* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Plant pathogenic bacteria
Survey
Document related concepts
Phospholipid-derived fatty acids wikipedia , lookup
Quorum sensing wikipedia , lookup
Infection control wikipedia , lookup
Trimeric autotransporter adhesin wikipedia , lookup
Sarcocystis wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Disinfectant wikipedia , lookup
Horizontal gene transfer wikipedia , lookup
Human microbiota wikipedia , lookup
Triclocarban wikipedia , lookup
Marine microorganism wikipedia , lookup
Bacterial cell structure wikipedia , lookup
Transcript
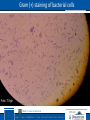
Plant pathogenic bacteria General characteristics Anton von Leuvenhook: first observation of bacteria 1683 TÁMOP-4.1.2.A/2-10/1-2010-0012 2 Koch’s postulates (1876) • Pathogen is the microorganism, which - can be isolated from the diseased organism, - can be grown in a pure culture - can infect the original host (with the original symptoms) - can be re-isolate from the new, infected host TÁMOP-4.1.2.A/2-10/1-2010-0012 3 Bacteria • • • • The smallest living organisms, Usually one cell, only few micrometer large, Shape: coccus, bacillus and spirillum Occurre everywhere, constant and important constituents of biosphere TÁMOP-4.1.2.A/2-10/1-2010-0012 4 Big discoveries in bacteriology • Leuwenhook 1683 First observation of bacteria • Koch 1876: anthrax bacillus: Bacillus anthracis • Burrill 1879: Erwinia amylovora – bacteria can cause disese of plants (fire blight) • Erwin Smith: First description of many bacteria • Pammel 1895: Xanthomonas campestris – from cabbage • Smith 1896: Pseudomonas solanacearum – from potato • Smith 1907: Agrobacterium tumefaciens – from fruit trees TÁMOP-4.1.2.A/2-10/1-2010-0012 5 Plant pathogenic bacteria 1950 Zoltán Klement the pioneer of plant bacteriology in Hungary Among 1600 bacterium genera only about 300 cause diseases on plants TÁMOP-4.1.2.A/2-10/1-2010-0012 6 Main properties of plant pathogenic bacteria • Heterotrophic organisms • Facultative parasites (usually can be cultured in pure cultures on artificial media) • Usually contains only single cells • Complex layer of cell wall • Shape: bacillus, coccus, spirillum • Majority Gram negative stained • Aerobe, seldom facultative anaerobe living style • No real nucleus, only chromatin, containing DNA • DNA containing small, round plasmids in the cytoplasm TÁMOP-4.1.2.A/2-10/1-2010-0012 7 Living forms of bacteria • Autotrophs (from the energy supply independent species, using the carbon dioxide as carbon source), chemotrophs, photoautotrops • Heterotrophs (using organic carbon source) TÁMOP-4.1.2.A/2-10/1-2010-0012 Main components of the bacterial cell • Cell membrane (lipid) • Cell wall (double structures of phospholipids and carbohydrates) • No independent membrane bound organelles (cell nucleus, plastids, endoplasmic reticulum) TÁMOP-4.1.2.A/2-10/1-2010-0012 Construction of bacterial cell • Complex cell wall, about 10-20 nm thick, mucoproteid skeleton, external membranes • (Gram + and -) groups • Plant pathogenic bacteria are usually Gram -, (G+ mucoproteid skeleton is loose, or G- is compact) - Inner cytoplasm membrane, - Periplasmic space (peptidoglucane), - Outer membrane (lipids, proteins, LPSlipopolysaccharids) - Cytoplasm: ribosomes, proteins, nucleic acids, plasmids - EPS envelope (extra-cellular polysaccharide) TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Bacterial taxons • Gram positive (no outer membrane) e.g. Actinobacteria • Gram negative (with outer membrane) e.g. Proteobacteria • Unknown, non classified bacteria e.g. Cyanobacteria TÁMOP-4.1.2.A/2-10/1-2010-0012 Extracellular structures • Bacterial cell wall (peptidoglucan) - Gram + type, thick, contains peptidoglucane and lipoteicnoinic acid - Gram – type thin, contains only lipopolysaccharides • Outher capsula (envelope) contains alginate and EPS (extra cellular polysaccharides) TÁMOP-4.1.2.A/2-10/1-2010-0012 Genetic material • No real, independent nucleus (prokaryotic organisms) • Irregular, round shaped DNAs in the nucleotide zone, only one giant chromosome • The genome contains about 300 genes with about 4Mbp TÁMOP-4.1.2.A/2-10/1-2010-0012 Cytoplasmic genetic elements • Chromatin (chromosome) • Plasmids: independent round shaped DNA molecules, coding few proeteins, responsibles for few properties (eg. antibiotic resistance) • Ribosomes and RNAs, producing proteins TÁMOP-4.1.2.A/2-10/1-2010-0012 Transposons • Special bacterial genetic elements, which can be translocated randomly from one part of the genome to other place TÁMOP-4.1.2.A/2-10/1-2010-0012 Movement organelles of bacterial cells • Flagellum (plural - flagella) 10-20 nm thick, 20-70 mm long, originated from thy cytoplasm • Types of flagella: - atrich (without flagellum); - monotrich (one): Xanthomonas; - lophotrich (lot in one side): Pseudomonas; - amphitrich (on two poles); - peritrich (around the whole surface) : Erwinia TÁMOP-4.1.2.A/2-10/1-2010-0012 16 Bacterial colonies • Development of colonies is characteristic for the bacteria • Emerging, round shape, slightly wave like…forms • Some are producing color substances (stains) (Corynebacterium michiganense yellow, C. insidiosum blue) TÁMOP-4.1.2.A/2-10/1-2010-0012 17 Reproduction types • • • • Rest (leg) phase, (time of DNS replication) Log phase (time of reproduction), 3-4 hrs Stationer phase, 24-78 hrs Asexual (division by bipartition) and sexual (conjugatio) by the pilus, with DNA • Decline phase • Plant pathogenic bacteria reproduce slower than animal ones TÁMOP-4.1.2.A/2-10/1-2010-0012 18 Environmental factors influencing the multiplication • Temperature: 2-5 oC minimum, 25-30 oC optimum, over 50 oC will die • Nutrients: water, carbohydrates, alkali pH. Autotrophic and heterotrophic type of living. Aerobe and anaerobe types • Light: Not as sensitives as the animal pathogens. UV light is harmful for DNA TÁMOP-4.1.2.A/2-10/1-2010-0012 19 Characterisation protocol of an unknown bacterium 1. • Isolation from diseased tissue on nutrient agar • Separation of colonies • Pathogenicity experiments on tobacco plants (HR) TÁMOP-4.1.2.A/2-10/1-2010-0012 Characterisation protocol of an unknown bacterium 2. • • • • • Testing of bacteria producing HR on tobacco plants Physiological and biochemical tests Pathogenicity tests Ice nucleation tests Production of antibiotics TÁMOP-4.1.2.A/2-10/1-2010-0012 Characterisation protocol of an unknown bacterium 3. Pathogenicity test • On young fruits • On young sprouts • On intact plants • On cuttings TÁMOP-4.1.2.A/2-10/1-2010-0012 Identification methods • Gram staining: (1884) pararosaniline (Gentiana violet+ KOH). Generally G-, but Clavibacterium G+ • Morphological properties: shape, form, flagella • In vitro studies: form, colour of colonies • Biochemical reactions: nitrate reduction, carbohydrate utilization, gelatine hydrolysis, gas production etc. • Pathogenicity according to Koch’s postulates TÁMOP-4.1.2.A/2-10/1-2010-0012 23 Gram (+) staining of bacterial cells Foto: T.Vigh TÁMOP-4.1.2.A/2-10/1-2010-0012 24 Diagnostic methods • • • • • Symptoms (macroscopic or microscopic) Isolation on artificial media, growing in pure cultures Colonies, type of colonies Microscopic studies (form, shape, flagella) Chemical diagnosis: production of different metabolites • Phage analysis • Molecular diagnosis: proteins and nucleic acids TÁMOP-4.1.2.A/2-10/1-2010-0012 25 Serological diagnosis of bacteria • Serological methods: - antigen – antibody relation, - serological reactions: agglutination (cell level), precipitation (in colloid form), immuno-electron microscopy, immuno blotts etc. Enyme Linked Serological Assay - ELISA • Antigens: lipoproteins, flagellin • Agglutination titre for estimation of taxonomic relation. Polyclonal and monoclonal antisera TÁMOP-4.1.2.A/2-10/1-2010-0012 26 Immunodiffusion test with serologically related strains Antiserum Antigen samples TÁMOP-4.1.2.A/2-10/1-2010-0012 