File - Ms M - EARL MARRIOTT SECONDARY
... Gamma radiation has no charge and no mass, and is represented by Gamma radiation is the highest-energy form of electromagnetic radiation. takes thick blocks of lead or concrete to stop gamma rays. Gamma decay results from energy being released from a high-energy nucleus. ...
... Gamma radiation has no charge and no mass, and is represented by Gamma radiation is the highest-energy form of electromagnetic radiation. takes thick blocks of lead or concrete to stop gamma rays. Gamma decay results from energy being released from a high-energy nucleus. ...
Chapter 25 – Types of Radiation 1. Alpha Radiation Alpha decay
... Positron decay is the mirror image of beta decay and can be described as: a. Something inside the nucleus breaks down causing a proton to become a neutron. b. It emits a positron which goes zooming off. c. The atomic number goes down by one and the mass number remains unchanged. Here is a typical po ...
... Positron decay is the mirror image of beta decay and can be described as: a. Something inside the nucleus breaks down causing a proton to become a neutron. b. It emits a positron which goes zooming off. c. The atomic number goes down by one and the mass number remains unchanged. Here is a typical po ...
Nuclear Decay
... • Alpha particles can burn your skin however they can also be stopped by a piece of paper ...
... • Alpha particles can burn your skin however they can also be stopped by a piece of paper ...
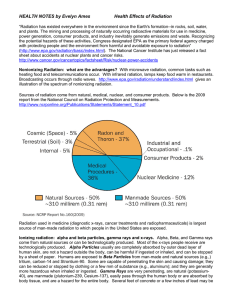
Health Effects of Radiation
... differ in origin; are generally lower in energy, therefore less penetrating than Gamma rays; and a few mm of lead can stop penetration of medical x-rays. How can alpha particles affect people’s health? The health effects of alpha particles depend heavily upon how exposure takes place. External expos ...
... differ in origin; are generally lower in energy, therefore less penetrating than Gamma rays; and a few mm of lead can stop penetration of medical x-rays. How can alpha particles affect people’s health? The health effects of alpha particles depend heavily upon how exposure takes place. External expos ...
Nuclear Chemistry
... Each of the two protons that form part of the Alpha particle emitted from an atom of uranium-238 possesses a unit of positive electric charge. Because the charge on the uranium-238 nucleus decreases by two units as a result of Alpha emission, the atomic number of the newly formed atom is 2 fewer tha ...
... Each of the two protons that form part of the Alpha particle emitted from an atom of uranium-238 possesses a unit of positive electric charge. Because the charge on the uranium-238 nucleus decreases by two units as a result of Alpha emission, the atomic number of the newly formed atom is 2 fewer tha ...
Chemistry: Matter and Change
... • Gamma rays (short wavelength) are photons, which are high-energy • Gamma rays have no mass or charge so the emission of gamma rays does not change the atomic number or mass number of a nucleus. • Gamma rays almost always accompany alpha and beta radiation. • X rays are a form of high-energy electr ...
... • Gamma rays (short wavelength) are photons, which are high-energy • Gamma rays have no mass or charge so the emission of gamma rays does not change the atomic number or mass number of a nucleus. • Gamma rays almost always accompany alpha and beta radiation. • X rays are a form of high-energy electr ...
Ch. 21.1 Nuclear Radiation
... • Easily pass through paper, wood, and human body (extremely dangerous). • They can be stopped, although not completely, by several meters of concrete or several centimeters of lead. ...
... • Easily pass through paper, wood, and human body (extremely dangerous). • They can be stopped, although not completely, by several meters of concrete or several centimeters of lead. ...
Radioactivity
... This means that they have no mass and no charge. • γ rays have a high penetrating power - it takes a thick sheet of metal such as lead, or concrete to reduce them significantly. • γ rays do not directly ionise other atoms, although they may cause atoms to emit other particles which will then cause i ...
... This means that they have no mass and no charge. • γ rays have a high penetrating power - it takes a thick sheet of metal such as lead, or concrete to reduce them significantly. • γ rays do not directly ionise other atoms, although they may cause atoms to emit other particles which will then cause i ...
nuclear test 2006
... Carbon Dating is a technique used to determine the age of organic material. The activity (rate of decay) of 14C atoms in the sample is measured. 14C has a half life of 5730 years. a) Explain how the activity of 14C in an organic sample can be used to determine its age. ...
