* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Corrosion - ThaparNotes
Survey
Document related concepts
Shape-memory alloy wikipedia , lookup
Energy applications of nanotechnology wikipedia , lookup
Work hardening wikipedia , lookup
Fatigue (material) wikipedia , lookup
Materials Research Science and Engineering Centers wikipedia , lookup
Flux (metallurgy) wikipedia , lookup
Condensed matter physics wikipedia , lookup
Semiconductor wikipedia , lookup
History of metamaterials wikipedia , lookup
Strengthening mechanisms of materials wikipedia , lookup
Transcript
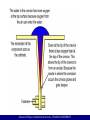
Materials Science & Engineering (UES012) Corrosion is the disintegration of metal through an unintentional chemical or electrochemical action, starting at its surface. Corrosive environment is needed for it All metals exhibit a tendency to be oxidized, some more easily than others. A tabulation of the relative strength of this tendency is called the galvanic series. Knowledge of a metal's location in the series is an important piece of information to have in making decisions about its potential usefulness for structural and other applications. The driving force that causes metals to corrode is a natural consequence of their temporary existence in metallic form. To reach this metallic state from their occurrence in nature in the form of various chemical compounds (ores), it is necessary for them to absorb and store up for later return by corrosion, the energy required to release the metals from their original compounds. School of Physics & Materials Science, THAPAR UNIVERSITY Standard EMF series This table is for reduction reactions. For oxidation reactions, the corresponding voltage will change sign. School of Physics & Materials Science, THAPAR UNIVERSITY Corrosion in metals It is the process by which metallic atoms leave the compounds in the presence of water or gas. Metal atoms leave the metal until it fails or erodes from oxidation. The tendency for oxidation is dependent on the metal. Metals used in construction, such as steel and copper alloys, are both subject to corrosion. Metals are chemical combinations of multiple elements. These metals are highly vulnerable because of the high energy content of the elements used in metallic form Corrosion is a natural process, which converts a refined metal to a more chemically-stable form, such as its oxide, hydroxide, or sulfide due to environmental conditions such as pollution, moisture etc For metallic materials, the corrosion process is normally electrochemical. Electrochemical reaction - a chemical reaction in which there is transfer of electrons from one chemical species to another. Metal atoms characteristically lose or give up electrons in what is called an oxidation reaction. M Mn++ ne- School of Physics & Materials Science, THAPAR UNIVERSITY 3 Reduction Reaction The electrons generated from each metal atom that is oxidized are transferred to and become a part of another chemical species. This is a reduction reaction. Reduction of hydrogen ions in an acid solution Reduction reaction in an acid solution containing dissolved oxygen Reduction reaction in a neutral or basic solution containing dissolved oxygen School of Physics & Materials Science, THAPAR UNIVERSITY Electrode potential (Principle of Corrosion) Not all metallic materials oxidize to form ions with same degree of ease. If the iron and copper electrodes are connected electrically, reduction will occur for copper at the expense of the oxidation of iron. Cu2+ + Fe Cu + Fe2+ In terms of half-cell reactions: Fe Fe2++ 2eCu2+ + 2e- Cu Cu2+ + Fe Cu + Fe2+ Cu2+ ions will deposit (electrodeposit) as metallic copper on the copper electrode, while iron dissolves (corrodes) on the other side of the cell and goes into solution as Fe2+ ions. School of Physics & Materials Science, THAPAR UNIVERSITY Standard half-cell Galvanic cell When externally connected, electrons generated from the oxidation of iron flow to the copper cell in order that Cu2+ be reduced. In addition, there will be some net ion motion from each cell to the other across the membrane. This is called a galvanic couple - two metals electrically connected in a liquid electrolyte wherein one metal becomes an anode and corrodes, while the other acts as a cathode. An electric potential or voltage will exist between the two cell halves. For example: Cu2+ + Fe Cu + Fe2+ Potential: 0.780 V Fe2+ + Zn Fe + Zn2+ Potential: 0.323 V Various electrode pairs have different voltages ! School of Physics & Materials Science, THAPAR UNIVERSITY Standard EMF series Cell voltages represent only differences in electrical potential. establish a reference point, or reference cell, to which other cell halves may be compared. Reference cell = standard hydrogen electrode Electromotive force (emf) series Connect a standard half cell of various metals to the standard hydrogen electrode. Measure the voltage of the cell. Rank the metals as per this