Geologic Time
... How many protons and neutrons does the nucleus of carbon-12 contain? Nucleus of carbon 12 Atomic number = ______ Number of Protons= ______ Number of Neutrons=______ How many protons and neutrons does the nucleus of carbon-14 contain? Number of Protons= ______ Number of Neutrons=______ One isotope of ...
... How many protons and neutrons does the nucleus of carbon-12 contain? Nucleus of carbon 12 Atomic number = ______ Number of Protons= ______ Number of Neutrons=______ How many protons and neutrons does the nucleus of carbon-14 contain? Number of Protons= ______ Number of Neutrons=______ One isotope of ...
Numerical Ages 1 -
... This system works if 1) you are not interested in very small blocks of time, and 2) you are only interested in fairly short blocks, or, alternately, 3) you are careful to light a new candle at the exact time the old one burns out, for as long as is necessary to measure a big block of time. This wor ...
... This system works if 1) you are not interested in very small blocks of time, and 2) you are only interested in fairly short blocks, or, alternately, 3) you are careful to light a new candle at the exact time the old one burns out, for as long as is necessary to measure a big block of time. This wor ...
SSPD Chapter 1_Part 5_Story of Atom-Solar
... Atoms are electrically neutral hence nucleus contains as many positive charges as are the electrons orbiting the nucleus. From this Model, most of the Atom is vacuum. Just as the Planets are orbiting the Sun, in an analogous manner electrons are orbiting the nucleus. Furthermore the nucleus contains ...
... Atoms are electrically neutral hence nucleus contains as many positive charges as are the electrons orbiting the nucleus. From this Model, most of the Atom is vacuum. Just as the Planets are orbiting the Sun, in an analogous manner electrons are orbiting the nucleus. Furthermore the nucleus contains ...
The Atomic Nucleus and Radioactivity Review questions pg. 657
... Ans. Gamma rays are photons of light and therefore do not have a charge. A magnetic field will only defect objects that have a charge and are moving or are magnetic. ...
... Ans. Gamma rays are photons of light and therefore do not have a charge. A magnetic field will only defect objects that have a charge and are moving or are magnetic. ...
Chapter 33: The Atomic Nucleus and Radioactivity
... Ans. A) Beta emission occurs when a neutron emits an electron as it changes into a proton. When 218 84 Po emits a beta particle, its atomic number increases by one and its atomic mass remains unchanged. The resulting atom will be atomic number 85 and its atomic mass is 218. The reaction can be writt ...
... Ans. A) Beta emission occurs when a neutron emits an electron as it changes into a proton. When 218 84 Po emits a beta particle, its atomic number increases by one and its atomic mass remains unchanged. The resulting atom will be atomic number 85 and its atomic mass is 218. The reaction can be writt ...
Chapter 33: The Atomic Nucleus and Radioactivity
... Ans. Beta emission occurs when a neutron emits an electron. The neutron changes into a proton in the process. The atomic nucleus now has one more proton that before the emission and thus is now an atom of a different element. ...
... Ans. Beta emission occurs when a neutron emits an electron. The neutron changes into a proton in the process. The atomic nucleus now has one more proton that before the emission and thus is now an atom of a different element. ...
Dominoes - Learning on the Loop
... The process of nuclei breaking up and emitting particles or waves to become stable ...
... The process of nuclei breaking up and emitting particles or waves to become stable ...
Nuclear Chemistry
... The half-life of a radioisotope (a radioactive isotope) is the time necessary for one-half of the atoms/nuclei to decay. The rate of decay is independent of environmental conditions such as pressure and temperature. Although the half-life remains the same, the number of nuclei decreases as a functio ...
... The half-life of a radioisotope (a radioactive isotope) is the time necessary for one-half of the atoms/nuclei to decay. The rate of decay is independent of environmental conditions such as pressure and temperature. Although the half-life remains the same, the number of nuclei decreases as a functio ...
Radiometric Dating
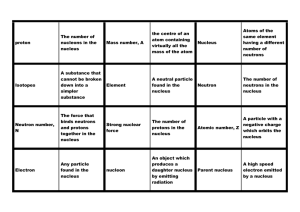
... protons that identifies the element; but may have varying numbers of neutrons • Atomic Number: sum of the protons in the nucleus of the atom • Atomic Mass: sum of the protons and neutrons in the nucleus of the isotope • Example: 39Ar is the isotope of the element argon that has an atomic mass of ...
... protons that identifies the element; but may have varying numbers of neutrons • Atomic Number: sum of the protons in the nucleus of the atom • Atomic Mass: sum of the protons and neutrons in the nucleus of the isotope • Example: 39Ar is the isotope of the element argon that has an atomic mass of ...
Nuclear Chemistry
... Electrochemistry refers to any reaction where any of the elements gain or lose electrons. Of course, before we can determine whether this is happening, we need to know how to figure out whether something has gained or lose electrons. To do this, we need to determine the oxidation states of the eleme ...
