* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Morphological Plasticity of the Mitotic Apparatus in
Survey
Document related concepts
Tissue engineering wikipedia , lookup
Endomembrane system wikipedia , lookup
Microtubule wikipedia , lookup
Cell encapsulation wikipedia , lookup
Extracellular matrix wikipedia , lookup
Programmed cell death wikipedia , lookup
Biochemical switches in the cell cycle wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cell culture wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Cell growth wikipedia , lookup
Kinetochore wikipedia , lookup
Spindle checkpoint wikipedia , lookup
List of types of proteins wikipedia , lookup
Transcript
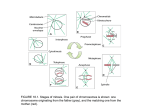
The Plant Cell, Vol. 5, 1001-1009, September 1993 O 1993 American Society of Plant Physiologists REVIEW AFITICLE Morphological Plasticity of the Mitotic Apparatus in Plants and Its Developmental Consequences Barry A. Palevitz Department of Botany, University of Georgia, Athens, Georgia 30602 INTRODUCTION More than 100 years after the discovery of mitosis, the role of the mitotic apparatus (MA) in cell and organismal development is still the subject of lively debate (e.g., Kaplan, 1992). The developmental impact of cell division looms perhapseven larger in plants than in other organisms because plant cells are not free to migrate during morphogenesis. Therefore, plants place great stock in modulating division planes to produce new directions for growth (Sinnott, 1960). In addition, precisely placed walls serve to set off unequal compartmentsthat then differentiate in divergent pathways. Thus, although their principal function, partitioning two equal sets of chromosomes to separate cytoplasmic compartments, remains the same, mitosis and cytokinesis assume an additional dimension in plants. Whereas animals also depend on precise division planes (Strome, 1993), the process occupies an especially prominent role in plant development (Gunning, 1982). My main purpose in this review is not to address division plane determination per se but to explore a characteristic of plant mitosis that has received much less attention. The subject of my discussion is the morphologicalplasticity of the MA, that is, its ability to be tilted, squeezed, or otherwise deformed under the confining influence of the cell wall and still work. Given new information about a number of proteins, we may be on the verge of understanding the molecular basis of mitotic architecture. Furthermore, with a fuller appreciation of mitotic plasticity, division plane determination can be seen from another perspective-that of a mechanismto compensate for MA asymmetryor deformation. During my discussion, 1’11 also make a brief foray into the evolution of microtubule organization patterns in plants. EXAMPLES OF PLASTlClTY OF THE MlTOTlC APPARATUS IN PLANTS The MA, also called the mitotic spindle or spindle apparatus, is the assemblage of microtubules and associated proteins responsible for separating the duplicated chromosomes. We tend to think of the MA as a symmetrical, spindle-shaped structure whose longitudinal axis is oriented perpendicular to the plane of the future cell plate. That may be true in many cases, but it is not so in many others. First of all, unlike the spindles in most animal cells, the MA in plants is anastral; its poles do not focus on distinct centrosomes from which astral microtubules radiate. Instead, it is roughly barrel shaped, its poles are relatively wide, and the only “astral” microtubules it contains are those that arise around the daughter nuclear masses in late anaphase. Hence, I refer to it here as the mitotic apparatus and not the spindle. In addition, the MA in most somatic plant cells is often tilted or distorted in various ways. For example, metaphase MASin root apical meristems are often obliquely oriented relative to the cell axes rather than longitudinallyor transversely aligned. Some MASin an onion root tip appear to be oblique, as shown in Figure 1A (Ngmec, 1899; Palevitz and Cresti, 1989), but many exhibit this arrangement in the cortical cells of broad bean roots (Oud and Nanninga, 1992). Nearly 1000/0 of the MASin guard mother cells of onion cotyledons are oblique, as shown in Figure 1B (Palevitz and Hepler, 1974; Palevitz, 1986), and similarly oriented MASare seen in guard mother cells of other species (Cho and Wick, 1989; Cleary and Hardham, 1989), as well as in fusiform cambial initials (Bailey, 1920), hair cells, and rib meristem cells (Bglãr, 1929; Wada, 1965). Not only is it often oblique, but also in some cases the MA is clearly distorted. For example, S-shaped MAS are present in a variety of cell types, including guard mother cells (BBlãr, 1929; Palevitz and Hepler, 1974). Distortion of the MA is perhaps most pronounced in the generative cell of Tradescanfia virginiana (spiderwort) (Palevitz and Cresti, 1989; Liu and Palevitz, 1991). During much of the division that creates the two sperm cells inside the growing pollen tube, the chromosomes are arranged in twisted, sometimes braided fashion. The kinetochores, labeled in Figure 1C with a human autoimmune serum (CREST EK; Liu and Palevitz, 1991), are distributed along the length and depth of the generative cell, often in tandem fashion, and are not clustered on an obvious metaphase plate. Kinetochore fibers are interlinked in tandem, 1002 The Plant Cell Figure 1. Examples of Deformed MAs in Three Cell Types. (A) Obliquely oriented metaphase or early anaphase MA in a root cell of onion viewed after immunofluorescence labeling of microtubules. Kinetochore fibers are grouped in clusters that focus separately at relatively broad poles. Bar = 10 urn. (B) Late anaphase in a live guard mother cell of onion viewed with differential-interference contrast optics. The kinetochores are clustered