27 Molecular diagnosis of bacteria • Extraction of proteins or total nucleic acids • Separation of proteins or nucleic acids by electrophoresis • Sequence analysis of nucleic acids by restriction endonucleases (produced by fungi) • PCR – polymerase chain reaction for amplification of nucleic acids • Hybridization (Western, Southern, Northern blot) TÁMOP-4.1.2.A/2-10/1-2010-0012 28 Phage analysis • 1915 Twort and D’Herelle – discovery of bacteriophages by specific lyses of bacterial colonies • Bacteriophages are viruses infecting bacteria (Delbrück and Luria): No independent metabolism, nucleoproteins, specific for bacterial genera • Adsorption, vegetative phase, lyses TÁMOP-4.1.2.A/2-10/1-2010-0012 29 Characteristics of phage infection • Adsorption, penetration of phage nucleic acid into the cell. Only one phage infects! • Vegetative phase: New protein synthesis, respiration does not change, division (reproduction) stops • Biosynthesis of new virions, disruption of cell walls (production about 10-300 new phages in a single bacterial cell) • Plaque formation (8-12 hrs). Plaque counting. Specific! TÁMOP-4.1.2.A/2-10/1-2010-0012 30 Transmission of bacteria • Mechanical way (through by wounds), through by the stomata and lenticels, etc. • By seed transmission, from the surface and from the endospermium • Pollen transmission: Erwinia amylovora • Vegetative mode: tubers, bulbs, grafting etc. • Vectors: insects, nematodes, humans etc. • Water (the most common) TÁMOP-4.1.2.A/2-10/1-2010-0012 31 Pathogenicity of plant pathogenic bacteria • Evolution of bacteria: saprophytic, symbiontic, parasitic (P. fluorescens - P. tabaci, AgrobacteriumRhizobacterium relationship) • Pathogenicity: toxin production, pectinases, hormones causing hypertrophy or other growth abnormalities TÁMOP-4.1.2.A/2-10/1-2010-0012 32 Infection conditions of phytopathogenic bacteria • Infection conditions: pathogen, susceptible plants, favourable environmental conditions • Infection by wounds and natural openings! • Starts fom the soil, through plant debrids, seeds, wounds of phloem and xyleme vessels • Vectors: insects, bees, etc. • Physiological and developmental stage of plants TÁMOP-4.1.2.A/2-10/1-2010-0012 33 Living types of bacteria Autotrophs (autonomous energy supply, species, using carbon dioxide of the air as carbon source), chemotrophs, photoautotrophs, etc. Heterotrophs (using organic carbon sources) TÁMOP-4.1.2.A/2-10/1-2010-0012 34 Bacterium domens • Archaea – thermophil fossil organisms from about 2,5 – 3 milliard years ago, altered type of evolution. Characterization on the basis of 16S ribosomal RNA • Bacteria - recent taxon, developed approx. 4 milliard years ago TÁMOP-4.1.2.A/2-10/1-2010-0012 35 Phases of bacterial infection • • • • • • Penetration into the intercellular space Avoid of defence reactions Alteration of host metabolism Genetic transfer by plasmids (Agrobacterium) Colonization of nutrient reach tissues Secondary colonization (soft rots) TÁMOP-4.1.2.A/2-10/1-2010-0012 36 Defence reactions against bacterial infection • General, non-specific defence reaction • Specific defence reaction (Hypersensitive reaction, HR) Resistant plant, incompatible - host parasite relation • Lack of defence reaction (susceptible plant) compatible host – parasite relation) TÁMOP-4.1.2.A/2-10/1-2010-0012 37 Disease induction (in susceptible host) • Susceptible plant (compatibility) • Avoid the recognition by EPS • Inhibition of HR (products of Avr genes do not react with the R genes) • Water and nutrient supply in the intercellular space • Alteration of pH (efflux of K ions and influx of H ions) • Moderate production of oxygen free radicals • Efflux of glucose into the intercellular spaces TÁMOP-4.1.2.A/2-10/1-2010-0012 38 Necrobiosis in the susceptible host • Rapid bacterial multiplication in the intercellular space (water soaking spots) • Glucose exhaustion, new bacteria will produced without EPS • Recognition of bacteria