversible, now withdrawn Because of potentially lethal dietary („cheese effect“ and drug interactions, hypertensive crisis MAOIs have been reserved as a last line of treatment, used only when other classes of antidepressant drugs have failed. Monoamine oxidase inhibitors (MAOIs) at present: reversible RIMA MAO –A serotonin, noradrenalin (norepinephrine), tyramine moclobemid treatment of depression treatment of anxiety disorders (OCD, panic disorders, phobia) dietary restrictions serotonin syndrome MAO –B dopamine selegiline no dietary restrictions treatment of Parkinson‘s disease Many drugs are targeted on enzymes and mostly act by inhibiting them: Therapeutic groups, indications Enzymes Inhibitors Cyclo-oxygenase aspirin, ibuprofen, diclofenac Monoamine oxidase moclobemide Acetylcholinesterase neostigmine, rivastigmin Parasympathomimetics, Anti-dementiadrugs Angiotensin-converting enzyme enalapril, ramipril Antihypertensives HMG-CoA reductase simvastatin, atorvastatin Lipid modifying agents; (hypercholesterolaemia) Xanthinoxidase allopurinol Drugs inhibiting uric acid production Phosphodiesterase type V sildenafil Drugs used in erectile dysfunction Dihydrofolate reductase trimethoprim Antiinflammatory and antirheumatic agents, analgesics Antidepressants Antimicrobial agents methotrexate Antimetabolites, folic acid analogues Neuroamidase oseltamivir Antivirals ( influenza virus) Thymidine kinase aciclovir Antivirals (Herpes virus) HIV protease saquinavir Antivirals (HIV), protease inhibitors Some drugs that produce active or toxic metabolites Inactive (prodrugs) Active drug Active metabolite Prednisone → Prednisolone Enalapril → Enalaprilat Toxic metabolite Diazepam → Nordiazepam → Oxazepam Morphine → Morphine 6-glucuronide Paracetamol → N-Acetyl-pbenzoquinone imine Drugs may also act as false substrates, where the drug molecule undergoes chemical transformation to form an abnormal product that subverts the normal metabolic pathway. An example is the anticancer drug fluorouracil, which replaces uracil as an intermediate in purine biosynthesis but cannot be converted into thymidylate, thus blocking DNA synthesis and preventing cell division Drug - cytochrome P450 interactions Cytochrome P450 (CYP) enzymes The most important enzymes involved in drug interactions are members of the cytochrome P450 (CYP) system that are responsible for many of the phase 1 biotransformations of drugs. These metabolic transformations, such as oxidation, reduction and hydrolysis, produce a molecule that is suitable for conjugation. Those of importance in the metabolism of psychotropic drugs are CYP1A2, CYP2C9, CYP2C19, CYP2D6 and CYP3A4, the last being responsible for the metabolism of more than 90% of psychotropic drugs that undergo hepatic biotransformation. a high affinity for one particular CYP enzyme but most are oxidised by more than one Many psychotropic drugs have Genetic effects: Genetic polymorphism The CYP enzymes that demonstrate pharmacogenetic polymorphism include CYP2C9, CYP2C19 and CYP2D6. In clinical practice, the polymorphism produces distinct phenotypes, described as poor metabolisers, extensive metabolisers (the most common type) and ultra-rapid metabolisers. Drug effects: CYP enzymes can be induced or inhibited by drugs or other biological substances, with a consequent change in their ability to metabolise drugs that are normally substrates for those enzymes. Enzymatic induction enzymatic induction can cause a decrease as well as an increase in the drug’s effect The onset and offset of enzyme induction take place gradually, usually over 7– 10 days The most important are inducers of CYP3A4 and include carbamazepine, phenobarbital, phenytoin, rifampicin and St John’s wort (Hypericum perforatum). An example of an interaction in psychiatric practice is the reduced efficacy of haloperidol (or alprazolam) when carbamazepine is started, resulting from induction of CYP3A4. Enzymatic inhibition enzymatic inhibition can cause an increase as well as a decrease in the drug’s effect Inhibition is usually due to a competitive action at the enzyme’s binding site. Therefore, in contrast to enzyme induction, the onset and offset of inhibition are dependent on the plasma level of the inhibiting drug Inhibition of CYP enzymes is the most common mechanism that produces serious and potentially life-threatening drug interactions Most hazardous drug interactions involve inhibition of enzyme systems, which increases plasma concentrations of the drugs involved, in turn leading to an increased risk of toxic effects. Amitriptyline + fluoxetine Fluoxetine inhibits 2D6 Amitriptyline is a substrate for 2D6 Amitriptyline + fluoxetine → increased plasma levels of amitriptyline and prolonged t1/2 → sometimes fatal consequences OTHER ADVERSE CLINICAL CONSEQUENCES OF DRUG INTERACTIONS* Profound oversedation Profound and prolonged sedation can be brought about by inhibition of CYP3A4 enzymes that are involved in the metabolism of anxiolytics and sedatives e.g. alprazolam, midazolam + ketoconazole/clarithromycine/grapefruit Severe sedation due to the additive effect (summation) of drugs with sedating properties is a particular problem in elderly and frail people, and it can lead to falls and injuries (especially fractures of the femoral neck). Excessively drowsy patients are also at increased risk of venous thromboembolism and, if confined to bed, of hypostatic pneumonia. In people who drive, increased sedation due to drug interactions carries a correspondingly increased risk of road traffic accidents.