File - Ms. Fox-Lent
... isotope is always the same; it doesn't depend on how many atoms you have or on how ...
... isotope is always the same; it doesn't depend on how many atoms you have or on how ...
Name:
... nucleus. When presented with problems you need to be able to tell me how many proton neutrons and electron are in an element or isotope based on limited information, such as: If I give you the hyphen notation or nuclear symbol of an uranium isotope you should be able to tell me how many protons, neu ...
... nucleus. When presented with problems you need to be able to tell me how many proton neutrons and electron are in an element or isotope based on limited information, such as: If I give you the hyphen notation or nuclear symbol of an uranium isotope you should be able to tell me how many protons, neu ...
CHAPTER 5 The Structure of Atoms
... Determined the charge and the mass of the electron from the oil drop experiment. (The second American to win Nobel prize in physics in 1923) 1910 – Ernest Rutherford Gave the first basically correct picture of the atom’s structure. (Nobel prize in chemistry in 1908) ...
... Determined the charge and the mass of the electron from the oil drop experiment. (The second American to win Nobel prize in physics in 1923) 1910 – Ernest Rutherford Gave the first basically correct picture of the atom’s structure. (Nobel prize in chemistry in 1908) ...
Chapter One
... Determined the charge and the mass of the electron from the oil drop experiment. (The second American to win Nobel prize in physics in 1923) 1910 – Ernest Rutherford Gave the first basically correct picture of the atom’s structure. (Nobel prize in chemistry in 1908) ...
... Determined the charge and the mass of the electron from the oil drop experiment. (The second American to win Nobel prize in physics in 1923) 1910 – Ernest Rutherford Gave the first basically correct picture of the atom’s structure. (Nobel prize in chemistry in 1908) ...
doc - The Crowned Anarchist Literature
... The chemical evidence that was so vital in leading Hahn and Strassmann to the discovery of nuclear fission was obtained by the application of carrier and tracer techniques. Since invisible amounts of the radioactive species were formed, their chemical identity had to be deduced from the manner in w ...
... The chemical evidence that was so vital in leading Hahn and Strassmann to the discovery of nuclear fission was obtained by the application of carrier and tracer techniques. Since invisible amounts of the radioactive species were formed, their chemical identity had to be deduced from the manner in w ...
PHY221 Lab-03-1: Rutherford Scattering
... 79mp = 79(1.67 × 10−27 kg) = 1.32 × 10−25 kg 2mp = 2(1.67 × 10−27 kg) = 3.34 × 10−27 kg +79(1.6 × 10−19 C) = 1.264 × 10e−17 C +2(1.6 × 10−19 C) = 3.2 × 10e−19 C h0, 0, 0i m h−2.7 × 10−12 , 0, 0i m h2 × 107 , 0, 0i m/s 9 × 109 Nm2 /C2 ...
... 79mp = 79(1.67 × 10−27 kg) = 1.32 × 10−25 kg 2mp = 2(1.67 × 10−27 kg) = 3.34 × 10−27 kg +79(1.6 × 10−19 C) = 1.264 × 10e−17 C +2(1.6 × 10−19 C) = 3.2 × 10e−19 C h0, 0, 0i m h−2.7 × 10−12 , 0, 0i m h2 × 107 , 0, 0i m/s 9 × 109 Nm2 /C2 ...
2 - The Student Room
... Beta radiation from a certain source can be stopped completely by a sheet of aluminium 3.0 mm thick. Calculate the mass of a square sheet of aluminium of this thickness measuring 1.0m x 1.0m. (Density of aluminium = 2.7 x 103 kgm-3 ) (2) To a fair approximation, the ability of any sheet of material ...
... Beta radiation from a certain source can be stopped completely by a sheet of aluminium 3.0 mm thick. Calculate the mass of a square sheet of aluminium of this thickness measuring 1.0m x 1.0m. (Density of aluminium = 2.7 x 103 kgm-3 ) (2) To a fair approximation, the ability of any sheet of material ...