by the host • Bacterial reproduction stops, • Bacteria will be killed by the necrosis • Typical bacterial symptoms will develop (necroses, wilting, yellowing etc.) TÁMOP-4.1.2.A/2-10/1-2010-0012 39 Specific defece reaction (HR) • Induction phase: attachment of bacteria on the cell surface • Activation of hrp (genes of hypersensitivity and pathogenicity) in the bacterial cell • Latency phase: introduction of Avr or Vir gene products into the host cell • Activation of R (resistant) genes of the plant • Increase and accumulation of reactive oxygen radicals • Activation of antioxidants and antioxidant enzymes • Necrotic phase: Active destruction of pathogen • Programmed cell death (HR) TÁMOP-4.1.2.A/2-10/1-2010-0012 40 Genes of hypersensitivity and pathogenicity (hrp) • Characterization by transposon mutagenesis • Because the spoiled genes of the mutants do not induce HR, their reproduction rate decrease • Organized in gene clusters 25-30 kb. (in the „island of pathogenicity”) • Localized in the chromosomes or in plasmids (Ralstonia) • Incorporation into pathogens will induce HR • Proteins of the III. rd type of secretion mechanism TÁMOP-4.1.2.A/2-10/1-2010-0012 41 Hypersensitivity • Hypersensitivity is the hyperergic expression of incompatibility • During the HR by the rapid death of the host cell the bacteria will also be killed • In the majority of cases the HR leads to resistance (but itself it is not the resistance, only the consequence of the resistance) TÁMOP-4.1.2.A/2-10/1-2010-0012 42 Main types symptoms caused by bacterial infection • Leaf spots, local necroses (Pseudomonas, Xanhomonas) • Cankers , wounds (Pseudomonas syringae, Erwinia amlylovora) • Wiltings (irreversible and reversible) (Ralstonia solanacearum) • Tuber soft rot (Erwinia carotovora) • Tumors (Agrobacterium tumefaciens) • Scab (Streptomyces scabies) TÁMOP-4.1.2.A/2-10/1-2010-0012 43 Necroses, leaf spots • Local spots, water soaked spots, later necroses • Necrotic spots on the leaves, stems, flowers and fruits • Bacterial slime • Chlorosis (effect of toxins), wilting • Largr necroses, cankers • Usually caused by Pseudomonads and Xanthomonads TÁMOP-4.1.2.A/2-10/1-2010-0012 44 Water soaking and necrosis (Pseudomonas syringae pv. phaseolicola) Photo: S. Kadlicskó TÁMOP-4.1.2.A/2-10/1-2010-0012 45 Necroses - Cankers • Cankers of woody plants: Pseudomonas morsprunorum • Apoplexy: Necrogenic bacteria, ice nucleation, exhaustion of glucose in the vascular system, frost injury, necrosis in cambium. Caused by: Pseudomonas syringae pv. syringae • Fire blight of apple trees: Bacterial mass blocks the nutrient transport, causing wilting and necrosis on twigs and trunks. Caused by: Erwinia amylovora. TÁMOP-4.1.2.A/2-10/1-2010-0012 46 Necrosis of fire blight (Erwinia amylovora) TÁMOP-4.1.2.A/2-10/1-2010-0012 47 Wilting • • • • Blocking of vascular system (ring necrosis) Wilting (sometimes temporary) Ralstonia solanacearum wilt Curtobacterium flaccumfaciens, P. syringae pv. phaseolicola, Xanthomonas campestris pv. phaseoli TÁMOP-4.1.2.A/2-10/1-2010-0012 48 Wilt caused by vascular block Ralstonia solanaceraum blocks the vascular system and causes temporary wilting of potato plants and ring necrosis on tubers TÁMOP-4.1.2.A/2-10/1-2010-0012 49 Soft rots caused by bacteria • Tubers of potato, bulbs of onion, cabbage head • Pectolitic enzymes (pectinases, pectin metylesterases) lisate the tuber tissues • Pathogenicity depends on the enzyme activities • Erwinia carotovora group TÁMOP-4.1.2.A/2-10/1-2010-0012 50 Soft rot of carrots caused by Erwinia carotovora Fotos: T. Vigh TÁMOP-4.1.2.A/2-10/1-2010-0012 51 Potato soft rot (Erwinia carotovora) TÁMOP-4.1.2.A/2-10/1-2010-0012 52 Bacterial tumors • Cell and tissue proliferation in Dicots by the transfer of bacterial plasmid DNA • Crown gall caused by Agrobacterium tumefaciens, A. vitis • Pseudomonas savastanoi on oil trees TÁMOP-4.1.2.A/2-10/1-2010-0012 53 Tumors on olive trees (Pseudomonas