near the corners of the cell, while the chromosome arms trail back toward the midzone. Same scale as (A). (C) Arrangement of kinetochore pairs in a metaphase generative cell of Tradescantia. The kinetochores, which were visualized by immunofluorescence using CREST serum, are arranged along the length and depth of the cell. Twelve pairs are evident, consistent with n = 12 in this species. A pair near the center is somewhat blurry, while the one at the far right is even more out of focus. Bar = 10 urn. From Liu and Palevitz (1991), with permission of the Company of Biologists, Ltd. and the metaphase MA not only lacks obvious bipolar architecture but also shows little resemblance to a conventional MA (Liu and Palevitz, 1991; Palevitz and Tiezzi, 1992). The Tradescantia generative cell MA falls on one end of a morphological continuum with conventional MAs at the other end and oblique and less seriously distorted MAs in between. Further evidence in support of this conclusion comes from observations that the Tradescantia MA is not typical of generative cells. Indeed, the MA in tobacco generative cells looks much more conventional, although it is almost always oblique and often shows signs of distortion (Palevitz, 1993). The MA therefore possesses a great deal of morphological plasticity. At first glance, unusual, seemingly abnormal configurations of the MA would seem to present significant developmental problems. Yet, amazingly, these distorted MAs still move chromosomes to the poles during anaphase, after which cytokinesis separates them into predetermined cytoplas- mic compartments. Division apparently proceeds with fidelity, because the progeny of guard mother cell division, for example, differentiate into a pair of mirror-image, functional guard cells, and generative cell division produces two similar sperm cells. Interestingly, the ability of deformed MAs to function correctly has been put to experimental use: flattened Haemanthus katherinae endosperm cells have provided excellent material to study mitosis and cytokinesis in plants (e.g., Inoue, 1964; Bajer, 1990). REASONS FOR MITOTIC APPARATUS DEFORMATION Why is the MA inclined or deformed in so many cells, and what properties of the MA account for this morphological plasticity? Two possible answers to the first question come Mitotic Plasticity in Plants immediately to mind. Either the MA is deformed because of mechanical constraints determined by cell geometry as defined by the cell wall or MA morphologyis governed by genetic and developmentalprograms expressed in each cell. A great deal of evidence strongly favors the first conclusion, but other data indicate that the second is important as well, at least in certain cell types. Numerous investigators have proposed that space constraints are responsible for oblique or deformed MAS. For example, Bailey (1920) mentionedthis as an obvious explanation for the oblique axes seen during radial longitudinal divisions of fusiform cambial initials. In cells of various wheat species, the inclination of the MA increases with the ploidy leve1 because cell volume does not increase proportionally to chromosome volume (Wada, 1965). Recently, Oud and Nanninga (1992) argued in favor of a prominent role of cell shape in MA orientation in root cells. lnterplay between cell geometry and the volume of the chromosomes andlor MA is also indicated by observations on the oblique divisions in onion guard mother cells. This cell is destined to divide longitudinally, as indicated by a longitudinal preprophase band (see below). When the MA first appears in prophase, it is aligned transversely, which might be expected based on its preset division polarity. However, as the MA grows in size, it shifts to an oblique orientation; by late metaphase, most MASare inclined (Mineyuki et al., 1988).Thus, as the MA becomes more confined by the wall, it occupies the maximum available dimensions (i.e., it becomes oblique). Space constraints have repeatedly been invoked to explain the arrangement of chromosomes in Tradescantia generative cells (Palevitz and Tiezzi, 1992); to wit, this is a fairly large cell (with 12 large chromosomes) that must divide within the confines of the narrow pollen tube. Hence, the chromosomes cannot fit on a single metaphase plate. Because the MA is flexible, it is deformed as well. It is noteworthy in this regard that in generative cells (e.g., of tobacco) with smaller chromosomes and MAS,the distortion is much less severe (Palevitz, 1993). On the other hand, MA morphology andlor alignment may be specifically programmed. For example, whereas Bailey (1920) suggested that the MASof fusiform cambial initials undergoing radial longitudinal divisions are oblique due to space constraints, the same argument cannot be used to explain the oblique pseudotransverse @e., tangential) divisions that increase the cambial circumference, where there often appears to be sufficient room to accommodate the MA. The recent results of Staiger and Cande (1990,1992) have shown that MA morphology and alignment have a genetic basis. lnstead of the sharply focused poles characteristic of wild-type maize meiocytes, spindles in the divergentspindle mutant have broad, divergent poles. Pointed spindles are also routinely found in cultured cells of spruce and larch (Fowke et al., 1990; I. Staxh and C. Bornman, personal communication). Interestingly,opposite poles of the same MA can differ in organization.During the first, asymmetrical division of pollen grains in orchids, the pole adjacent to the site of the future generative cell is broad, 1003 while that in the future vegetative cytoplasm is more pointed (Brown and Lemmon, 1992). MECHANISMS OF MlTOTlC APPARATUS PLASTlClTY Whether its morphology is determined by cell wall constraints or genetic programming, the MA must have properties