Cyanide Destruction with Chlorine Dioxide
... makes chlorine dioxide a powerful nonchlorinating oxidizing agent useful in many water treating applications for which chlorine and other oxidizing agents are unsuitable. Unlike most oxidants, chlorine dioxide may be used over a broad pH range. Application Description Cyanides (CN-) are used to solu ...
... makes chlorine dioxide a powerful nonchlorinating oxidizing agent useful in many water treating applications for which chlorine and other oxidizing agents are unsuitable. Unlike most oxidants, chlorine dioxide may be used over a broad pH range. Application Description Cyanides (CN-) are used to solu ...
File
... 5.2 Oxidation Numbers Oxidation number the apparent charge an atom would have if it gained or lost its bonding electrons Consider the example of sulfur dioxide, SO2. In sulfur dioxide, oxygen is more electronegative than sulfur. Since oxygen gains two electrons to form the oxide ion, O-2, in ionic c ...
... 5.2 Oxidation Numbers Oxidation number the apparent charge an atom would have if it gained or lost its bonding electrons Consider the example of sulfur dioxide, SO2. In sulfur dioxide, oxygen is more electronegative than sulfur. Since oxygen gains two electrons to form the oxide ion, O-2, in ionic c ...
Sources of CO in your home - Livingston Public Schools
... are the particles that generally pass through the throat and nose and enter the lungs. Once inhaled, these particles can affect the heart and lungs and cause serious health effects. EPA groups particle pollution into two categories: – "Inhalable coarse particles," such as those found near roadways a ...
... are the particles that generally pass through the throat and nose and enter the lungs. Once inhaled, these particles can affect the heart and lungs and cause serious health effects. EPA groups particle pollution into two categories: – "Inhalable coarse particles," such as those found near roadways a ...
Unit F335/01
... (c) At 500 K, the equilibrium constant for equation 1.1 is 7.76 × 10–3. In an equilibrium mixture at 500 K, the concentrations of hydrogen and carbon dioxide are: [H2] = 1.00 × 10–5 mol dm–3 [CO2] = 3.46 × 10–5 mol dm–3 Calculate the equilibrium concentrations of H2O and CO at 500 K. Assume the H2O ...
... (c) At 500 K, the equilibrium constant for equation 1.1 is 7.76 × 10–3. In an equilibrium mixture at 500 K, the concentrations of hydrogen and carbon dioxide are: [H2] = 1.00 × 10–5 mol dm–3 [CO2] = 3.46 × 10–5 mol dm–3 Calculate the equilibrium concentrations of H2O and CO at 500 K. Assume the H2O ...
T -I O -D
... and carbon dioxide is produced, making an appropriate atmosphere for anaerobic photosynthesizers. The media and chamber work as follows: the presence of sodium sulfide and cysteine (an amino acid with an exposed sulfhydryl group) helps maintain reducing conditions; it is also a key component for ano ...
... and carbon dioxide is produced, making an appropriate atmosphere for anaerobic photosynthesizers. The media and chamber work as follows: the presence of sodium sulfide and cysteine (an amino acid with an exposed sulfhydryl group) helps maintain reducing conditions; it is also a key component for ano ...
Acid Precipitation
... The ancient organisms also contained proteins, however, and these proteins contained sulfur and nitrogen. The sulfur, therefore, remains as a contaminant in fossil fuels. When the fossil fuels are burned, the sulfur is heated to very high temperatures and reacts with oxygen in the air. Write a combu ...
... The ancient organisms also contained proteins, however, and these proteins contained sulfur and nitrogen. The sulfur, therefore, remains as a contaminant in fossil fuels. When the fossil fuels are burned, the sulfur is heated to very high temperatures and reacts with oxygen in the air. Write a combu ...
Elemental Sulfur Corrosion in Sour Gas and Claus Sulfur Recovery
... Primary Corrosion in Sulfur Tanks – Air Drafted Systems ...
... Primary Corrosion in Sulfur Tanks – Air Drafted Systems ...
File - chemistryattweed
... Most sulfur dioxide (SO2) released into the atmosphere comes from the burning of such coal or oil in electric power stations. SO2 is the major contributor to acid rain that can affect places thousands of kilometres from the source. The low sulfur content of Australian coal is one reason why Australi ...
