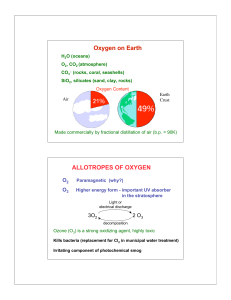
Name - TeacherWeb
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
Lecture 11 - Biosynthesis of Amino Acids
... Amino acid biosynthesis pathways Regulation of amino acid biosynthesis. Amino acids as precursors to other biological molecules. e.g., Nucleotides and porphoryns ...
... Amino acid biosynthesis pathways Regulation of amino acid biosynthesis. Amino acids as precursors to other biological molecules. e.g., Nucleotides and porphoryns ...
p-Block Elements, Part 1
... e.g. Li2O = 2Li+ O2− Peroxide Ion ⇒ O22− = −O – O− e.g. Na2O2 = 2 Na+ −O – O − Also, H2O2 (hydrogen peroxide) Superoxide Ion ⇒ O2− e.g. KO2 = K+ O2− Can have positive oxidation states in combination with fluorine + 2 in OF2 ...
... e.g. Li2O = 2Li+ O2− Peroxide Ion ⇒ O22− = −O – O− e.g. Na2O2 = 2 Na+ −O – O − Also, H2O2 (hydrogen peroxide) Superoxide Ion ⇒ O2− e.g. KO2 = K+ O2− Can have positive oxidation states in combination with fluorine + 2 in OF2 ...
Practice Toxins Mid-Unit Test 08-09
... (A) A mole equals 6.02x1023 atoms (B) A mole of substance has a specific mass. (C) Two moles equals 1.204x1024 atoms (D) A mole of any substance has the same mass. ______10. How many moles of potassium iodide, KI, are there in 100 grams? (A) 0.6 moles (B) 1660 moles (C) 3.01 x 1025 moles (D) 5.0 x 1 ...
... (A) A mole equals 6.02x1023 atoms (B) A mole of substance has a specific mass. (C) Two moles equals 1.204x1024 atoms (D) A mole of any substance has the same mass. ______10. How many moles of potassium iodide, KI, are there in 100 grams? (A) 0.6 moles (B) 1660 moles (C) 3.01 x 1025 moles (D) 5.0 x 1 ...
Ionic Bonding
... 4. Which of the Lewis structures in questions 2 and 3 include coordinate covalent bonds? 5. Distinguish between bonding electrons and lone pairs. 6. Are the following pairs of atoms more likely to form ionic or covalent bonds? (a) sulfur and oxygen (b) iodine and iodine (c) calcium and chlorine (d) ...
... 4. Which of the Lewis structures in questions 2 and 3 include coordinate covalent bonds? 5. Distinguish between bonding electrons and lone pairs. 6. Are the following pairs of atoms more likely to form ionic or covalent bonds? (a) sulfur and oxygen (b) iodine and iodine (c) calcium and chlorine (d) ...
Chapter 17 – Amino Acid Metabolism
... Chapter 17 – Amino Acid Metabolism By Dr. Mohammad College of Science for Women / Babylon University Will be interested in two things: 1) origin of nitrogen atoms and their incorporation into amino group 2) origin of carbon skeletons ...
... Chapter 17 – Amino Acid Metabolism By Dr. Mohammad College of Science for Women / Babylon University Will be interested in two things: 1) origin of nitrogen atoms and their incorporation into amino group 2) origin of carbon skeletons ...
C454_lect11 - chem.uwec.edu - University of Wisconsin
... Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire ...
... Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire ...
pblock - Chemistry Courses
... 2nd period: Only s and p orbitals are possible with n = 2 Therefore, the maximum number of bonds is 4 (single and/or double bonds) Examples: CH4, NF4+, BH43rd (and higher periods): can use d-orbitals to make bonds E.g. ...
... 2nd period: Only s and p orbitals are possible with n = 2 Therefore, the maximum number of bonds is 4 (single and/or double bonds) Examples: CH4, NF4+, BH43rd (and higher periods): can use d-orbitals to make bonds E.g. ...
26.4 The ureo cyde
... deamination also has produced ammonia, however. And ammonia, even in low concentrations, is especially toxic to brain cells and can result in coma and death. A Closer Look Hyperammonemia examines some reasons for this toxicity. It is important that ammonia be removed from the body-This is accomplish ...
... deamination also has produced ammonia, however. And ammonia, even in low concentrations, is especially toxic to brain cells and can result in coma and death. A Closer Look Hyperammonemia examines some reasons for this toxicity. It is important that ammonia be removed from the body-This is accomplish ...
A1979HN47900001
... The work showed that both light and substrate were involved in induction of the enzyme and that the measured activity was related to the accumulation of protein by the plant. The concurrent, and subsequent development of physiological and biochemical techniques for studying the characteristics of is ...
... The work showed that both light and substrate were involved in induction of the enzyme and that the measured activity was related to the accumulation of protein by the plant. The concurrent, and subsequent development of physiological and biochemical techniques for studying the characteristics of is ...
IJCA 40A(6) 652-655
... v(C=N) and ring breathing modes • The appearance of v(C=N) and ring breathing modes due to the ring nitrogen in the complex under study at 1601 and 1086 em·', respectively, (as compared to that of the free ligand at 1590 and 994 em·', respectively) is indicative of the coordination of this ligand th ...