savastanoi) TÁMOP-4.1.2.A/2-10/1-2010-0012 54 Crown gall caused by Agrobacterium vitis TÁMOP-4.1.2.A/2-10/1-2010-0012 55 Actinomyces are bacteria, not fungi! • 1875 first report on potato scab • 1891 Streptomyces scabies • Formerly independent taxon, recently belongs to bacteria (Actinomycetales, Streptomyces) • No real nucleus, cell wall does not contain chitin TÁMOP-4.1.2.A/2-10/1-2010-0012 56 Plant pathogenic bacteria Economically important bacterial diseases Taxonomy of most important bacteria Class Family Genus Alpha Proteobacteria Rhizobiaceae Agrobacterium Beta Proteobacteria Ralstoniaceae Ralstonia Gamma Proteobacteria Pseudomonadaceae Pseudomonas Xanthomonadaceae Xanthomonas Enterobacteriaceae Erwinia Actinobacteria Microbacteriaceae Corineabacteriaceae Streptomycetaceae TÁMOP-4.1.2.A/2-10/1-2010-0012 Clavibacter Curtobacterium Rhodococcus Streptomyces Taxonomy of Pseudomonas genus • • • • • • Bacteria (Gram -) Phylum: Proteobacteria Class: Gamma Proteobacteria Order: Pseudomonadales Family: Pseudomonadaceae Genus: Pseudomonas • Inside the Pseudomonas genus there are animal-, human- and plant pathogenic species TÁMOP-4.1.2.A/2-10/1-2010-0012 59 Plant pathogenic Pseudomonas species • Pseudomonas species: • Pseudomonas syringae: species with wide range of hosts, over 50 different pathovars • Pseudomonas savastanoi – infects oil trees • Pseudomonas viridiflava – infects sweet pepper TÁMOP-4.1.2.A/2-10/1-2010-0012 60 Pseudomonas syringae • Rod shaped, Gram-negative bacterias with polar flagella • Infect a range of plant species • Presence of INA (ice nucleation active) proteins • Infection: wet, cool temperature (optimal 12-25 Co), could be seed-borne, can live as saprophyte • Pathovars: - P. s. pv. atrofaciens – infects Triticum aestivum - P. s. pv. pisi – infects Pisum sativum - P. s. pv. syringae – infect Syringa, Prunus, Phaseolus species TÁMOP-4.1.2.A/2-10/1-2010-0012 61 Bacterial necroses on tobacco leaf Serious disease of tobacco plants caused by Pseudomonas syringae pv. tabaci TÁMOP-4.1.2.A/2-10/1-2010-0012 62 Pseudomonas syringae pv. syringae TÁMOP-4.1.2.A/2-10/1-2010-0012 63 Apoplexy of apricot trees • • • • Rapid wilting and necrosis of apricot branches Necrosis in vascular system Causal agent: Pseudomonas syringae pv. syringae Bacteria multiply in vascular tissues, utilizing its sugar content • Ice nucleating strains cause frost damage and stem necrosis • Control: removal of infected parts (truncation) • Pruning not in winter, but in spring TÁMOP-4.1.2.A/2-10/1-2010-0012 64 Truncated apricot tree after bacterial infection TÁMOP-4.1.2.A/2-10/1-2010-0012 65 Died apricot tree : Apoplexy TÁMOP-4.1.2.A/2-10/1-2010-0012 66 Pseudomonas syringae pv. phaseoli TÁMOP-4.1.2.A/2-10/1-2010-0012 67 Pseudomonas viridiflava • Fluorescent soil borne pathogen infects tomato (stem necrosis, dark blotches on pruning sites of the stem), soft rot on sweet pepper, the runner beans etc. TÁMOP-4.1.2.A/2-10/1-2010-0012 68 Pseudomonas viridiflava infection on sweet pepper Photo:Z. Klement TÁMOP-4.1.2.A/2-10/1-2010-0012 69 Pseudomonas savastanoi TÁMOP-4.1.2.A/2-10/1-2010-0012 70 Taxonomy of Xanthomonas genus • Gram negative bacteria, with rod shaped flagella bacteria grow almost exclusively in plants • Phylum: Proteobacteria • Class: Gamma Proteobacteria • Order: Xanthomonadales • Family: Xanthomonadaceae • Genus: Xanthomonas TÁMOP-4.1.2.A/2-10/1-2010-0012 71 Xanthomonas campestris • • • • • • • • Cause a large variety of plant diseases Pathovars classification based on the host plant Important pathovars: - pv. campestris. - pv. caroteae - pv. juglandis - pv. malvacearum - pv. pelargonii TÁMOP-4.1.2.A/2-10/1-2010-0012 72 Xanthomonas campestris pv. vesicatoria • Aerobic. Gram negative, mobile (single polar flagellum) • Principal hosts: tomatoes, sweet pepper and other Solanaceous plants • Occurs widely in pepper and tomato growing areas, infects under warmer, humid conditions (above 30 oC) • Survives in seeds, infested