... Most sulfur dioxide (SO2) released into the atmosphere comes from the burning of such coal or oil in electric power stations. SO2 is the major contributor to acid rain that can affect places thousands of kilometres from the source. The low sulfur content of Australian coal is one reason why Australi ...
Proton n.m.r, spectroscopic evidence for sulfur
... bChemiC'JI shift difference between the aromatic signals. ...
... bChemiC'JI shift difference between the aromatic signals. ...
Nitrogen and Sulfur
... Atmospheric sulfur • Air pollutant from burning coal and oil. • Pollution can supply ½ of plants sulfur requirements. • 2-8 lb/A sulfur added to soil annually from rain in Oklahoma… more in E • Irrigation water can also be high in sulfate • Irrigation water containing 1 ppm sulfate adds 2.7 lbs S pe ...
... Atmospheric sulfur • Air pollutant from burning coal and oil. • Pollution can supply ½ of plants sulfur requirements. • 2-8 lb/A sulfur added to soil annually from rain in Oklahoma… more in E • Irrigation water can also be high in sulfate • Irrigation water containing 1 ppm sulfate adds 2.7 lbs S pe ...
The Role of Organosulfur Compounds in the Growth of PbS
... 1-Decanethiol Titration of PbS QDs. We performed a control experiment where we added 100, 500, 1000, and 5000 molar equivalents of 1-decanethiol per PbS QD as estimated from NIR band-edge absorption spectroscopy. These were stirred for 18 hours under N2 and then the 1HNMR spectrum acquired in CDCl3 ...
... 1-Decanethiol Titration of PbS QDs. We performed a control experiment where we added 100, 500, 1000, and 5000 molar equivalents of 1-decanethiol per PbS QD as estimated from NIR band-edge absorption spectroscopy. These were stirred for 18 hours under N2 and then the 1HNMR spectrum acquired in CDCl3 ...
Rana ridibunda
... higher in cells actively performing some anabolic activity as protein synthesis, lipogenesis, or glycogenesis (ELLMAN & GAN 1969) which may be reflected in a higher content of rough endoplasmic reticulum. Sulfhydryl levels therefore represent a measure of the activity potential of cells for maintena ...
... higher in cells actively performing some anabolic activity as protein synthesis, lipogenesis, or glycogenesis (ELLMAN & GAN 1969) which may be reflected in a higher content of rough endoplasmic reticulum. Sulfhydryl levels therefore represent a measure of the activity potential of cells for maintena ...
Bacteria on the edge: redox and pH • Anaerobic bacterial
... • Created naturally and anthropogenically – natural sources • production of sulfurous gases in hydrothermal vents and some hot springs • metabolic activities of certain acidophiles themselves. ...
... • Created naturally and anthropogenically – natural sources • production of sulfurous gases in hydrothermal vents and some hot springs • metabolic activities of certain acidophiles themselves. ...
Helen`s Project4
... parvum, gen. nov. and sp. nov., a new purple non-sulfur bacterium with unusual light absorption properties. Arch Microbiol (1999) vol. 171 pp. 405-416 Selective enrichment with light instead of media. Reinhartz et al. Sulfide oxidation in the phototrophic sulfur bacterium Chromatium vinosum. Arch Mi ...
... parvum, gen. nov. and sp. nov., a new purple non-sulfur bacterium with unusual light absorption properties. Arch Microbiol (1999) vol. 171 pp. 405-416 Selective enrichment with light instead of media. Reinhartz et al. Sulfide oxidation in the phototrophic sulfur bacterium Chromatium vinosum. Arch Mi ...
Recognizing Wine Off-Characters of Microbial Origin
... • Character interacts with other characters to give a more complex off-character: fecal, burnt rubber • Wine aroma is attenuated at levels below recognition threshold ...
... • Character interacts with other characters to give a more complex off-character: fecal, burnt rubber • Wine aroma is attenuated at levels below recognition threshold ...
Soil and Applied Sulfur (A2525)
... Authors: E.E. Schulte and K.A. Kelling are professors of soil science, College of Agricultural and Life Sciences, University of WisconsinMadison and University of Wisconsin-Extension, Cooperative Extension. The authors wish to thank L.M. Walsh, professor of soil science, University of Wisconsin-Madi ...