... Electrochemistry refers to any reaction where any of the elements gain or lose electrons. Of course, before we can determine whether this is happening, we need to know how to figure out whether something has gained or lose electrons. To do this, we need to determine the oxidation states of the eleme ...
Nuclear Chemistry and Electrochemistry
... Electrochemistry refers to any reaction where any of the elements gain or lose electrons. Of course, before we can determine whether this is happening, we need to know how to figure out whether something has gained or lose electrons. To do this, we need to determine the oxidation states of the eleme ...
... Electrochemistry refers to any reaction where any of the elements gain or lose electrons. Of course, before we can determine whether this is happening, we need to know how to figure out whether something has gained or lose electrons. To do this, we need to determine the oxidation states of the eleme ...
To balance a nuclear equation, the mass number and
... is used to describe nuclear reactions. This style is of the form A(b,c)D, which is equivalent to A + b gives c + D. Common light particles are often abbreviated in this shorthand, typically p for proton, n forneutron, d for deuteron, α representing an alpha particle or helium-4, β for beta particle ...
... is used to describe nuclear reactions. This style is of the form A(b,c)D, which is equivalent to A + b gives c + D. Common light particles are often abbreviated in this shorthand, typically p for proton, n forneutron, d for deuteron, α representing an alpha particle or helium-4, β for beta particle ...
STEM Fair Introduction Beanium Isotopes Lab
... Neutrons are made of one “up” quark and two “down” quarks ...
... Neutrons are made of one “up” quark and two “down” quarks ...
STEM Fair Introduction Beanium Isotopes Lab
... Neutrons are made of one “up” quark and two “down” quarks ...
... Neutrons are made of one “up” quark and two “down” quarks ...
1. The gravitational force is the fundamental force that exists
... A) Because according to the laws of probability, if there are only two possible outcomes for a random event, then either outcome is equally likely. You have a 50% chance of flipping heads, but you can’t predict in advance which flip will fall heads. 27. What does this have to do with radioactive dec ...
... A) Because according to the laws of probability, if there are only two possible outcomes for a random event, then either outcome is equally likely. You have a 50% chance of flipping heads, but you can’t predict in advance which flip will fall heads. 27. What does this have to do with radioactive dec ...
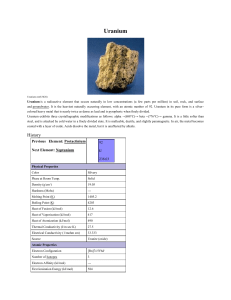
Uranium
... This nuclear conversion can be brought about in breeder reactors where it is possible to produce more new fissionable material than the fissionable material used in maintaining the chain reaction. Uranium-235 (235U) is of even greater importance because it is the key to utilizing uranium. 235U, whil ...
... This nuclear conversion can be brought about in breeder reactors where it is possible to produce more new fissionable material than the fissionable material used in maintaining the chain reaction. Uranium-235 (235U) is of even greater importance because it is the key to utilizing uranium. 235U, whil ...
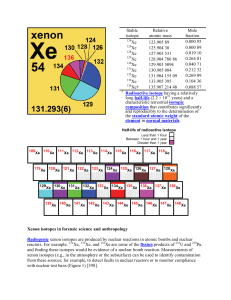
Xenon isotopes in forensic science and anthropology Radiogenic
... than those of X-rays; thus, gamma rays have greater penetrating power. half-life (radioactive) – the time interval that it takes for the total number of atoms of any radioactive isotope to decay and leave only one-half of the original number of atoms. [return] isotope – one of two or more species of ...
... than those of X-rays; thus, gamma rays have greater penetrating power. half-life (radioactive) – the time interval that it takes for the total number of atoms of any radioactive isotope to decay and leave only one-half of the original number of atoms. [return] isotope – one of two or more species of ...
PPT - hrsbstaff.ednet.ns.ca
... positron from within the nucleus There are two types of beta decay (β- and β+) Beta particles can penetrate matter to a greater extent than alpha particles; they can penetrate about 0.1mm of lead or 10m of air They are also a form of ionizing radiation but less damaging than alpha particles ...
... positron from within the nucleus There are two types of beta decay (β- and β+) Beta particles can penetrate matter to a greater extent than alpha particles; they can penetrate about 0.1mm of lead or 10m of air They are also a form of ionizing radiation but less damaging than alpha particles ...
Topic 7: Atomic and nuclear physics 7.1 The atom
... • A Geiger–Müller tube consists of a tube filled with a low-pressure (~0.1 Atm) inert gas such as helium, neon or argon and an organic vapor or a halogen gas and contains electrodes, between which there is a potential difference of several hundred volts, but no current flowing. The walls of the tube ...
... • A Geiger–Müller tube consists of a tube filled with a low-pressure (~0.1 Atm) inert gas such as helium, neon or argon and an organic vapor or a halogen gas and contains electrodes, between which there is a potential difference of several hundred volts, but no current flowing. The walls of the tube ...