that impart plasticity, that is, allow it to be deformed yet carry out its function successfully. The organizationof the spindle poles and the operation of specific proteins may help account for this plasticity. Organization of the Poles Many of the microtubules in the MA are organized into groups called kinetochore fibers, based on their attachment to specialized regions of the chromosomal centromeres known as kinetochores.Thus, chromosomes are attached to the MA via their kinetochorefibers (Baskin and Cande, 1990). Moreover, it is on the kinetochore fibers that the chromosomes actually move to the poles. In most animal cells, each pole consists of adistinct centrosome upon which the spindle microtubules sharply focus (Cande, 1990). The centrosomes serve as microtubule organizing centers (MTOCs), generating spindle microtubules from a shell of nucleating material surrounding two centrioles. Additional astral microtubules radiate in all directions away from the centrosomes. In contrast, the acentriolar, anastral MASof higher plants generally have broad poles (Bajer, 1990; Baskin and Cande, 1990).The maize meiocytes and cultured spruce cells already mentioned are notable exceptions. In fact, through much of division the MA in most plant cells is actually multipolar (Figure lC),in that groups of kinetochore fibers in each half of the MA converge at several separate foci (e.g., Schmit et al., 1983; Bajer, 1990; Smirnova and Bajer, 1992). The poles do not contain a single obvious, focused centrosome. Therefore, because the kinetochore fibers may be anchored at several locations at each broad pole, a loose, splayed arrangement of the MA is possible. That is, the kinetochore fibers appear to have a high degree of positional freedom relativeto each other. This looser arrangement at the poles may be crucial in allowing the MA to adjust to spatial constraints in narrow or small cells. This argument is reinforced by the observations of Cleary et al. (1992a) on the division of monoplastidic guard mother cells of Selaginella. Newly divided plastids are the sites of MA nucleation in these cells, as they are in many other lower plants (Busby and Gunning, 1989; Brown and Lemmon, 199Oa). Spindle orientation is also governed by the plastids, which migrate to positions along the sides of the cells. Unlike in onion, the MA remains transverse and never becomes oblique, even though the Selaginella guard mother cell is small. Although the poles are broad, they are associatedwith the plastids. Thus, because they maintain their lateral position during division, 1004 The Plant Cell we can assume that the plastids anchor the MA in a transverse orientation. We can then deduce that in the absence of this anchorage, i.e., in higher plants, the broad poles would be free to rotate. Any model of MA plasticity must also account for the impressive number of interlinkages between kinetochore fibers in plant cells. That is, numerous microtubulessplay from each fiber (like the branches of afir tree; Palevitz, 1988; Bajer, 1990), only to link up with neighboring fibers. Similar observations have been made in algae (Schibler and Pickett-Heaps, 1987). In fact, it can be argued that there are few nonkinetochore microtubules; at some level, most are associated with the kinetochore fibers. These linkagesappear to be crucial for MA organization and function (Bajer, 1990; Smirnova and Bajer, 1992), because kinetochore fibers are linked to their neighbors along their entire length. Surrounding endoplasmic reticulum membranes could provide additional stabilization (as well as function in Ca2+regulation; Hepler, 1992). Thus, the whole MA plus poles could move in a coordinated fashion in responseto spatial constraints. Alternatively, the shape of the MA could be deformed while maintaining the links between neighboring kinetochore fibers. Taking into account the interkinetochore fiber linkages and the structure of the poles, Schibler and Pickett-Heaps(1987) wondered whether the integrity of the MA is maintained in a different fashion than in animal cells. Roles of Specific Proteins The MA is composed of numerous proteins in addition to tubulin. Many of these govern its function, but others probably play major roles in determining its physical properties, including its shape and plasticity. Considerable recent attention has been directed at motor proteins such as those belonging to the kinesin and dynein superfamilies (e.g., Bloom, 1992). It now appears that kinesinsmay be important in defining MA architecture during mitosis and meiosis (Hatsumi and Endow, 1992; Saunders and Hoyt, 1992; Sawin et al., 1992). Although kinesins have been studied in diatoms (Hogan et al., 1992), few data are available from higher plants (and none deals with the MA; Cai et ai., 1993). Although plants lack discrete, focused centrosomes of the kind seen in animal cells and fungi, they probablycontain centrosomal protein homologs that control conserved aspects of microtubule nucleation (Lambert, 1993). Numerous reports have now appeared on centrosomal constituents in other organisms, including phosphoproteins and other potential regulatory elements (e.g., Vandr6 et al., 1984; Rout and Kilmartin, 1990; Balczon and West, 1991; Leslie et al., 1991; Maekawa et al., 1991). The challenge isto identify plant centrosomal proteins and account for their unique spatial distribution patterns (see below). Unfortunately, attempts to identify such proteins using heterologous antibody probes have produced mixed results (Baskin and Cande, 1990; Palevitz, 1991; Chevrier et al., 1992; Traas et al., 1992). New work on y-tubulin may ameliorate this situation, however. ylrubulin is a newly discovered member of the tubulin superfamily(Oakley, 1992), which also includes a- and P-tubulins (Fosket and Morejohn, 1992). It was discovered in