... Authors: E.E. Schulte and K.A. Kelling are professors of soil science, College of Agricultural and Life Sciences, University of WisconsinMadison and University of Wisconsin-Extension, Cooperative Extension. The authors wish to thank L.M. Walsh, professor of soil science, University of Wisconsin-Madi ...
Lecture #7: Composition of the Present Day Atmosphere
... subsequently rains out of the atmosphere). Carbon dioxide (CO2), carbon monoxide (CO), and methane (CH4): Carbon dioxide is important because it is the 2nd most abundant greenhouse gas in the atmosphere (after water vapor). Carbon dioxide is not toxic, so it does not directly impact human health. Ho ...
... subsequently rains out of the atmosphere). Carbon dioxide (CO2), carbon monoxide (CO), and methane (CH4): Carbon dioxide is important because it is the 2nd most abundant greenhouse gas in the atmosphere (after water vapor). Carbon dioxide is not toxic, so it does not directly impact human health. Ho ...
Elements, Compounds and Mixtures.
... letter = a new chemical symbol/element) • NaCl = sodium chloride (table salt) • H2O2 = hydrogen peroxide • C6H12O6 = glucose (sugar plants make from photosynthesis) ...
... letter = a new chemical symbol/element) • NaCl = sodium chloride (table salt) • H2O2 = hydrogen peroxide • C6H12O6 = glucose (sugar plants make from photosynthesis) ...
Air Pollution Causes and Effects
... of carbon monoxides (CO) and a major source of hydrocarbons (HC) and nitrogen oxides (NOx). Whereas, fuel combustion in stationary sources is the dominant source of sulfur dioxide (SO2). Carbon Dioxide ...
... of carbon monoxides (CO) and a major source of hydrocarbons (HC) and nitrogen oxides (NOx). Whereas, fuel combustion in stationary sources is the dominant source of sulfur dioxide (SO2). Carbon Dioxide ...
Inorganic Chemistry
... With metal cations, phosphate forms a variety of salts. These solids are polymeric, featuring P-O-M linkages. When the metal cation has a charge of 2+ or 3+, the salts are generally insoluble, hence they exist as common minerals. Many phosphate salts are derived from hydrogen phosphate (HPO42-). PCl ...
... With metal cations, phosphate forms a variety of salts. These solids are polymeric, featuring P-O-M linkages. When the metal cation has a charge of 2+ or 3+, the salts are generally insoluble, hence they exist as common minerals. Many phosphate salts are derived from hydrogen phosphate (HPO42-). PCl ...
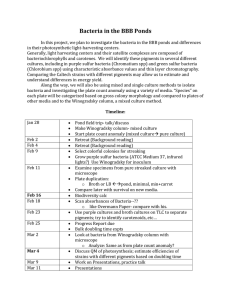
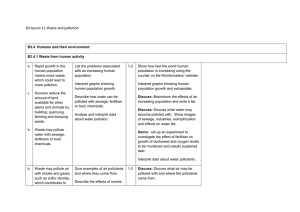
B3 lesson 11 Waste and pollution B3.4 Humans and their
... How might these affect life in rivers ...
... How might these affect life in rivers ...
Biochemistry of Sulfur
... Thiosulfate Oxidation: A tetrathionate-forming, membrane-bound thiosulfate:quinone oxidoreductase (TQO) was isolated from aerobically grown Ac. ambivalens cells. Optimal activity was observed at 85 ˚C and pH 5. The 102 kDa glycosylated holoenzyme had a α2β2 stoichiometry. Oxygen electrode measureme ...
... Thiosulfate Oxidation: A tetrathionate-forming, membrane-bound thiosulfate:quinone oxidoreductase (TQO) was isolated from aerobically grown Ac. ambivalens cells. Optimal activity was observed at 85 ˚C and pH 5. The 102 kDa glycosylated holoenzyme had a α2β2 stoichiometry. Oxygen electrode measureme ...
Sulfur dioxide
Sulfur dioxide (also sulphur dioxide) is the chemical compound with the formula SO2. At standard atmosphere, it is a toxic gas with a pungent, irritating, and rotten smell. The triple point is 197.69 K and 1.67 kPa. It is released naturally by volcanic activity. Sulfur dioxide was used by the Romans in winemaking when they discovered that burning sulfur candles inside empty wine vessels kept them fresh and free from vinegar smell.