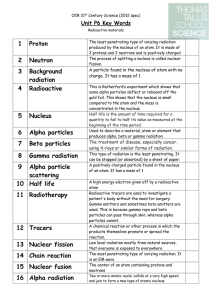
Nuclear Reactions
... • Beta particles are faster and more penetrating than Alpha particles • Can be stopped by a sheet of aluminum foil. ...
... • Beta particles are faster and more penetrating than Alpha particles • Can be stopped by a sheet of aluminum foil. ...
Radioactivity - Miami Beach Senior High School
... which are radioactive. • More neutrons are released. • The additional neutrons released may also hit other uranium nuclei and cause them to split. Even more neutrons are then released, which in turn can split more uranium nuclei. This is called a chain reaction. The chain reaction in nuclear reactor ...
... which are radioactive. • More neutrons are released. • The additional neutrons released may also hit other uranium nuclei and cause them to split. Even more neutrons are then released, which in turn can split more uranium nuclei. This is called a chain reaction. The chain reaction in nuclear reactor ...
Powerpoint Slides
... We can detect the radiation from a radioactive source. Say we get X counts/minute (cpm). ...
... We can detect the radiation from a radioactive source. Say we get X counts/minute (cpm). ...
Nuclear Chemistry
... interior of Earth. •Radiation occurs in all of our surroundings: air, water, soil. •Cosmic radiation reaches us every day. ...
... interior of Earth. •Radiation occurs in all of our surroundings: air, water, soil. •Cosmic radiation reaches us every day. ...
on Nuclear Physics - Good Earth School
... RUTHERFORD studied experimentally that the radiations given out by a radioactive substance in the presence of either a magnetic or an electric field gets separated into three constituents. Those which turn towards the negative plate are positively charged alpha α particles Those which turn towar ...
... RUTHERFORD studied experimentally that the radiations given out by a radioactive substance in the presence of either a magnetic or an electric field gets separated into three constituents. Those which turn towards the negative plate are positively charged alpha α particles Those which turn towar ...
Radioactivity2015
... • This type of decay process leaves the mass number of the nuclei unchanged. • The element is transformed to a new element • A beta particle is minute in comparison to that of an alpha particle and has about one hundred times the penetrating ability. Where an alpha particle can be stopped by a piece ...
... • This type of decay process leaves the mass number of the nuclei unchanged. • The element is transformed to a new element • A beta particle is minute in comparison to that of an alpha particle and has about one hundred times the penetrating ability. Where an alpha particle can be stopped by a piece ...
SCIENCE 10: (7.1) ATOMIC THEORY, ISOTOPES
... the mass numbers are equal on both sides of the arrow. Also the product nucleus has an atomic number that is greater, making it an atom of the next higher element and a mass number that equal to the reactant because the beta particle emitted was an electron) ...
... the mass numbers are equal on both sides of the arrow. Also the product nucleus has an atomic number that is greater, making it an atom of the next higher element and a mass number that equal to the reactant because the beta particle emitted was an electron) ...
Nuclear Chemistry - Northwest ISD Moodle
... http://www.esrl.noaa.gov/gmd/outreach/isotopes/images/beta_decay.jpg ...
... http://www.esrl.noaa.gov/gmd/outreach/isotopes/images/beta_decay.jpg ...
Nuclear - chemmybear.com
... (c) The neutron/proton ratio in Sr-90 and Cs-137 is (c) Gamma rays are observed during the radioactive too large and they emit beta particles (converting decay of carbon-11. Why is it unnecessary to inneutrons into protons) to lower this ratio. clude the gamma rays in the radioactive decay (d) Large ...
... (c) The neutron/proton ratio in Sr-90 and Cs-137 is (c) Gamma rays are observed during the radioactive too large and they emit beta particles (converting decay of carbon-11. Why is it unnecessary to inneutrons into protons) to lower this ratio. clude the gamma rays in the radioactive decay (d) Large ...
Summative Assessment Review!
... • Atoms with the same number of protons but a different number of neutrons. • Example: an atom with one proton, one electron and one neutron is hydrogen. • However, if the hydrogen atoms has two particles (neutrons) but the same number of protons (in the case of hydrogen one) it is an isotope of hyd ...
... • Atoms with the same number of protons but a different number of neutrons. • Example: an atom with one proton, one electron and one neutron is hydrogen. • However, if the hydrogen atoms has two particles (neutrons) but the same number of protons (in the case of hydrogen one) it is an isotope of hyd ...
Chp 7.1 Atomic Theory and Radioactive Decay
... • Radioactivity results from having an unstable nucleus. • When these nuclei lose energy and break apart, decay occurs. • Radioactive decay releases energy from the nucleus as radiation. • Radioactive atoms release energy until they become stable, often as different atoms. • An element may have only ...
... • Radioactivity results from having an unstable nucleus. • When these nuclei lose energy and break apart, decay occurs. • Radioactive decay releases energy from the nucleus as radiation. • Radioactive atoms release energy until they become stable, often as different atoms. • An element may have only ...
Multiple Choice Questions
... (1) small amounts of energy are converted into large amounts of matter (2) small amounts of matter are converted into large amounts of energy (3) heavy nuclei are split into lighter nuclei (4) light nuclei are combined into heavier nuclei ...
... (1) small amounts of energy are converted into large amounts of matter (2) small amounts of matter are converted into large amounts of energy (3) heavy nuclei are split into lighter nuclei (4) light nuclei are combined into heavier nuclei ...
Half-life and Radioactive Decay guided notes
... and two neutrons released from the nucleus of the radioactive atom. This means that alpha particles have a ...
... and two neutrons released from the nucleus of the radioactive atom. This means that alpha particles have a ...