Aspergillus nidulans and later found in other organisms (fission yeast, Drosophila, mammals), where it is present at relatively low abundance and concentratedat MTOCs such as spindle pole bodies and centrosomes. By virtue of interactions between y- and P-tubulin mutations, it has been proposed that the protein may nucleate and/or anchor microtubulesand set up their characteristic minus end proximal polarity relative to the centrosome (Oakley, 1992). Microtubulesare polar structures, and their two ends differ kinetically. The end located dista1 to an MTOC is called the plus end and is distinguished by faster rates of assembly and disassemblythan the minus end, which is anchored at an MTOC (Fosket and Morejohn, 1992). Recently, y-tubulin has been identified in the cells of onion, soybean, tobacco, and Arabidopsis (Liu et al., 1993; McDonald et al., 1993) using an antibody generated against a synthetic peptide corresponding to a conserved region of the protein (Joshi et al., 1992). y-Tubulin genes have also been cloned in Arabidopsis (C. D. Silflow and D. P. Snustad, personal communication). The polypeptide has an M, of 58,000 (compared to49,OOO in fungi and animals) and is more widely distributed along plant microtubule arrays (Liu et al., 1993). For example, during prophase, the plant protein becomesclusteredover the nuclear caps, which are broad regions at the ends of the nuclei associated with MA formation. In metaphase, it is localized over long stretches of the kinetochore fibers but not near the plus ends at the kinetochores. Thus, y-tubulin is biased toward the minus ends of microtubulesas in animal cells (see Oakley, 1992, for review; Joshi et al., 1992; Gueth-Hallonet et al., 1993). In anaphase, y-tubulin again aggregates over broad, caplike regions of the poles. yTubulin has also been localized over microtubules and their organizing centers in lower plants (K.C. Vaughn, personal communication). The broad spatial distribution of y-tubulin, as well as new information at the molecular level, may help to explain fascinating aspects of microtubuleorganizationin plants, includingthe structure and interconnections of kinetochore fibers, behavior of the poles, and, hopefully, MA morphology. EVOLUTiON OF PLANT MICROTUBULE ARRAYS AND THE MlTOTlC APPARATUS Anastral, multipolar MASare probably one of many cytoskeletal modifications involved in the evolution of land plants (Pickett-Heaps, 1969; Graham, 1985; Graham and Kaneko, 1991). For example, although exceptions exist, microtubules appear to control cellulose orientation in many cell types (Giddings and Staehelin, 1991). The elaboration of a complex cell wall thus entailed the utilization of cortical microtubules Mitotic Plasticity in Plants to align cellulose microfibrils. This in turn necessitated the dispersal of microtubule nucleation capacity away from the cell center to the cortex. In animal cells, interphase microtubules are nucleatedat the centrosome adjacent to the nucleus and permeate the entire cytoplasm (duplication of the centrosome and formation of the MA occur later, prior to division). By contrast, interphase microtubules in plant cells are primarily cortical. Considerable evidence, including the presence of y-tubulin in cortical arrays, supports the idea that those microtubules are nucleated and aligned in the cortex, although this issue has yet to be resolved (e.g., many microtubules are also nucleated at the nuclear envelope in G2 and telophase, where y-tubulin is also located, and these could move to the cortex; see Palevitz, 1991, for review). Centrioles (and basal bodies when in association with cilia and flagella) were probably lost severa1times during alga1and plant evolution (Graham and Kaneko, 1991). Pickett-Heaps (1969) concludedthat centrioles are in effect “disposable” baggage and are not essential for centrosomal activity at all, thereby echoing a proposal of Sharp (1912). Centrioles, in this view, merely use the spindle and centrosome to ensure accurate dissemination of basal bodies in organisms that must generate cilia or flagella. Because walled, sessile land plants cannot locomote, basal bodies and centrioles were unnecessary and were retained only in the divisions immediately preceding the formation of motile sperm in “lower” land plants (Hepler and Myles, 1977; Graham and Kaneko, 1991). In what could be considereda corollary argument to PickettHeaps’, Mazia (1984) has championed the idea that the centrosome is flexible and not necessarily fixed around the centrioles. In their absence, the centrosome is free to assume various morphologies. Thus, the loss of centrioles made the MA poles in plants more diffuse and aided the dispersal of centrosomal material elsewhere in the cell (see also Graham and Kaneko, 1991). The more diffuse poles and attendant multipolarity were additionally advantageous because they potentiated increased MA flexibility, allowing the spindle to operate under spatial constraints imposed by the wall. The broad distribution of y-tubulin over the kinetochore fibers in plant cells compared to its relatively confined location at the centrosomes in most animal cells could be interpreted as support for the dispersal of centrosomal equivalents. Additional support comes from the observation that spindles of early mouse embryos (which lack centrioles) show a dispersed y-tubulin distribution somewhat similar to that seen in plants (Gueth-Hallonet et al., 1993). The hypothesis that plants contain dispersed centrosomal equivalents is attractive. However, an alternative view has been offered by Smirnova and Bajer (1992), who believe that patterns of microtubule nucleation in plants do not require centrosomal homologs but instead rest on the capacity of plant microtubules to undergo self-reorganization.The distribution patterns of y-tubulin in plants (Liu