... v(C=N) and ring breathing modes • The appearance of v(C=N) and ring breathing modes due to the ring nitrogen in the complex under study at 1601 and 1086 em·', respectively, (as compared to that of the free ligand at 1590 and 994 em·', respectively) is indicative of the coordination of this ligand th ...
Nutritional Importance of Proteins
... Severe persistent malnutrition may have deleterious effects on the intellectual capacity in later life. ...
... Severe persistent malnutrition may have deleterious effects on the intellectual capacity in later life. ...
Equilibrium Constant- Keq
... 3. Sulfur dioxide gas (0.141 mol/L) and oxygen gas (0.25 mol/L) are produced when sulfur trioxide gas (1.6 mol/L) is decomposed. a) Write a balanced chemical equation b) Write the equilibrium law c) Calculate the equilibrium constant d) Describe the percent reaction. 4. Hydrogen Chloride is produced ...
... 3. Sulfur dioxide gas (0.141 mol/L) and oxygen gas (0.25 mol/L) are produced when sulfur trioxide gas (1.6 mol/L) is decomposed. a) Write a balanced chemical equation b) Write the equilibrium law c) Calculate the equilibrium constant d) Describe the percent reaction. 4. Hydrogen Chloride is produced ...
How do bacteria respond to their environment?
... Experiment • Add uncharged tRNA to ribosomes • See if pppGpp increases Need: • In vitro system where charging of tRNA can be controlled • Assay for level of pppGpp ...
... Experiment • Add uncharged tRNA to ribosomes • See if pppGpp increases Need: • In vitro system where charging of tRNA can be controlled • Assay for level of pppGpp ...
Technical Data Sheet Yeast Extract 19512
... ≤ 5 000 /g ≤ 10 /g Absence / g Absence / 25 g Absence / g ≤ 100 /g The information contained in this publication is based on our own research and development work and is to the best of our knowledge true and accurate. Users should, however, conduct their own tests to determine the suitability of our ...
... ≤ 5 000 /g ≤ 10 /g Absence / g Absence / 25 g Absence / g ≤ 100 /g The information contained in this publication is based on our own research and development work and is to the best of our knowledge true and accurate. Users should, however, conduct their own tests to determine the suitability of our ...
FINAL EXAM REVIEW PROBLEMS
... b. Gaseous nitrogen monoxide decomposes to produce dinitrogen monoxide gas and nitrogen dioxide gas. c. Liquid nitric acid decomposes to reddish brown nitrogen dioxide gas, liquid water, and oxygen gas. 23. Predict whether a solid will form when the following pairs of solution are mixed. If so, iden ...
... b. Gaseous nitrogen monoxide decomposes to produce dinitrogen monoxide gas and nitrogen dioxide gas. c. Liquid nitric acid decomposes to reddish brown nitrogen dioxide gas, liquid water, and oxygen gas. 23. Predict whether a solid will form when the following pairs of solution are mixed. If so, iden ...
Lab 10: Ammonium Nitrogen
... Some trees and grasses are able to absorb ammonium ions directly, but most require their conversion to nitrate. This process, called nitrification, is usually accomplished by bacteria in the soil or water. In the first step of nitrification, ammonium ions are oxidized into nitrite. The nitrite is th ...
... Some trees and grasses are able to absorb ammonium ions directly, but most require their conversion to nitrate. This process, called nitrification, is usually accomplished by bacteria in the soil or water. In the first step of nitrification, ammonium ions are oxidized into nitrite. The nitrite is th ...
Assessment of grapevine nitrogen status and optimized nitrogen
... also important in those situations (e.g., Figure 1) where vineyards are being located on steep slopes in order to minimize the potential for soil erosion. The under-trellis (also called intra-row) cover crops are becoming more widely used in the Virginia industry and are either intentionally planted ...
... also important in those situations (e.g., Figure 1) where vineyards are being located on steep slopes in order to minimize the potential for soil erosion. The under-trellis (also called intra-row) cover crops are becoming more widely used in the Virginia industry and are either intentionally planted ...
Chapter 26 Outline Assimilation of Inorganic Nitrogen
... ATP requirement is explained as energy needed to overcome a high activation energy for breaking the N2 triple bond. Other texts suggest the ATP lowers the reduction potential of the reductase complex. Rhizobia grow in symbiotic association with leguminous plants and fix nitrogen for them. Chemical f ...
... ATP requirement is explained as energy needed to overcome a high activation energy for breaking the N2 triple bond. Other texts suggest the ATP lowers the reduction potential of the reductase complex. Rhizobia grow in symbiotic association with leguminous plants and fix nitrogen for them. Chemical f ...
12.3 - heoldduscience
... nothing can escape), equilibrium is reached when both reactions occur at exactly the same rate in each direction. The relative amounts of all the reacting substances at equilibrium depend on the conditions of the reaction. ...
... nothing can escape), equilibrium is reached when both reactions occur at exactly the same rate in each direction. The relative amounts of all the reacting substances at equilibrium depend on the conditions of the reaction. ...
NO 2
... H2S is: – toxic to bacteria, algae, workers (ppm) – smelly – reactive with metals gives FeS – precursor for acid formation (when oxidised by Thiobacillus) – cause of steel and concrete corrosion ...
... H2S is: – toxic to bacteria, algae, workers (ppm) – smelly – reactive with metals gives FeS – precursor for acid formation (when oxidised by Thiobacillus) – cause of steel and concrete corrosion ...