debris, stalks • In glasshouses seed borne infection (survive on tomato and pepper seeds for 10 years) • Disseminated by rain splash, irrigation, handling, aerosols TÁMOP-4.1.2.A/2-10/1-2010-0012 73 Symptoms on pepper caused by X. vesicatoria pv. vesicatoria • Corky spots and scabs, water soaking margins , black necrotic lesions on the leaves with yellow haloes TÁMOP-4.1.2.A/2-10/1-2010-0012 74 Xanthomonas campestris ssp. vesicatoria Control: healthy seed, care in handling, phytosanitary measures, resistant variety TÁMOP-4.1.2.A/2-10/1-2010-0012 75 Xanthomonas campestris pv. juglandis TÁMOP-4.1.2.A/2-10/1-2010-0012 76 Ralstonia solanacearum • Quarantine, aerobic, Gram-negative, soil-borne bacteria with polar flagella • One of the most important bacterial pathogen, infects over 250 species of 50 families • Phylum: Proteobacteria • Class: Beta Proteobacteria • Order: Burkholderiales • Family: Ralstoniaceae • Genus: Ralstonia TÁMOP-4.1.2.A/2-10/1-2010-0012 77 Ralstonia solanacearum • Host range: potato, tomato, pepper, tobacco, eggplant, banana, etc. • Symptoms: reversible, later irreversible wilting of the leaves. Bacterial exudates accumulate in the vascular system. Ring necrosis in potato tubers • Overwinters in diseased plants, plant debris, in seed and tubers. Occurs in surface waters, including rivers. Can be spread by irrigation , mechanical tools and by seeds • Infects by wounds. Wilting occurs at high concentration of bacteria in the xylem (EPS) TÁMOP-4.1.2.A/2-10/1-2010-0012 78 Erwinia amylovora • Contagious, quarantine, Gram-negative bacteria, causal agent of „fireblight disease” of apple trees, affecting apples, pears, quinces and other members of family Rosaceae (Cotoneaster, Pyracanthus etc.), Class: Gamma Protobacteria • Order: Enterobacteriales • Family: Enterobacteriaceae • Infects in warm, wet conditions in spring in blossom time. Affected areas are dying quickly and tender new shoots and leaves. Bacterial exudates • Dissemination by rain, honeybees, birds and insects TÁMOP-4.1.2.A/2-10/1-2010-0012 79 „Shepherds crook” symptom of fire blight Photo: Cs. Pintér TÁMOP-4.1.2.A/2-10/1-2010-0012 80 Symtoms of late infection caused by Erwinia amylovora TÁMOP-4.1.2.A/2-10/1-2010-0012 81 Secondary infection of fire blight on pear TÁMOP-4.1.2.A/2-10/1-2010-0012 82 Control of fire blight disease • • • • • Resistant (?) variety Rather difficult. Routine inspection in the orchard Removal and burning of infected branches Antibiotics are not allowed! Chemical control by copper containing fungicides with special care of honeybees • Quarantine measures TÁMOP-4.1.2.A/2-10/1-2010-0012 83 Erwinia infection on apple shoots TÁMOP-4.1.2.A/2-10/1-2010-0012 Apple shoot necrosis TÁMOP-4.1.2.A/2-10/1-2010-0012 Canker of apple trees caused by Erwinia amylovora TÁMOP-4.1.2.A/2-10/1-2010-0012 86 Erwinia carotovora • Rod shaped Gram negative, peritrichously flagellated facultative anaerobe bacterium • Class: Beta Proteobacteria, • Order: Enterobacteriales, • Class: Enterobacteriaceae • Causes diseases of many plants • Infects a wide host range, causing soft rot on carrots, potato, tomato, cucurbits, onion, etc. • Important as post-harvest pathogen • Produce proteolitic enzymes (pectinases, cellulases) TÁMOP-4.1.2.A/2-10/1-2010-0012 87 Erwinia carotovora on cabbagge TÁMOP-4.1.2.A/2-10/1-2010-0012 88 Erwinia carotovora infection TÁMOP-4.1.2.A/2-10/1-2010-0012 89 Erwinia carotovora on carrot Photos: T. Vigh TÁMOP-4.1.2.A/2-10/1-2010-0012 90 Soft rot of paotato caused by Erwinia carotovora TÁMOP-4.1.2.A/2-10/1-2010-0012 91 Potato soft rot TÁMOP-4.1.2.A/2-10/1-2010-0012 92 Test for pectolitic enzymes on potato slices Photo: M. Kállay TÁMOP-4.1.2.A/2-10/1-2010-0012 93 Agrobacterium tumefaciens • Rod shaped, Gram negative soil bacterium with flagella • Causal agent of crown gall disease • Ti plasmid is responsible for tumor formation • Class: Alpha Proteobacteria • Order: Rhizobiales • Family: Rhizobiaceae • Genus: Agrobacterium