et al., 1993) could be interpreted as support for this idea. All of these ideas await rigorous experimental proof. Indeed, the universalityof the hypothesis that centrioles are not neces- 1005 sary for mitosis was recently challenged on the basis that certain animal cells cannot divide when experimentally deprived of this organelle even though microtubule nucleation capacity is eventually restored (Maniotis and Schliwa, 1991). DEVELOPMENTAL CONSEQUENCES OF MlTOTlC PLASTICITY Chromosome Separation Cells with oblique or deformed MASfind themselves in a rather sticky situation in two important ways. First, if the deformation is extreme, as is the case in Tradescantia generative cells, how are the chromosomes segregated with fidelity? If the kinetochore pairs are distributed along the length of the cell, once separated the kinetochoreswould seem to have to pass each other going in opposite directions during anaphase! How could this occur? Two possibilities come to mind: that the chromosomes separate by a mechanism that does not depend on an ordered bipolar MA or that the MA is not as deformed as it appears, at least by the onset of anaphase. Support for both possibilities can be found in the literature. The first idea is consistent with an old hypothesis that the MA in such cells is not a single entity. Instead, each chromosomal pair organizes its own MA (Saxand OMara, 1941; Lewandowska and Charzynska, 1977); in effect, the dividing Tradescantia generative cell contains 12 different spindles. Although this idea may seem radical, certain data make it less so. Specifically, separate chromosomes do organizetheir own minispindles in insect cells (e.g., Hughes-Schrader, 1924, 1942; Church et al., 1986; Hatsumi and Endow, 1992; note, however, that Rieder and Nowogrodzki, 1983, disagree). In addition, experimental evidence indicates that the seat of chromosome motion is actually at the kinetochore, where mechanochemical transduction by motor proteins such as kinesin and dynein, in combination with microtubule depolymerization, is responsible for the kinetochore“chewing” its way to the poles (Gorbsky et al., 1987; Nicklas, 1989). Thus, in the case of Tradescantia, once kinetochores are pointed in the “right” direction (which is imposed by the elongate spindle shape of the cell and a longitudinalsystem of microtubule bundles), they would naturally and independently arrive at opposite ends of the generative cell regardless of their starting positions. It seems entirely feasible that chromosomes could organize separate spindles and move chromosomes independently of each other. The latter could explain chromosome motion in severely distorted spindles. However, the validity of 12 separate MASin Tradescantia generative cells remainsto be tested. Actually, it should be recognizedthat these kinetochorefibers are interlinked in tandem arrays (Palevitz and Cresti, 1989), which argues against completely independent MASand in favor of some degree of coordinated action. 1006 The Plant Cell On the other hand, at the onset of anaphase, deformation of the MA may be alleviated. At about this time, microtubule rearrangements make the MA more obviously bipolar (Liu and Palevitz, 1992a). Kinetochore fibers belonging to each set of chromosomes become interlinked on two superbundles with opposing structural polarities. Each superbundle then “delivers” its attached kinetochores to the opposite ends of the cell like two groups of fish on strings. Clearly, both microtubule rearrangements and the pac man-like action of the kinetochores may operate in this situation. Division Planes The second dilemma of MA deformation is just as important as the first: how is the division plane achieved if the MA is deformed or aligned incorrectly? Because the phragmoplast is defined by the midzone of the MA in late anaphase-telophase, then if the MA is oblique, the cell plate should be as well. Indeed, this is the case in guard mother cells (Palevitz and Hepler, 1974; Palevitz, 1986). But such a situation is at odds with the fact that the cortex contains “information” that determines final cell wall position and orientation. This information defining the position and plane of the future cell plate is inscribed prior to division. In most higher plant cells, the plane is marked by a group of microtubules called the preprophase band, which appears early in the G2 phase of the cell cycle (Gunning, 1982; Wick, 1991). The preprophase band somehow denotes a specialized “division site” in the cortex or plasma membranethat is active in telophase, long after the band itself disappears late in prophase (Gunning, 1982). In many highly vacuolate cells, the band borders a “raft” of cytoplasm, termed the phragmosome, that forms prior to division and ultimately acts as a scaffold for the nucleus, MA, and phragmoplast(Staiger and Lloyd, 1991).The preprophase band contains actin microfilaments and other proteins in addition to microtubules (e.g., Wick, 1991; Cleary et al., 1992b; Eleftheriou and Palevitz, 1992; Liu and Palevitz, 1992b). Thus, the division site is complex and associated with a variety of structural organization from G2 through cytokinesis. Various hypotheses have been offered to explain the way the division site functions. For example, evidence including microtubule linkages between the preprophase band and the early MA at prophase (e.g., Mineyuki et al., 1991), as well as coordinate effects of the anti-actin drug cytochalasin D on both the width of the band and the placement of the nucleus, MA, and cell plate (Mineyuki and Palevitz, 1990; Eleftheriou and Palevitz, 1992), pointsto the preprophase band somehow positioning the nucleus and MA (Wick, 1991). Nevertheless, this influence is not absolute. As described above, the MA (and thus the midzone and phragmoplast)are often forced into the ”wrong” (e.g., oblique) orientationthrough spatial effects. What happens then? Such “mistakes” are corrected through a second activity of the division site in the cortex: its interactionwith the expanding phragmoplast. This interaction is evident from a variety of obsetvations (e.g., Ôta, 1961; Gunning, 1982; Galatis et al., 1984; Wick, 1991). In onion guard mother cells, the oblique phragmoplastclearly respondsto the division site and snaps into the correct longitudinal alignment when it reaches the cortex (Palevitz, 1986). lnjury to the division site prevents this interaction (Gunning and Wick, 1985). In other words, the preprophase band and division site not only embody basic morphogeneticinformationabout the position and orientation of the new wall but also act to secure compliance with that information should the MA be forced into an incompatible orientation. Out of necessity I have, of course, generalized certain topics and concepts. For example, oblique MASare sometimes corrected in anaphase (e.g., stamen hairs; Bglãr, 1929; Zhang et al., 1990) before the phragmoplastappears. In addition, plants clearly have other means to secure division planes. In lower plants, MA orientation and perhaps even the preprophase band are determined by polar organizers such as those associated with plastids (Busby and Gunning, 1989; Brown and Lemmon, 1990a, 1990b; Cleary et ai., 1992a). Preprophase bands are not present in the divisions that create the male and female gametophytesof angiosperms (e.g., during microsporogenesis and generative cell formation), yet division planes are precise (Brown and Lemmon, 1991; Staiger and Cande, 1992). Nevertheless, the higher plant preprophase band illustrates an important point: MA deformations happen, probably because of the wall. The resulting malplacementof the cell plate could be developmentallycatastrophic, but built-insystems that compensate for the lack of locomotion imposed by the wall also correct for MA deformation. CONCLUDING REMARKS In the preceding discussion I have described an aspect of the dynamic properties of the MA in plants: its ability to respond to its immediate environment, most notably the cell wall. That responsivenessand the machinery used to determine the division plane are products of evolution and are undoubtedly determined genetically. The challenge for the near future is to identify the genes and proteins involved by harnessingthe combined powers of molecular and cell biology. ACKNOWLEDGMENTS I am indebted to severa1 people for important conversations andlor critical reading of the manuscript, includingAndrew Bajer, W.Z. (Zac) Cande, Linda Graham, Peter Hepler, Bo Liu, Rosa McDonald, Bruce Nicklas, and lrena Staxh. I also thank Bo Liu for help with the figures. The work in my laboratory is supported by Grant No. DCB-9019285 from the National Science Foundation. Received May 19, 1993; accepted July 9, 1993. Mitotic Plasticity in Plants 1007 REFERENCES Cleary, A.L., Gunning, B.E.S., Wasteneys, G.O., and Hepler, P.K. (1992b). Microtubuleand F-actin dynamics at the division site in living Tradescantia stamen hair cells. J. Cell Sci. 103, 977-988. Bailey, I.W. (1920). The cambium and its derivatives. 111. A reconnaissance of cytological phenomena in the cambium. Am. J. Bot. 7, 417-434. Eleftheriou, E.P., and Palevitz, B.A. (1992). The effect of cytochalasin D on preprophase band organization in root tip cells of Allium. J. Cell Sci. 103, 989-998. Bajer, A.S. (1990). The elusive organization of the spindle and the kinetochore fiber: A conceptual retrospective.Adv. Cell Biol. 3,65-93. Fosket, D.E., and Morejohn, L.C. (1992). Structural and functional organization of tubulin. Annu. Rev. Plant Physiol. Plant MOI.Biol. 43, 201-240. Balczon, R., and West, K. (1991). The identification of mammalian centrosomal antigens using human autoimmune anticentrosomal antisera. Cell Motil. Cytoskel. 20, 121-135. Baskin, T., and Cande, W.Z. (1990). The structure and function of the mitotic spindle in flowering plants. Annu. Rev. Plant Physiol. Plant MOI. Biol. 41, 277-315. BBIHr, K. (1929). Beitrage zur Kausalanalyse der Mitose. 111. Untersuchungen an den Staubfadenhaarzellen und Blattmeristemzellen von Tradescantia virginica. Zeit. Zellforsch. 10, 73-134. Bloom, G.S. (1992). Motor proteinsfor cytoplasmic microtubules. Curr. Opin. Cell Biol. 4, 66-73. Brown, R.C., and Lemmon, B.E. (199Oa). Monoplastidiccell division in lower land plants. Am. J. Bot. 77, 559-571. Brown, R.C., and Lemmon, B.E. (1990b). Polar organizers mark division axis prior to preprophaseband formation in mitosis in the hepatic Reboulia hemisphaerica (Bryophyta). Protoplasma 156, 74-81. Brown, R.C., and Lemmon, B.E. (1991). The cytokinetic apparatus in meiosis: Control of division plane in the absence of a preprophase band of microtubules. In The Cytoskeletal Basis of Plant Growth and Form, C.W. Lloyd, ed (London: Academic Press), pp. 259-273. Brown, R.C., and Lemmon, B.E. (1992). Pollen development in orchids. 4. Cytoskeletonand ultrastructure of the unequal pollen mitosis in Phalaenopsis. Protoplasma 167, 183-192. Busby, C.H., and Gunning, B.E.S. (1989). Development of the quadripolar meiotic apparatus in Funaria spore mother cells: Analysis by means of antimicrotubule drug treatments. J. Cell Sci. 93,267-277. Cai, G., Bartalesi, A., De1Casino, C., Moscatelli, A., Tieui, A., and Cresti, M. (1993).The kinesin immunoreactivehomologuefrom Nicotiana tabacum L. pollen tubes: Biochemical properties and subcellular localization. Planta, in press. Cande, W.Z. (1990). Centrosomes: Compositionand reprcduction. Curr. Opin. Cell Biol. 2, 301-305. Chevrier, V., Komesli, S., Schmit, A.-C., Vantard, M., Lambert, A.-M., and Job, D. (1992). A monoclonal antibody, raised against mammalian centrosomes and screened by recognition of plant microtubule organizing centers, identifies a pericentriolar component in different cell types. J. Cell Sci. 101, 823-835. Cho, S.