... Carbon Dating is a technique used to determine the age of organic material. The activity (rate of decay) of 14C atoms in the sample is measured. 14C has a half life of 5730 years. a) Explain how the activity of 14C in an organic sample can be used to determine its age. ...
Radioactivity
... • describe the structure of an atom in terms of protons, neutrons and electrons and use symbols to describe particular nuclei • understand the terms atomic (proton) number, mass (nucleon) number and isotope • understand that ionising radiations can be detected using a photographic film or a GeigerMu ...
... • describe the structure of an atom in terms of protons, neutrons and electrons and use symbols to describe particular nuclei • understand the terms atomic (proton) number, mass (nucleon) number and isotope • understand that ionising radiations can be detected using a photographic film or a GeigerMu ...
NUCLEAR CHEMISTRY
... 1. A very heavy nucleus splits into more stable nuclei of intermediate mass 2. The mass of the products is less than the mass of the reactants. Missing mass is converted to energy a. Small amounts of missing mass are converted to HUGE amounts of energy (E = mc2) ...
... 1. A very heavy nucleus splits into more stable nuclei of intermediate mass 2. The mass of the products is less than the mass of the reactants. Missing mass is converted to energy a. Small amounts of missing mass are converted to HUGE amounts of energy (E = mc2) ...
File
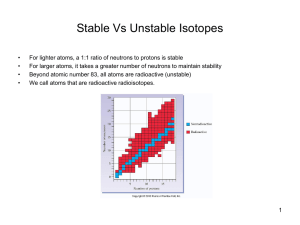
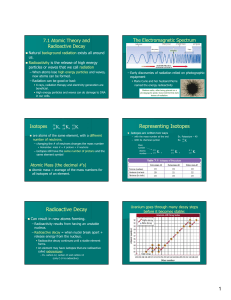
... Radioactivity: “the release of nuclear radiation in the form of particles & rays from a radioactive element.” Isotopes are often unstable – they have more neutrons than the element “wants” The isotopes are naturally occurring & decompose at different rates depending on the type of element. A ...
... Radioactivity: “the release of nuclear radiation in the form of particles & rays from a radioactive element.” Isotopes are often unstable – they have more neutrons than the element “wants” The isotopes are naturally occurring & decompose at different rates depending on the type of element. A ...
Representing Isotopes Radioactive Decay
... Radioactive decay continues until a stable element forms. An element may have isotopes that are radioactive called radioisotopes – Ex. carbon-12, carbon-13 and carbon-14 (only C-14 is radioactive) ...
... Radioactive decay continues until a stable element forms. An element may have isotopes that are radioactive called radioisotopes – Ex. carbon-12, carbon-13 and carbon-14 (only C-14 is radioactive) ...
The Manhattan Project and Beyond
... DE-NAZIFICATION after WWII Germany was divided into 4 parts Controlled by France, United States, Great Britain, and USSR Stalin agreed that “FREE ELECTIONS” would take place in Eastern (Soviet controlled) Germany THIS DID NOT HAPPEN and led to hostility during the Cold War between the USSR and ...
... DE-NAZIFICATION after WWII Germany was divided into 4 parts Controlled by France, United States, Great Britain, and USSR Stalin agreed that “FREE ELECTIONS” would take place in Eastern (Soviet controlled) Germany THIS DID NOT HAPPEN and led to hostility during the Cold War between the USSR and ...
Chemistry Test: Transmutation Multiple Choice 1. Identify the new
... Identify the new element when an alpha particle is emitted from Pu-244. a. Cm-240 b. Am-244 c. U-240 d. Np-244 Which of the following lists ranks nuclear radiation from most massive to least massive? a. alpha, beta, and gamma c. gamma, alpha, and beta b. beta, gamma, and alpha d. gamma, beta, and al ...