voltage. H2 gas 1 atm pressure School of Physics & Materials Science, THAPAR UNIVERSITY Corrosion rate The corrosion rate, or the rate of material removal as a consequence of the chemical action, is an important corrosion parameter. Corrosion penetration rate (CPR), or the thickness loss of material per unit of time. where W = weight loss after exposure time t ; = density, and A = exposed specimen area, K is a constant. There is an electric current associated with electrochemical corrosion reactions. Corrosion rate r can be written as r = i/nF i = current density (the current per unit surface area of material corroding) n = number of electrons associated with the ionization of each metal atom F = 96,500 C/mol. School of Physics & Materials Science, THAPAR UNIVERSITY Forms of corrosion Uniform Attack - Occurs with equivalent intensity over the entire exposed surface - Often leaves behind a scale or deposit. Examples: General rusting of steel and iron, Tarnishing of silverware Galvanic Corrosion - occurs when two metals or alloys having different compositions are electrically coupled while exposed to an electrolyte. - The less noble or more reactive metal in the particular environment will experience corrosion; the more inert metal, the cathode, will be protected from corrosion. Lower in the series experiences corrosion Example: Steel screws corrode when in contact with brass in a marine environment; If copper and steel tubing are joined in a domestic water heater, the steel will corrode in the vicinity of the junction. School of Physics & Materials Science, THAPAR UNIVERSITY Forms of corrosion Crevice Corrosion (a narrow opening especially in a rock or wall/ crack) In small opening where solution is not in flow and is stagnent - Arises as a consequence of concentration differences of ions or dissolved gases in the electrolyte solution, and between two regions of the same metal piece. - For such a concentration cell, corrosion occurs in the locale that has the lower concentration. Example: occurs in crevices and recesses or under deposits of dirt or corrosion products where the solution becomes stagnant and there is localized depletion of dissolved oxygen. The crevice must be wide enough for the solution to penetrate, yet narrow enough for stagnancy galvanic corrosion two connected metals + single environment crevice corrosion one metal part + two connected environments This form of attack is generally associated with the presence of small volumes of stagnant solution in occluded interstices, beneath deposits and seals, or in crevices, e.g. at nuts and rivet heads. It is a localized form of attack, where there is a breakdown of the surface passive layer, in crevices or on 'shielded' areas beneath surface deposits. Engineered or 'designed in' crevices can be set up at bolted and other joints, beneath flanges or between flanges and gaskets or other contact areas such as valve seats. Crevices make a chemical environment which is different from that of freely exposed surfaces and therefore accelerate corrosion. This environment keeps moisture, traps pollutants, concentrates corrosion products. . School of Physics & Materials Science, THAPAR UNIVERSITY School of Physics & Materials Science, THAPAR UNIVERSITY Forms of corrosion Stress Corrosion - results from the combined action of an applied tensile stress and a corrosive environment; humidity, pollution both influences are necessary. - In fact, some materials that are virtually inert in a particular corrosive medium become susceptible to this form of corrosion when a stress is applied. - Small cracks form and then propagate in a direction perpendicular to the stress, with the result that failure may eventually occur. - Failure behavior is characteristic of that for a brittle material, even though the metal alloy is intrinsically ductile. - Furthermore, cracks may form at relatively low stress levels, significantly below the tensile strength. - Most alloys are susceptible to stress corrosion in specific environments, especially at moderate stress levels. Example: Most stainless steels stress corrode in solutions containing chloride ions, A bent wire will corrode easily at the bend. School of Physics & Materials Science, THAPAR UNIVERSITY Hydrogen embrittlement The embrittlement of a metal or alloy by atomic hydrogen involves the ingress of hydrogen into a component, an event that can seriously reduce the ductility and load-bearing capacity, cause cracking and catastrophic brittle failures at stresses below the yield stress of susceptible materials. Hydrogen embrittlement occurs in a number of forms but the common features are an applied tensile stress and hydrogen dissolved in the metal. Examples of hydrogen embrittlement are cracking of weldments or hardened steels when exposed to conditions which inject hydrogen into the component. Presently this phenomenon is not completely understood and hydrogen embrittlement detection, in particular, seems