* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Cell Energy
Efficient energy use wikipedia , lookup
Dark energy wikipedia , lookup
William Flynn Martin wikipedia , lookup
Open energy system models wikipedia , lookup
Energy subsidies wikipedia , lookup
Potential energy wikipedia , lookup
100% renewable energy wikipedia , lookup
Energy storage wikipedia , lookup
Kinetic energy wikipedia , lookup
Low-Income Home Energy Assistance Program wikipedia , lookup
Public schemes for energy efficient refurbishment wikipedia , lookup
Zero-energy building wikipedia , lookup
Low-carbon economy wikipedia , lookup
World energy consumption wikipedia , lookup
Regenerative brake wikipedia , lookup
Energy Charter Treaty wikipedia , lookup
Alternative energy wikipedia , lookup
Energy policy of Australia wikipedia , lookup
Gibbs free energy wikipedia , lookup
International Energy Agency wikipedia , lookup
Energy returned on energy invested wikipedia , lookup
Energy harvesting wikipedia , lookup
Distributed generation wikipedia , lookup
Energy efficiency in transport wikipedia , lookup
Energy policy of the United Kingdom wikipedia , lookup
Internal energy wikipedia , lookup
Energy policy of Finland wikipedia , lookup
Life-cycle greenhouse-gas emissions of energy sources wikipedia , lookup
Negawatt power wikipedia , lookup
Energy in the United Kingdom wikipedia , lookup
Energy policy of the European Union wikipedia , lookup
Conservation of energy wikipedia , lookup
United States energy law wikipedia , lookup
Energy efficiency in British housing wikipedia , lookup
Energy Independence and Security Act of 2007 wikipedia , lookup
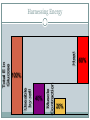
Energy Transformations An intro to energy conversions in living things Molecular Biology Sumner High School Created by Mr. Woodbury, Modified by K. Slater Big Question What is the relationship between life, matter and energy? Our two first definitions Energy: The capacity to do work. Examples: Energy stored in gasoline to make a car go, food to supply energy for motion. Food: Molecules that contain energy that can be converted into a useable form in organisms. Where is the energy stored? Within strong covalent bonds. Glucose: C6H12O6 Two categories of energy Potential Energy: stored energy and the energy of position Chemical Nuclear Gravitational Stored mechanical The other form... Kinetic Energy: the energy of motion Motion… duh Radiant (electromagnetic waves… aka light energy) Sound Thermal (heat) Electrical Quick Check Which of the following is an example of potential energy? A)The sound of a teacher’s voice B)Warmth from a heater C)Sugar D)Radio signal from a remote control Answer: C What types of PE, KE and energy transformations are in this pic? #8. Identify one energy transformation… My list... Potential Energy Diver waiting to jump: gravitational People: chemical Sun: nuclear Kinetic Energy Diver: motion People: sound & thermal Sun: thermal & radiant Ocean: motion Energy Conversions Diver: gravitational (PE) to motion (KE) People: chemical (PE) into thermal, sound & motion (KEs) Sun: nuclear (PE) into thermal & radiant (KEs) Next Question Do these forms of energy always stay in the same form? No, they can be transformed from one type to another. Energy transformation = changing one form of energy into other forms as a result of some event or process. Energy Transformations Remember: energy cannot be… We don’t really “use” energy, we just alter the form or type Especially in living systems A mental inquiry... What are the energy transformations done by an iPod? List the transformation and the start and end energy types. Foundation level: transformations done by the iPod itself (general types of energy: potential & kinetic) Challenge level: include transformations that happen several steps before/after the iPod (detailed types of energy) The iPod inquiry Foundation Level: Electricity stored in battery (kinetic to potential) Battery powers light (potential to kinetic) Battery powers headphones (potential to kinetic) Any ideas for Challenge Level? Thermodynamics These are the laws of energy First Law of Thermodynamics: “Energy can be transferred and transformed, but it cannot be created or destroyed.” Also known as the principle of conservation of energy. Key idea: there is a fixed amount of energy in the universe. Example: Plants do not produce energy; they transform light energy to chemical energy. Thermodynamics Second Law: every energy transfer or transformation increases the entropy (randomness or “chaos”) of the universe. Things are going from being organized to random. Key idea: No transformation is 100% efficient. Energy flows towards making more heat, which is the energy of random molecular motion. Example: nuclear energy of the sun is converted into radiant AND thermal energy Useable by cell 40% Muscle Contraction Heat Total E in Glucose Harnessing Energy 100% 20% 60% Summarize... Plan to take 2-3 minutes to silently summarize all of the information so far into one big idea (20 word max?) I will quasi-randomly call on you to share so we can hear from a variety of people. My summary... Living systems transform energy between various forms by controlling the rearrangement of molecules. Energy conversions in cell processes... Photosynthesis in the _____________: (organelle name) Radiant energy (KE) → chemical energy (PE) Cellular respiration in the _____________: (organelle name) Chemical energy (glucose / PE) → chemical energy (ATP / PE) → motion (KE) Energy Sources... Create a list of as many types of energy that you can think of (including those in today’s PPT) For each type of energy, identify it as potential or kinetic energy