* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Ocaliva
Survey
Document related concepts
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Drug design wikipedia , lookup
Discovery and development of angiotensin receptor blockers wikipedia , lookup
Pharmacognosy wikipedia , lookup
Drug discovery wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Neuropharmacology wikipedia , lookup
Drug interaction wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
Transcript
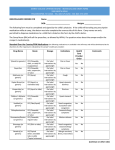
New Drug Introduction: Ocaliva / obeticholic acid Pharmacology Manufacturer Approval Date Indications Contraindications Black Box Warnings Warnings/Precautions Pregnancy/Lactation Pharmacokinetics Drug Interactions – Object Drugs All drug interaction effects listed at: 10 mg Ocaliva Drug Interactions – Precipitant drugs Adverse Effects (Treatment%) [Placebo%] Monitoring Efficacy Monitoring Toxicity Dosing - Initial Dosing - Usual Dosing - Max Renal Adjustment Hepatic Adjustment Farnesoid X Receptor Agonist Intercept Pharmaceuticals, Inc 5/27/2016 Treatment of primary biliary cholangitis (PBC) as monotherapy or in combination with ursodeoxycholic acid (UDCA) Patients with complete biliary obstruction None Liver – Related Adverse Reactions Severe Pruritus Reduction in HDL-C Limited human data available. No fetal harm at exposures at 13 and 6 times the max amount in rats and rabbits, respectively. No information on drug transfer in lactation. A – Cmax occurred ~1.5 hrs and follows linear kinetics. Food did not alter extent of absorption. D – Volume is 618 L and binds to >99% of human plasma protein. M – Conjugated with glycine or taurine in liver and secreted into bile, these metabolites are pharmaceutically active. Conjugates are absorbed in the small intestine leading to enterohepatic recirculation. E – 87% excretion in feces through biliary secretion. <3% was excreted in urine with no detection of obeticholic acid. CYP1A2: potential for increased exposure to CYP1A2 substrates. 25 mg warfarin – 13% systemic exposure 200 mg caffeine – 42% AUC and 6% in Cmax 20 mg omeprazole – 32% AUC and 33% in Cmax 20 mg rosuvastatin – 22% AUC and 27% in Cmax Not metabolized to significant extent by CYP450 20 mg omeprazole <1.2 fold exposure. Determined to be not clinically significant. Pruritus (70%) [38%] Fatigue (25%) [15%] Rash (10%) [8%] Abdominal pain and discomfort(19%) [14%] Improvement in lowering ALP, a liver enzyme and biomarker used to track disease progression in PBC. No current mortality or symptom relief benefits LFTs, including alkaline phosphatase and bilirubin, lipid profile; signs/symptoms of hepatic adverse reactions and/or pruritus 5 mg daily 10 mg daily if adequate reduction not achieved in 3 months 10 mg daily None In moderate to severe impairment (Child-Pugh B and C): Initial: 5 mg PO weekly. If inadequate reduction in ALP and/or total bilirubin after 3 months, increase to 5 mg twice weekly (at least 3 days apart) and subsequently to 10 mg twice weekly (at least 3 days apart). Cost: Source: Uptodate. Accessed 09/01/2016 Dose(s) Brand- generic 5 mg Ocaliva – obeticholic 10 mg acid 250 mg (Urso 250) ursodiol tablets 500 mg (Urso Forte) dosed: 250 mg generic 13-15mg/kg/day 500 mg generic in 2-4 doses $ (30d) $6840 (30 tabs) $6840 (30 tabs) $528 (100 tabs) $936 (100 tabs) $268 (100 tabs) $475 (100 tabs) Summary Ocaliva™ - obeticholic acid is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodiol (ursodeoxycholic acid) in adults with an inadequate response to ursodiol, or as monotherapy in adults unable to tolerate ursodiol. This medication provides an alternative treatment option for PBC through a unique, novel mechanism of action. Obeticholic acid is a farnesoid X receptor agonist. Initial dosing for Ocaliva is 5 mg daily, titrated to 10 mg if adequate reduction not achieved in 3 months. o Child-Pugh B and C: 5 mg weekly References: 1. https://ocaliva.com/ 2. Ocaliva package insert. Intercept Pharmaceutical. May 2016. 3. Nevens F, Andreone P, Mazzella G, et al. A Placebo-Controlled Trial of Obeticholic Acid in Primary Biliary Cholangitis. N Engl J Med. 2016;375(7):631-43. Date Prepared: 09/23/2016 Editor: Peter G. Koval, Pharm.D., BCPS Author: Han Lin, Pharm.D. Candidate, UNC Eshelman School of Pharmacy