142KB - NZQA
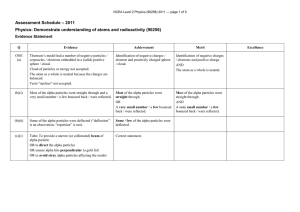
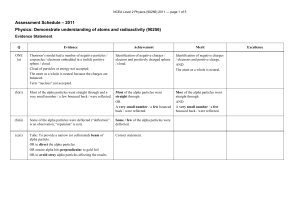
... • Flemings left hand rule (or other suitable hand rule) predicts this Alpha has a small / smaller deflection because • Alpha has much greater mass (8000 compared with beta) so it affected less by the force produced by the magnetic field. • Alpha has a much smaller charge / mass ratio (x 1 / 4000 c ...
... • Flemings left hand rule (or other suitable hand rule) predicts this Alpha has a small / smaller deflection because • Alpha has much greater mass (8000 compared with beta) so it affected less by the force produced by the magnetic field. • Alpha has a much smaller charge / mass ratio (x 1 / 4000 c ...
NCEA Level 2 Physics (90256) 2011 Assessment Schedule
... • Flemings left hand rule (or other suitable hand rule) predicts this Alpha has a small / smaller deflection because • Alpha has much greater mass (8000 × compared with beta) so it affected less by the force produced by the magnetic field. • Alpha has a much smaller charge / mass ratio (x 1 / 4000 c ...
... • Flemings left hand rule (or other suitable hand rule) predicts this Alpha has a small / smaller deflection because • Alpha has much greater mass (8000 × compared with beta) so it affected less by the force produced by the magnetic field. • Alpha has a much smaller charge / mass ratio (x 1 / 4000 c ...
PP - myndrs.com
... • Particle emitted: No particle, but gives off excess energy • Result: No change to type of nucleus, it just has less energy. • Ba → Ba + Gamma radiation • Ba → Ba + ...
... • Particle emitted: No particle, but gives off excess energy • Result: No change to type of nucleus, it just has less energy. • Ba → Ba + Gamma radiation • Ba → Ba + ...
Atomic Structure
... If the substance produced is radioactive it can also decay and the process can continue until a stable isotope is reached. For example uranium 238 decays through many steps to lead 206. The graph of amount of substance versus time is not a straight line for a radioactive substance but is an exponent ...
... If the substance produced is radioactive it can also decay and the process can continue until a stable isotope is reached. For example uranium 238 decays through many steps to lead 206. The graph of amount of substance versus time is not a straight line for a radioactive substance but is an exponent ...
Nuclear Chemistry – Chapter 25, chapter 4, section 4
... (100000) HIGH: need to be protected by thick concrete or metal like lead ...
... (100000) HIGH: need to be protected by thick concrete or metal like lead ...
In 1896, Henri Becquerel was working with compounds containing
... most beta particles An example of such a process is: ...
... most beta particles An example of such a process is: ...
11.6 Nuclear Radiation
... either the atomic mass or atomic number. It does however increase the stability of the nucleus, as it reduces its energy level. The gamma rays are weakly ionising, as they tend to pass through a substance rather than collide with its atoms. When gamma rays do collide with an atom, energy is transfer ...
... either the atomic mass or atomic number. It does however increase the stability of the nucleus, as it reduces its energy level. The gamma rays are weakly ionising, as they tend to pass through a substance rather than collide with its atoms. When gamma rays do collide with an atom, energy is transfer ...
Atomic Structure
... particles together. This force is only effective at very small distances. Protons and neutrons are composed of still smaller particles called quarks. Electrons are not composed of quarks and cannot be subdivided. ...
... particles together. This force is only effective at very small distances. Protons and neutrons are composed of still smaller particles called quarks. Electrons are not composed of quarks and cannot be subdivided. ...
radioactivity PowerPoint Presentation
... • Nucleus changes mass by four units and charge by two units – Alpha particle easily stopped – 4 x nucleon mass ...
... • Nucleus changes mass by four units and charge by two units – Alpha particle easily stopped – 4 x nucleon mass ...
File - Carroll`s Cave of Knowledge
... isotopes are types of the same element (same # of protons), with different atomic mass numbers (due to the # of neutrons present). Carbon 12 has 6 protons + 6 neutrons Carbon 14 has 6 protons + 8 neutrons a nucleus is radioactive when it is unstable and can spontaneously emit a particle and become t ...