-O., and Wick, S.M. (1989). Microtubule orientation during stomatal differentiation in grasses. J. Cell Sci. 92, 581-594. Church, K., Nicklas, R.B., and Lin, H.-P. (1986). Micromanipulated bivalents can trigger mini-spindle formation in Drosophila melanogaster spermatocyte cytoplasm. J. Cell Biol. 103, 2765-2773. Fowke, L.C, Attree, S.M., Wang, H., and Dunstan, D.I. (1990). Microtubule organization and cell division in embryogenic pfutoplast cultures of white spruce. Protoplasma 158, 86-94. Galatis, B., Apostolakos, P., and Katsaros, C. (1984). Experimental studies on the function of the cortical cytoplasmic zone of the preprophase microtubule band. Protoplasma 122, 11-26. Giddings, T.H., and Staehelin, L.A. (1991). Microtubule-mediatedcontrol of microfibril deposition: A re-examination of the hypothesis. In The Cytoskeletal Basis of Plant Growth and Form, C.W. Lloyd, ed (London: Academic Press), pp. 85-99. Gorbsky, G.J., Sammak, P.J., and Borisy, G.G. (1987). Chromosomes move poleward in anaphase along stationary microtubulesthat coordinately disassemble from their kinetochore ends. J. Cell Biol. 104, 9-18. Graham, L.E. (1985). The origin of the life cycle of land plants. Am. Sci. 73, 178-186. Graham, L.E., and Kaneko, Y. (1991). Subcellular structures of relevance to the origin of land plants (embryophytes) from green algae. CRC Crit. Rev. Plant Sci. 10, 323-342. Gueth-Hallonet, C, Antony, C, Aghion, J., Santa-Maria, A., LajoieMizenc, I., Wright, M., and Maro, 8. (1993). y-Tubulin is present in acentriolar MTOCs during early mouse development. J. Cell Sci. 105, 157-166. Gunning, B.E.S. (1982). The cytokinetic apparatus: Its development and spatial regulation. In The Cytoskeleton in Plant Growth and Development, C.W. Lloyd, ed (London: Academic Press), pp. 229-292. Gunning, B.E.S., and Wick, S.M. (1985). Preprophase bands, phragmoplasts, and spatial control of cytokinesis. J. Cell Sci. 2 (suppl.), 157-179. Hatsumi, M., and Endow, S.A. (1992). Mutants of the microtubule motor protein, nonclaret disjunctional, affect spindle structure and chromosome movement in meiosis and mitosis. J. Cell Sci. 101, 545-559. Hepler, P.K. (1992). Calcium and mitosis. Int. Rev. Cytol. 138,239-268. Hepler, P.K., and Myles, D.G. (1977). Spermatcgenesisin Marsilea: An example of male gamete development in plants. In lnternational Cell Biology 1976-1977, B.R. Brinkley and K.R. hrter, eds (New York: Rockefeller University Press), pp. 569-579. Hogan, C.J., Stephens, L., Shimizu, T., and Cande, W.Z. (1992). Physiologicalevidence for involvement of a kinesin-relatedprotein during anaphase spindle elongation in diatom central spindles. J. Cell Biol. 119, 1277-1286. Cleary, A.L., and Hardham, A.R. (1989). Microtubule organization during development of stomatal complexes of Lolium rigidum. Protoplasma 149, 67-81. Hughes-Schrader, S. (1924). Reproduction in Acroschismus wheeleri Pierce. J. Morphol. 39, 157-205. Cleary, A.L., Brown, R.C, and Lemmon, B.E. (1992a). Establishment of division plane and mitosis in monoplastidic guard mother cells of Selaginella. Cell Motil. Cytoskel. 23, 89-101. Hughes-Schrader, S. (1942). The chromosomes of Nautococcus schraderae Vays, and the meiotic division figure of male llaveiine coccids. J. Morphol. 70, 261-299. 1008 The Plant Cell Inou(~, S. (1964). Organization and function of the mitotic spindle. In Primitive Motile Systems in Cell Biology, R.D. Allen and N. Kamiya, eds (New York: Academic Press), pp. 549-598. Joahi, H.C., Palacioa, M.J., McNamara, L., and Cleveland, D.W. (1992). y-Tubulin is a centrosomal protein required for cell cycledependent microtubule nucleation. Nature 356, 80-83. Kaplan, D.R. (1992). The relationship of cells to organisms in plants: Problem and implications of an organismal perspective. Int. J. Plant £ci. 153, £28-£37. Lambert, A.-M. (1993). Microtubule-organizing centers in higher plants. Curt. Opin. Cell Biol. 5, 116-122. Leslie, R.J., Kohler, E., and Wilson, L. (1991). A component of the interphase cytoskeleton is cyclically recruited into spindle poles during mitosis. Cell Motil. Cytoskel. 19, 80-90. Lewandowska, E., and Charzynska, M. (1977). Tradescantia bracteata pollen in vitro: Pollen tube development and mitosis. Acta Soc. Bot. Polon. 46, 587-597. Liu, B., and Palevitz, B.A. (1991). Kinetochore fiber formation in dividing generative cells of Tradescantia. Kinetochore reorientation associated with the transition between lateral microtubule interactions and endon kinetochore fibers. J. Cell Sci. 98, 475-482. Liu, B., and Palevitz, B.A. (1992a). Anaphase chromosome separation in dividing generative cells of Tradescantia. Changes in microtubule organization and kinetochore distribution visualized by antitubulin and CREST immunocytochemistry. Protoplasma 166, 122-133. Liu, B., and Palevitz, B.A. (1992b). Organization of cortical microfilaments in dividing root cells. Cell Motil. Cytoskel. 23, 252-264. Liu, B., Marc, J., Joshi, H.C., and Palevitz, B.A. (1993). A y-tubulinrelated protein associated with the microtubule arrays of higher plants in a cell cycle-dependent manner. J. Cell Sci. 104, 1217-1228. Maekawa, T., Leslie, R., and Kuriyama, R. (1991). Identification of a minus end-specific microtubule-associated protein located at the mitotic poles in cultured mammalian cells. Eur. J. Cell Biol. 54, 255-267. Maniotia, A., and Schliwa, M. (1991). Microsurgical removal of centrosomes blocks cell reproduction and centriole generation in BSC-1 cells. Cell 67, 495-504. Mazia, D. (1984). Centrosomes and the mitotic poles. Exp. Cell Res. 153, 1-15. McDonald, A.R., Llu, B., Joshi, H., and Palevitz, B.A. (1993). ~-Tubulin is associated with a cortical microtubute organizing zone in the developing guard cells of Allium cepa L. Planta, in press. Mineyuki, Y., and Palevitz, B.A. (1990). Relationship between preprophase band organization, F-actin and the division site in AIlium. J. Cell Sci. 97, 283-295. Mineyukl, Y., Marc, J., and Palevltz, B.A. (1988). Formation of the oblique spindle in dividing guard mother cells of Allium. Protoplasma 147, 200-203. Mineyuki, Y., Marc, J., and Palevitz, B.A. (1991). Relationship between the preprophase band, nucleus and spindle in dividing Allium cotyledon cells. J. Plant Physiol. 