to be one of the most difficult aspects of the problem. Hydrogen embrittlement does not affect all metallic materials equally. The most vulnerable are high-strength steels, titanium alloys and aluminum alloys. Sources of hydrogen causing embrittlement have been encountered in the making of steel, in processing parts, in welding, in storage or containment of hydrogen gas, and related to hydrogen as a contaminant in the environment that is often a by-product of general corrosion. It is the latter that concerns the nuclear industry. Hydrogen may be produced by corrosion reactions such as rusting, cathodic protection, and electroplating. Hydrogen may also be added to reactor coolant to remove oxygen from reactor coolant systems. Hydrogen entry, the obvious pre-requisite of embrittlement, can be facilitated in a number of ways summarized below: by some manufacturing operations such as welding, electroplating, phosphating and pickling; if a material subject to such operations is susceptible to hydrogen embrittlement then a final, baking heat treatment to expel any hydrogen is employed as a by-product of a corrosion reaction such as in circumstances when the hydrogen production reaction described here acts as the cathodic reaction since some of the hydrogen produced may enter the metal in atomic form rather than be all evolved as a gas into the surrounding environment. In this situation, cracking failures can often be thought of as a type of stress corrosion cracking. If the presence of hydrogen sulfide causes entry of hydrogen into the component, the cracking phenomenon is often termed “sulphide stress cracking (SSC)” the use of cathodic protection for corrosion protection if the process is not properly controlle School of Physics & Materials Science, THAPAR UNIVERSITY Erosion corrosion is a degradation of material surface due to mechanical action, often by impinging liquid, abrasion by a slurry, particles suspended in fast flowing liquid or gas, bubbles or droplets, cavitation, etc. The mechanism can be described as follows: mechanical erosion of the material, or protective (or passive) oxide layer on its surface, enhanced corrosion of the material, if the corrosion rate of the material depends on the thickness of the oxide layer. School of Physics & Materials Science, THAPAR UNIVERSITY Protection against corrosion • Use of noble metals – least susceptible to corrosion (from emf series) • Avoid physical contact between dissimilar metals – No galvanic cell formation • For dissimilar metal contacts, anode should have a larger surface area than cathode. Corrosion rate is dependent on current density. Ex: Cu nut & bolt on large steel plate • Materials with two phase structure – formation of galvanic couple, i.e., one phase acts as anode while another one acts as cathode. Ex: cementite in steel is more noble (cathode) than ferrite. Protection by passivation layers Ex: Aluminum coating on both sides of duralumin sheets • Use of inhibitors – formation of a protective coating • Use of non-metallic coating such as enamel, oil, paint, etc. School of Physics & Materials Science, THAPAR UNIVERSITY So far… Classification of materials Crystal Structure Chemical Bonding Structure of Solids: Crystalline and non-crystalline materials, Inorganic solids, Silicate structures and its applications. Crystal Imperfections: Point defects, Line defects, Surface defects, Movement of Dislocation, Dislocation energy. Diffusion: Laws of diffusion, Temperature dependence of diffusion coefficient, Determination of activation energy. Mechanical Properties: Elastic, Anelastic and Viscoelastic behaviour, Plastic behaviour, Critical shear stress, Twinning and slipping phenomenon, Creep. Equilibrium Diagram: Solids solutions and alloys, Gibbs phase rule, Isomorphous and eutectic phase diagrams and their construction, Lever- arm rule, Application of phase diagrams, Zone refining. Corrosion Process School of Physics & Materials Science, THAPAR UNIVERSITY Properties of Materials One goal of materials engineering is to select materials with suitable properties for a given application. Mechanical properties A. Elasticity and stiffness (recoverable stress vs. strain) B. Plasticity (non-recoverable stress vs. strain) C. Strength D. Brittleness or Toughness E. Fatigue Electrical properties A. Electrical conductivity and resistivity B. Semiconducting materials School of Physics & Materials Science, THAPAR UNIVERSITY 17 Properties of Materials Dielectric properties A. Polarizability B. Capacitance C. Ferroelectric properties D. Piezoelectric properties E. Pyroelectric properties Magnetic properties A. Paramagnetic properties B. Diamagnetic properties C. Ferromagnetic properties Optical properties A. Refractive index B. Absorption, reflection, and transmission C. Birefringence (double refraction) Corrosion properties Biological properties A. Toxicity B. bio-compatibility School of Physics & Materials Science, THAPAR UNIVERSITY 18