... isotopes are types of the same element (same # of protons), with different atomic mass numbers (due to the # of neutrons present). Carbon 12 has 6 protons + 6 neutrons Carbon 14 has 6 protons + 8 neutrons a nucleus is radioactive when it is unstable and can spontaneously emit a particle and become t ...
Nuclear Change
... Produced when a proton “captures” an electron from the orbital nearest the nucleus and changes into a neutron Decreases the atomic number by one This process causes an “excited nucleus” A gamma ray is emitted by the nucleus to become more stable ...
... Produced when a proton “captures” an electron from the orbital nearest the nucleus and changes into a neutron Decreases the atomic number by one This process causes an “excited nucleus” A gamma ray is emitted by the nucleus to become more stable ...
Document
... Write simple nuclear reactions and apply the law of conservation of mass Calculate half-life. Calculate mass remaining of an element after half-lifes have occurred. Use dimensional analysis to solve problems involving the relationships between grams, moles, particles, and liters. ...
... Write simple nuclear reactions and apply the law of conservation of mass Calculate half-life. Calculate mass remaining of an element after half-lifes have occurred. Use dimensional analysis to solve problems involving the relationships between grams, moles, particles, and liters. ...
Radioactive Decay
... • I can figure out how old a substance is using the half-life • I can interpret a graph on half-life ...
... • I can figure out how old a substance is using the half-life • I can interpret a graph on half-life ...
The buoyant force on an object totally submerged in a fluid depends
... And the Messiah cometh in the fulness of time, that he may redeem the children of men from the fall. And because that they are redeemed from the fall they have become free forever, knowing good from evil; to act for themselves and not to be acted upon, save it be by the punishment of the law at the ...
... And the Messiah cometh in the fulness of time, that he may redeem the children of men from the fall. And because that they are redeemed from the fall they have become free forever, knowing good from evil; to act for themselves and not to be acted upon, save it be by the punishment of the law at the ...
Chem Ch. 4.4
... electrons called beta particles. They have a 1- charge. – Symbol for a beta particle is 00 1ß or -1e. – Example of beta decay ...
... electrons called beta particles. They have a 1- charge. – Symbol for a beta particle is 00 1ß or -1e. – Example of beta decay ...
HW Notes: Nuclear Chemistry - Liberty Union High School District
... The ability of radioactive particles to pass through air and other materials is inversely related to their mass. • Alpha particles – the least penetrating, they travel only a few centimeters through air. They can be stopped by a single sheet of paper. • Beta particles – more penetrating, they travel ...
... The ability of radioactive particles to pass through air and other materials is inversely related to their mass. • Alpha particles – the least penetrating, they travel only a few centimeters through air. They can be stopped by a single sheet of paper. • Beta particles – more penetrating, they travel ...
Calutron

A calutron is a mass spectrometer originally designed and used for separating the isotopes of uranium. It was developed by Ernest O. Lawrence during the Manhattan Project and was based on his earlier invention, the cyclotron. Its name was derived from California University Cyclotron, in tribute to Lawrence's institution, the University of California in Berkeley, California, where it was invented. Calutrons were used in the industrial-scale Y-12 uranium enrichment plant at the Clinton Engineer Works in Oak Ridge, Tennessee. The uranium-235 produced there was used in the Little Boy atomic bomb that was detonated over Hiroshima on 6 August 1945.The calutron is a type of sector mass spectrometer, an instrument in which a sample is ionized and then accelerated by electric fields and subsequently deflected by magnetic fields. The ions ultimately collide with a plate and produce a measurable electric current. Since the ions of the different isotopes have the same electric charge but different masses, the heavier isotopes are bent less by the magnetic field, causing the beam of particles to separate out into several beams by mass, striking the plate at different locations. The mass of the ions can be calculated according to the strength of the field and the charge of the ions. During World War II, calutrons were developed to use this principle to obtain substantial quantities of high-purity uranium-235, by taking advantage of the small mass difference between uranium isotopes.Electromagnetic separation for uranium enrichment was abandoned in the post-war period in favor of the more complicated, but more efficient, gaseous diffusion method. Although most of the calutrons of the Manhattan Project were dismantled at the end of the war, some remained in use to produce isotopically enriched samples of naturally-occurring elements for military, scientific and medical purposes.