138, 640-649. N~mec, B. (1899). Ueber die karyokinetische kerntheilung in der wurzelspitze von Allium cepa. Jahrb. Wiss. Bot. 33, 313-336. Nlcklae, R.B. (1989). The motor for poleward chromosome movement in anaphase is in or near the kinetochore. J. Cell Biol. 109, 2245--2255. Oakley, B.R. (1992). 7-Tubulin:The microtubule organizer? Trends Cell Biol. 2, 1-5. Ota, T. (1961).The role of cytoplasm in cytokinesis in plant cells. Cytologia 26, 428-447. Oud, J.L., and Nanninga, N. (1992). Cell shape, chromosome orientation and the position of the plane of division in Vicia faba root cortex cells. J. Cell Sci. 103, 847-855. Palevitz, B.A. (1986). Division plane determination in guard mother cells of A/lium: Video time-lapse analysis of nuclear movements and phragmoplast rotation in the cortex. Dev. Biol. 117, 644-654. Palevitz, B.A. (1988). Microtubular fir-trees in mitotic spindles of onion roots. Protoplasma 142, 74-78. Palevitz, B.A. (1991). Potential significance of microtubule rearrangement, translocation and reutilization in plant cells. In The Cytoskeletal Basis of Plant Growth and Form, C.W. Lloyd, ed (London: Academic Press), pp. 45-55. Palevitz, B.A. (1993). Organization of the mitotic apparatus during generative cell division in Nicotiana tabacum. Protoplasma 174, 25-35. Palevitz, B.A., and Hepler, P.K. (1974). The control of the plane of division during stomatal differentiation in Allium. I. Spindle reorientation. Chromosoma 46, 297-326. Palevitz, B.A., and Cresti, M. (1989). Cytoskeletal changes during generative cell division and sperm formation in Tradescantia virginiana. Protoplasma 150, 54-71. Pelevitz, B.A., and Tlezzi, A. (1992). Organization, composition, and function of the generative cell and sperm cytoskeleton. Int. Rev. Cytol. 140, 149-185. Pickett-Heaps, J.D. (1969). The evolution of the mitotic apparatus: An attempt at comparative ultrastructural cytology in dividing plant cells. Cytobios 3, 257-280. Rieder, C.L., and Nowogrodzki, R. (1983). Intranuclear membranes and the formation of the first meiotic spindle in Zenos peckii (Acroschismus wheelerii) oocytes. J. Cell Biol. 97, 1144-1155. Rout, M.R, and Kilmartin, J.V. (1990). Components of the yeast spindle and spindle pole body. J. Cell Biol. 111, 1913-1927. Saundem, W.S., and Hoyt, M.A. (1992). Kinesin-related proteins required for structural integrity of the mitotic spindle. Cell 70, 451-458. Sawln, K.E., LeGuellec, K., Philllpe, M., and Mltchlson, T.J. (1992). Mitotic spindle organization by a plus-end-directed microtubule motor. Nature 359, 540-543. Sex, K., end O'Mera, J.G. (1941). Mechanism of mitosis in pollen tubes. Bot. Gaz. 102, 629-636. Schlbler, M.J., and Pickett-Heaps, J.D. (1987). The kinetochore fiber structure in the acentric spindles of the green alga Oedogonium. Protoplasma 137, 29-44. Schmlt, A.-C., Vantard, A.-M., de Mey, J., and Lambert, A.-M. (1983). Aster-like microtubule centers establish spindle polarity during interphase-mitosis transition in higher plant cells. Plant Cell Rep. 2, 285-288. Sharp, L.W. (1912). Spermatogenesis in Equisetum. Bot. Gaz. 54, 89-119. Sinnott, E.W. (1960). Plant Morphogenesis. (New York: McGraw-Hill). Smirnova, E.A., and BaJer, A.S. (1992). Spindle poles in higher plant mitosis. Cell Motil. Cytoskel. 23, 1-7. Mitotic Plasticity in Plants Staiger, C.J., and Cande, W.Z. (1990). Microtubule distribution in dv, a maize meiotic mutant defective in the prophase to metaphase transition. Dev. Biol. 138, 231-242. Staiger, C.J., and Cande, W.Z. (1992). Arneiotic, a gene that controls meiotic chromosome and cytoskeletal behavior in maize. Dev. Biol. 154, 226-230. Staiger, C.J., and Lloyd, C.W. (1991). The plant cytoskeleton. Curr. Opin. Cell Biol. 3, 33-42. Strome, S. (1993). Determination of division planes. Cell 72, 3-6. Traas, J., Beven, A.F., Doonan, J.H., Cordewener, J., and Shaw, RJ, (1992). Cell-cycle-dependent changes in labelling of specific 1009 phosphoproteins by the monoclonal antibody MPM-2 in plant cells. Plant J. 2, 723-732. Vandr6, D.D., Davis, F.M., Rao, RN., and Borisy, G.G. (1984). Phosphoproteins are components of mitotic microtubule organizing centers. Proc. Natl. Acad. Sci. USA 61, 4439-4443. Wada, B. (1965). Analysis of mitosis. Cytologia 30 (suppl.), 92-149. Wick, S.M. (1991). The preprophase band. In The Cytoskeletal Basis of Plant Growth and Form, C.W. Lloyd, ed (London: Academic Press), pp. 231-244. Zhang, D., Wadsworth, R, and Hepler, RK. (1990). Microtubule dynamics in living dividing plant cells: Confocal imaging of microinjected fluorescent brain tubulin. Proc. Natl. Acad. Sci. USA 87, 8820-8824. Morphological Plasticity of the Mitotic Apparatus in Plants and Its Developmental Consequences. B. A. Palevitz Plant Cell 1993;5;1001-1009 DOI 10.1105/tpc.5.9.1001 This information is current as of June 14, 2017 Permissions https://www.copyright.com/ccc/openurl.do?sid=pd_hw1532298X&issn=1532298X&WT.mc_id=pd_hw1532298X eTOCs Sign up for eTOCs at: http://www.plantcell.org/cgi/alerts/ctmain CiteTrack Alerts Sign up for CiteTrack Alerts at: http://www.plantcell.org/cgi/alerts/ctmain Subscription Information Subscription Information for The Plant Cell and Plant Physiology is available at: http://www.aspb.org/publications/subscriptions.cfm © American Society of Plant Biologists ADVANCING THE SCIENCE OF PLANT BIOLOGY