TÁMOP-4.1.2.A/2-10/1-2010-0012 94 Infection by Agrobacterium tumefaciens • Bacteria chemotactically move to the root cells • Vir A and transmembrane proteins recognize the cell exudates • Bacteria synthesize cellular fibrils, anchoring bacteria to the host cell • Cut out of transfer DNA (T-DNA) from the circular (Titumor inducing) plasmid • One copy of T-DNA (activated by vir genes) transferred into the plant cells by T pilus • Incorporation of T-DNA into the plant chromosome TÁMOP-4.1.2.A/2-10/1-2010-0012 95 Genes in the Ti plasmid transfer (T) DNA • • • • Vir genes cut out T-DNA region from Ti plasmid Gene of indole acetic acid (IAA) production Gene of cytokinin production Gene for encoding opines (octopine and nopaline • Only one copy of T-DNA is exported to the host cell! TÁMOP-4.1.2.A/2-10/1-2010-0012 96 Agrobacterium as gene vector • Cloning and amplification of a desired gene (DNA) sequences (e.g. in Escherichia coli) • Incorporation of the cloned DNA into Agrobacterium tumefaciens Ti plasmid (transformed bacteria) • Agroinfection of isolated plant cells or protoplast by transformed A. tumefaciens • Transformation of host cells by modified Ti plasmid containing the desired gene • In vivo culture of transformed cells to plants in tissue cultures TÁMOP-4.1.2.A/2-10/1-2010-0012 97 Tumor formation on fruit tree Infection of Agrobacterium means a natural genetic transformation, a genetically modified plant! TÁMOP-4.1.2.A/2-10/1-2010-0012 98 Crown gall caused by Agrobacterium TÁMOP-4.1.2.A/2-10/1-2010-0012 Tumor formation of Agrobacterium vitis • A. vitis DNA has small differences in its DNA from A. tumefaciens •A. vitis causes systemic infection on grapes! •Properties: similar to Agrobacterium tumefaciens •Control: Eradication, sanitary measures TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Agrobacterium rhizogenes • • • • Symptoms: root proliferation Host plants: fruit trees Agent: Agrobacterium rhizogenes Contains Ri plasmid responsible for the infection and symptoms • Control: see A. tumefaciens TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Control of bacterial diseases • Use of resistant (or tolerant) varieties • Quarantine measures: Erwinia amylovora, Ralstonia solanacearum • Agro technical methods: prevention, hygiene, prevention of transmission, soil disinfection • Chemical methods: antibacterial chemicals (no antibiotics are allowed !) • Biological methods: antagonistic bacteria, e.g. Agrobacterium K84 strain produces bacteriocin against Ti plasmid containing Agrobacterium strains TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Agrotechnical and chemical control • Keep the quarantine rules • Agro technical methods: • Proper nutrient supply, isolated propagation, preventive measures to avoid of infection, soil disinfection, forecasting • Hygienic rules • Chemical control: bactericides, no antibiotics allowed! TÁMOP-4.1.2.A/2-10/1-2010-0012 Use of resistant varieties to bacterial infection Resistant and susceptible cabbage lines in the experimental field Photo: Z. Klement TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Taxonomic position of Actinomycetes • Group of soil-and water-borne, Gram positive organisms, probably the oldest living organism on Earth • Domain: Bacteria • Phylum: Actinobacteria • Class: Actinobacteridaceae • Order: Actinomycetales • Specific group of bacteria, morphologically resembling to the fungi, because of elongated cells or filaments or hyphae. TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Streptomyces scabies • Unusual filamentous pathogen, similar to fungi • Causative agent of common scab of potato and other root crops • Present in soils of potato growing areas • Symptoms: russet and pitted lesions, later necrotic areas on the surface, corky tissues • Gray, spiral vegetative hyphae are fragmented into spores • Spores survive in the soil, spread through water infect by wounds TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Phytoplasma • A distinct group of plant pathogenic organisms, belonging to the bacteria • There are living in the intercellular space and in the phloem tissues of the plants • Its genome contains both DNA and RNA • Have no differentiated cell wall, only cell membrane • Therefore, their shape is generally round, but variable (pleomorph), about 100 nm in diameter • Spread by leafhoppers in a persistent manner • Diseases can be cured by antibiotics TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Most important properties of phytoplasma • 1967 Yoi Doi et al. separation from viruses according to the type of genome (RNA and DNA) • Propagation in artificial media is not successful • Two living cycles: in planta and in the vector • Symptoms: dwarfing, yellowing, deformation of flowers, witches broom, hormonal abnormalities • Taxonomy: formerly according to the symptoms, recently: Candidatus, on the basis of 16r (ribosomal) DNA TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Special characteristics of phytoplasma • Now total DNA sequences of two species (AYWB and OY) are known (530-1350 kb), with about 671-754 genes • Have plasmids (similarly to bacteria) • There are missing some genes characteristic to bacteria (for de novo amino acid, fatty acid, nucleotide- and ATP synthesis) • There are depended on the metabolism of their host (plant or leafhopper) • Membrane transport is affected (no tubulin has found necessary for cell division) • The most primitive forms of auto replication • Because of repetitive DNA sequences, horizontal gene transfer is possible (recombination among the chromosomes and the plasmids) TÁMOP-4.1.2.A/2-10/1-2010-0012 10 Aster yellows phytoplasma • Chronic, phloem limited bacterial-like organisms affects approx. 300 species in 38 families (onion, letuce, celery, carrot etc.) • Symptoms: virescence, chlorosis, stunting, sterility of flowers • Vectored by leafhopper Macrosteles quadrilineatus • Control: infected plants should be removed and eliminated. Control of insect vectors and weeds. No cure possibilities TÁMOP-4.1.2.A/2-10/1-2010-0012 11 Aster yellows phytoplasma on cabbage TÁMOP-4.1.2.A/2-10/1-2010-0012 11 Stolbur phytoplasma • The most common phytoplasma agent in Hungary • Spreads by leafhoppers • Infects tomato (flower deformation) - green pepper (wilting) - potato (air bulbs) - tobacco (flower abnormalities) - grapes (yellowing, redding, gummy shoots) - and a lot of other plants TÁMOP-4.1.2.A/2-10/1-2010-0012 Flower abnormalities caused by stolbur phytoplasma TÁMOP-4.1.2.A/2-10/1-2010-0012 11 Yellowing and formation of new branches of tomato TÁMOP-4.1.2.A/2-10/1-2010-0012 11 Stolbur phytoplasma on tobacco TÁMOP-4.1.2.A/2-10/1-2010-0012 11 Wilt of pepper plants caused by Stolbur phytoplasma TÁMOP-4.1.2.A/2-10/1-2010-0012 11 Aerial bulbs on Stolbur phytoplasma infected potato TÁMOP-4.1.2.A/2-10/1-2010-0012 11 Convolvulus sp. infected by stolbur phytoplasma TÁMOP-4.1.2.A/2-10/1-2010-0012 Stolbur infection on almond and Polygonathum TÁMOP-4.1.2.A/2-10/1-2010-0012 Stolbur infection on white grape TÁMOP-4.1.2.A/2-10/1-2010-0012 Matured and non matured grape shoots TÁMOP-4.1.2.A/2-10/1-2010-0012 European stone fruit phytoplasma • • • • Infects fruit trees (apricot, peach, plum, etc.) Transmitted by leafhoppers in persistent manner Control of vector species Characterization according to 16S rDNA TÁMOP-4.1.2.A/2-10/1-2010-0012 Symptoms caused by European stone fruit phytoplasma TÁMOP-4.1.2.A/2-10/1-2010-0012 Symptoms caused by European stone fruit phytoplasma TÁMOP-4.1.2.A/2-10/1-2010-0012 Clover green pethal phytoplasma • • • • Phyllody and greening of flowers Disseminated by leafhoppers No economic importance Differentiation on the basis of 16S rDNA TÁMOP-4.1.2.A/2-10/1-2010-0012 Clower green petal phytoplasma Healthy Diseased TÁMOP-4.1.2.A/2-10/1-2010-0012 12 Növénypatogén baktériumok Dr. Richard Gáborjányi Georgikon Kar Növényvédelmi Intézet AZ ELŐADÁS LETÖLTHETŐ: -