... Identify the new element when an alpha particle is emitted from Pu-244. a. Cm-240 b. Am-244 c. U-240 d. Np-244 Which of the following lists ranks nuclear radiation from most massive to least massive? a. alpha, beta, and gamma c. gamma, alpha, and beta b. beta, gamma, and alpha d. gamma, beta, and al ...
solutions - Physicsland
... 11. The proton “bullets” need enough momentum to overcome the electric force of repulsion they experience once they get close to the atomic nucleus. 14.The fusing of hydrogen atoms into helium atoms suggests that the electric force of repulsions can be overcome by the strong nuclear force if two ato ...
... 11. The proton “bullets” need enough momentum to overcome the electric force of repulsion they experience once they get close to the atomic nucleus. 14.The fusing of hydrogen atoms into helium atoms suggests that the electric force of repulsions can be overcome by the strong nuclear force if two ato ...
AP Exam Questions: Nuclear
... (c) An alpha particle, or He nuclei, has a 2+ charge and would be attracted to the (-) side of the electric field. A beta particle, , or electron, has a single negative charge and is attracted to the positive side of the electric field, but since it is much lighter and faster than an alpha it wou ...
... (c) An alpha particle, or He nuclei, has a 2+ charge and would be attracted to the (-) side of the electric field. A beta particle, , or electron, has a single negative charge and is attracted to the positive side of the electric field, but since it is much lighter and faster than an alpha it wou ...
Document
... Beta Decay • Beta particles β: e- ejected from the nucleus when neutrons decay • Beta particles have the same charge & mass as e-. • Can be stopped by aluminum foil or a block of wood. ...
... Beta Decay • Beta particles β: e- ejected from the nucleus when neutrons decay • Beta particles have the same charge & mass as e-. • Can be stopped by aluminum foil or a block of wood. ...
Senator Joseph McCarthy Hunts Communists at Home
... The Rosenbergs became the first American civilians to be executed for spying when they were put to death at Sing Sing Prison in Ossining, New York. After Julius Rosenberg became an engineer with the U.S. Army Signal Corps in 1940, Ethel's brother, Sgt. David Greenglass, who worked on the atomic bomb ...
... The Rosenbergs became the first American civilians to be executed for spying when they were put to death at Sing Sing Prison in Ossining, New York. After Julius Rosenberg became an engineer with the U.S. Army Signal Corps in 1940, Ethel's brother, Sgt. David Greenglass, who worked on the atomic bomb ...
Radioactivity and Nuclear Reactions
... • radioactivity—process of nuclear decay • All nuclei that contain more than 83 protons are radioactive. • Almost all elements with more than 92 ...
... • radioactivity—process of nuclear decay • All nuclei that contain more than 83 protons are radioactive. • Almost all elements with more than 92 ...
Untitled
... Cosmic rays are streams of electrically charged particles. Collisions between cosmic rays And Earth’s atmosphere shower the surface below with nuclear radiation. ...
... Cosmic rays are streams of electrically charged particles. Collisions between cosmic rays And Earth’s atmosphere shower the surface below with nuclear radiation. ...
Word - The Chemistry Book
... emitted from a nucleus as it changes from an excited state to a ground energy state 2. Gamma rays are produced when nuclear particles undergo transitions in energy levels; beta and gamma rays are usually emitted together 3. Gamma emission usually follows other types of decay that leave the nucleus i ...
... emitted from a nucleus as it changes from an excited state to a ground energy state 2. Gamma rays are produced when nuclear particles undergo transitions in energy levels; beta and gamma rays are usually emitted together 3. Gamma emission usually follows other types of decay that leave the nucleus i ...
Fallout shelter

A fallout shelter is an enclosed space specially designed to protect occupants from radioactive debris or fallout resulting from a nuclear explosion. Many such shelters were constructed as civil defense measures during the Cold War.During a nuclear explosion, matter vaporized in the resulting fireball is exposed to neutrons from the explosion, absorbs them, and becomes radioactive. When this material condenses in the rain, it forms dust and light sandy materials that resembles ground pumice. The fallout emits alpha and beta particles, as well as gamma rays.Much of this highly radioactive material falls to earth, subjecting anything within the line of sight to radiation, becoming a significant hazard. A fallout shelter is designed to allow its occupants to minimize exposure to harmful fallout until radioactivity has decayed to a safer level.