* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download L 35 Modern Physics [1]
Renormalization wikipedia , lookup
Franck–Condon principle wikipedia , lookup
Particle in a box wikipedia , lookup
James Franck wikipedia , lookup
Bremsstrahlung wikipedia , lookup
Atomic orbital wikipedia , lookup
Hydrogen atom wikipedia , lookup
Tight binding wikipedia , lookup
Double-slit experiment wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Wheeler's delayed choice experiment wikipedia , lookup
Matter wave wikipedia , lookup
Astronomical spectroscopy wikipedia , lookup
Ultraviolet–visible spectroscopy wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Electron configuration wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Delayed choice quantum eraser wikipedia , lookup
Ultrafast laser spectroscopy wikipedia , lookup
Atomic theory wikipedia , lookup
Population inversion wikipedia , lookup
Wave–particle duality wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
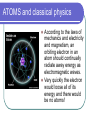
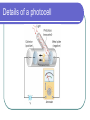
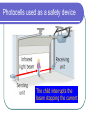
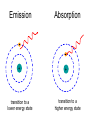
L 35 Modern Physics [1] Introduction- quantum physics Particles of light PHOTONS The photoelectric effect Photocells & intrusion detection devices The Bohr atom emission & absorption of radiation LASERS Scanners CD readers Modern Physics- Introduction “Modern” – 20th Century By the end of the 19th century it seemed that all the laws of physics were known However, there were a few problems where classical physics (pre-20th century) didn’t seem to work. It became obvious that Newton’s laws could not explain atomic level phenomena ATOMS and classical physics According to the laws of mechanics and electricity and magnetism, an orbiting electron in an atom should continually radiate away energy as electromagnetic waves. Very quickly the electron would loose all of its energy and there would be no atoms! Newton’s Laws have flaws! Newton’s laws, which were so successful in allowing us to understand the behavior of big objects such as the motions of the planets, failed when pushed to explain atomic size phenomena. The discovery of the laws of atomic physics led to every important 20th century discovery that have transformed our lives, the electronic revolution. The photoelectric effect- photons LIGHT photoelectrons Metal plate When light shines on a metal surface, electrons pop out Photoelectrons are only emitted if the wavelength of the light is shorter than some maximum value, no matter how intense the light is. Details of a photocell Photocells used as a safety device The child interrupts the beam stopping the current Photoelectric effect defies a classical explanation According to classical physics, if the intensity of the light is strong enough, enough energy should be absorbed by the electrons to make them pop out The wavelength of the light should not make a difference. What is going on ? ? ? Einstein explains the PE effect, receives Nobel Prize in 1921 A radical idea was needed to explain the photoelectric effect. Light is an electromagnetic wave, but when it interacts with matter (the metal surface) it behaves like a particle, a light particle called a photon. A beam of light is thought of as a beam of photons. PE – explanation continued The energy of a photon depends on the wavelength or frequency of the light Recall that speed of light = wavelength () x frequency (f) Photon energy E = Planck’s constant x frequency = h f Shorter wavelength (higher f) photons have a higher energy PE – explanation continued A certain amount of energy is required to make an electron pop out of a metal An photoelectron is emitted if it absorbs a photon from the light beam that has enough energy (high enough frequency) No matter how many photons hit the electron, if they don’t have the right frequency the electron doesn’t get out Blue and red photons -example How much energy does a photon of wavelength = 350 nm (nanometers) have compared to a photon of wavelength = 700 nm? Solution: The shorter wavelength photon has the higher frequency. The 350 nm photon has twice the frequency as the 700 nm photon. Therefore, the 350 nm photon has twice the energy as the 700 nm photon. The quantum concept The photon concept is a radial departure from classical thinking. In classical physics, energy can come in any amounts In modern physics, energy is QUANTIZED comes in definite packets photons of energy h f. In the PE effect energy is absorbed by the electrons only in discreet amounts Video recorders and digital cameras A CCD (charge coupled device) can be used to capture photographic Images using the photoelectric effect. http://money.howstuffworks.com/camcorder2.htm THE LASER: a product of 20th Century Physics Light Amplification by Stimulated Emission of Radiation. Niels Bohr and his five sons The quantum concept and the Bohr Atom Niels Bohr, a Danish physicist, used the quantum concept to explain the nature of the atom. Recall that the orbiting electrons, according to classical ideas, should very quickly radiate away all of its energy If this were so, then we would observe that atoms emit light over a continuous range of wavelengths (colors) NOT SO! Line spectra of atoms Line spectra are like atomic fingerprints. Forensic scientists use line spectra to identify substances. The Bohr Atom Nucleus + Ei Ef The orbits farther from the nucleus are higher energy states than the closer ones • The electrons move in certain allowed, “stationary” orbits or states in which then do not radiate. • The electron in a high energy state can make a transition to a lower energy state by emitting a photon whose energy was the difference in energies of the two states, hf = Ei - Ef Line spectra of atomic hydrogen The Bohr model was successful in determining Where all the spectral lines of H should be. Emission and Absorption When an electron jumps from a high energy state to a low energy state it emits a photon emission spectrum An electron in a low energy state can absorb a photon and move up to a high energy state absorption spectrum Emission + transition to a lower energy state Absorption + transition to a higher energy state




















![L 35 Modern Physics [1] Modern Physics](http://s1.studyres.com/store/data/003926344_1-b779c05b753c6dc3972377c21f9bdcd3-150x150.png)


![L 35 Modern Physics [1]](http://s1.studyres.com/store/data/000572764_1-c4bf5ed66474525e3cf4981a43e1bbe1-150x150.png)

![L 35 Modern Physics [1] Modern Physics](http://s1.studyres.com/store/data/001558975_1-84d6e03bc786b63795533f59711ce2f4-150x150.png)





![L 33 Modern Physics [1] Modern Physics](http://s1.studyres.com/store/data/003217156_1-265c5a519e2bca3f33717b4abd842898-150x150.png)