Chemistry Vocabulary List
... charge, nearly all of the mass, but a very small fraction of the volume of an atom. 2. Proton => Positively charged particle located in the nucleus of an atom; charge = +1, mass = 1 atomic mass unit 3. Neutron => Neutrally charged particle located in the nucleus of an atom; charge = 0, mass = 1 atom ...
... charge, nearly all of the mass, but a very small fraction of the volume of an atom. 2. Proton => Positively charged particle located in the nucleus of an atom; charge = +1, mass = 1 atomic mass unit 3. Neutron => Neutrally charged particle located in the nucleus of an atom; charge = 0, mass = 1 atom ...
Radioactivity Mid-Unit Review Questions
... of protons, it goes down and up by 2, meaning it’s the same at the end as it was at the beginning. Also, since there are 2 neutrons lost in the release of the alpha particle, the atomic mass goes down by 2. The atomic mass in our calculations would not be changed during beta decay, since each neutro ...
... of protons, it goes down and up by 2, meaning it’s the same at the end as it was at the beginning. Also, since there are 2 neutrons lost in the release of the alpha particle, the atomic mass goes down by 2. The atomic mass in our calculations would not be changed during beta decay, since each neutro ...
Lecture 14 - Experimental Achievements in Physics
... - Henri Becquerel (1852-1908) knew of Rontgen’s experiments, and decided to investigate phosphorescent materials (materials that would glow) - He took a photographic plate and wrapped it in heavy black paper, blocking exposure to sunlight - He then took a thin crust of potassium uranyl sulphate crys ...
... - Henri Becquerel (1852-1908) knew of Rontgen’s experiments, and decided to investigate phosphorescent materials (materials that would glow) - He took a photographic plate and wrapped it in heavy black paper, blocking exposure to sunlight - He then took a thin crust of potassium uranyl sulphate crys ...
Radiometric Dating - EHS
... neutrons making them unstable. The nuclei of radioactive atoms change or decay by giving off radiation in the form of particles or electromagnetic waves until the atom reaches a stable state. ...
... neutrons making them unstable. The nuclei of radioactive atoms change or decay by giving off radiation in the form of particles or electromagnetic waves until the atom reaches a stable state. ...
URANIUM: A RADIOACTIVE CLOCK
... properties of daughter products are so very different, any geological transformation of a rock sample will have quite different effects on the sample’s daughter isotope contents. Potassium and rubidium frequently occur together in rock samples, making this pair particularly important for radiochemic ...
... properties of daughter products are so very different, any geological transformation of a rock sample will have quite different effects on the sample’s daughter isotope contents. Potassium and rubidium frequently occur together in rock samples, making this pair particularly important for radiochemic ...
Uranium: A Radioactive Clock
... seconds for Po-214 to 2.5 × 105 years for U-234. Two other such radioactive series occur in nature. They start with U-235 and Th-232. The uranium radioactive series has been used to estimate the age of the oldest rocks in the Earth's crust. The ratio of U-238 to Pb-206 in a rock changes slowly as th ...
... seconds for Po-214 to 2.5 × 105 years for U-234. Two other such radioactive series occur in nature. They start with U-235 and Th-232. The uranium radioactive series has been used to estimate the age of the oldest rocks in the Earth's crust. The ratio of U-238 to Pb-206 in a rock changes slowly as th ...
Calutron

A calutron is a mass spectrometer originally designed and used for separating the isotopes of uranium. It was developed by Ernest O. Lawrence during the Manhattan Project and was based on his earlier invention, the cyclotron. Its name was derived from California University Cyclotron, in tribute to Lawrence's institution, the University of California in Berkeley, California, where it was invented. Calutrons were used in the industrial-scale Y-12 uranium enrichment plant at the Clinton Engineer Works in Oak Ridge, Tennessee. The uranium-235 produced there was used in the Little Boy atomic bomb that was detonated over Hiroshima on 6 August 1945.The calutron is a type of sector mass spectrometer, an instrument in which a sample is ionized and then accelerated by electric fields and subsequently deflected by magnetic fields. The ions ultimately collide with a plate and produce a measurable electric current. Since the ions of the different isotopes have the same electric charge but different masses, the heavier isotopes are bent less by the magnetic field, causing the beam of particles to separate out into several beams by mass, striking the plate at different locations. The mass of the ions can be calculated according to the strength of the field and the charge of the ions. During World War II, calutrons were developed to use this principle to obtain substantial quantities of high-purity uranium-235, by taking advantage of the small mass difference between uranium isotopes.Electromagnetic separation for uranium enrichment was abandoned in the post-war period in favor of the more complicated, but more efficient, gaseous diffusion method. Although most of the calutrons of the Manhattan Project were dismantled at the end of the war, some remained in use to produce isotopically enriched samples of naturally-occurring elements for military, scientific and medical purposes.