Radioactivity
... • When an unstable nucleus emits one or more particles or energy • When these particles are emitted, the element changes to another isotope or to a different element • Nuclear radiation refers to radiation resulting from nuclear changes ...
... • When an unstable nucleus emits one or more particles or energy • When these particles are emitted, the element changes to another isotope or to a different element • Nuclear radiation refers to radiation resulting from nuclear changes ...
radioisotopes and radiotherapy - video
... 1. Isotopes are nuclides with the same number of protons but different number of neutrons. Isotopes have same atomic number but different mass number. 2. Radioisotopes are unstable isotopes, where their nuclei decay to a more stable state or type of nuclei, giving off matter and energy as radiation. ...
... 1. Isotopes are nuclides with the same number of protons but different number of neutrons. Isotopes have same atomic number but different mass number. 2. Radioisotopes are unstable isotopes, where their nuclei decay to a more stable state or type of nuclei, giving off matter and energy as radiation. ...
nuclear force
... Alpha particles consist of two protons and two neutrons, and are emitted during some kinds of radioactive decay. ...
... Alpha particles consist of two protons and two neutrons, and are emitted during some kinds of radioactive decay. ...
3 Background radiation
... A high energy electron given off by a radioactive atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. ...
... A high energy electron given off by a radioactive atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. ...
Unit 14 Notes - shscience.net
... protons, 2 neutrons 42He nucleus Beta Particle (β-) Electron emitted from the nucleus 0-1e Positron Particle (β+) Mass of an electron but positive charge 0+1e Gamma Radiation (γ) High energy radiation (higher than x-ray) No mass and no charge ...
... protons, 2 neutrons 42He nucleus Beta Particle (β-) Electron emitted from the nucleus 0-1e Positron Particle (β+) Mass of an electron but positive charge 0+1e Gamma Radiation (γ) High energy radiation (higher than x-ray) No mass and no charge ...
Radioactivity Reading Assignment Name: Chemistry Date: Hour
... Compared to light, each bit (photon) of gamma radiation has 1 million times as much energy (or 1000 time more energy than an X-ray photon). Gamma rays travel at the speed of light. They happily travel through centimeters of lead and travel easily through air. They are a danger to the human body even ...
... Compared to light, each bit (photon) of gamma radiation has 1 million times as much energy (or 1000 time more energy than an X-ray photon). Gamma rays travel at the speed of light. They happily travel through centimeters of lead and travel easily through air. They are a danger to the human body even ...
Isotopes and Radioactive Decay
... GAMMA RADIATION: Radiation that is made up of ________________________ rays. A gamma ray is high-energy and contains no _____________ and is represented by the symbol __________. Gamma rays usually accompany ___________________ and ___________________ radiation. Gamma rays also account for _________ ...
... GAMMA RADIATION: Radiation that is made up of ________________________ rays. A gamma ray is high-energy and contains no _____________ and is represented by the symbol __________. Gamma rays usually accompany ___________________ and ___________________ radiation. Gamma rays also account for _________ ...
printable version
... b. Beta emission occurs in isotopes that have too many neutrons ( more neutrons than protons in small atoms) like Cobalt – 60 ( 27 protons and 33 neutrons) c. Isotopes that have too few neutrons (like nitrogen-13) would attain stability by electron capture or positron emission ...
... b. Beta emission occurs in isotopes that have too many neutrons ( more neutrons than protons in small atoms) like Cobalt – 60 ( 27 protons and 33 neutrons) c. Isotopes that have too few neutrons (like nitrogen-13) would attain stability by electron capture or positron emission ...
File
... Beta Particles are faster than alpha because they’re smaller & lighter so they penetrate deeper into material they hit. •Pass through paper and skin •Aluminum foil will stop a beta particle •Can damage human cells if released inside the body ...
... Beta Particles are faster than alpha because they’re smaller & lighter so they penetrate deeper into material they hit. •Pass through paper and skin •Aluminum foil will stop a beta particle •Can damage human cells if released inside the body ...
File
... There are three types of radiation most frequently emitted from radioactive nuclei. 1. Alpha particles: They consist of two protons and two neutrons. Identical with the helium nucleus. The range of α particles in air is about 5 cm, and less than 100µ in tissue. 2. Beta particles: Are ...
... There are three types of radiation most frequently emitted from radioactive nuclei. 1. Alpha particles: They consist of two protons and two neutrons. Identical with the helium nucleus. The range of α particles in air is about 5 cm, and less than 100µ in tissue. 2. Beta particles: Are ...
What do I know about……
... understand that alpha and beta particles and gamma rays are ionising radiations emitted from unstable nuclei in a random process describe the nature of alpha and beta particles and gamma rays and recall that they may be distinguished in terms of penetrating power describe the effects on the atomic a ...
... understand that alpha and beta particles and gamma rays are ionising radiations emitted from unstable nuclei in a random process describe the nature of alpha and beta particles and gamma rays and recall that they may be distinguished in terms of penetrating power describe the effects on the atomic a ...
Revision of Atomic Structure and Nuclide Notations Nuclide
... A neutron has been changed into a proton and an electron. Gamma emission often happens at the same time but has no effect on the mass number or atomic number. It is not included in nuclear equations. Balancing the Numbers! In nuclear equations the top numbers (mass numbers) must add up to the same n ...
... A neutron has been changed into a proton and an electron. Gamma emission often happens at the same time but has no effect on the mass number or atomic number. It is not included in nuclear equations. Balancing the Numbers! In nuclear equations the top numbers (mass numbers) must add up to the same n ...
Fallout shelter

A fallout shelter is an enclosed space specially designed to protect occupants from radioactive debris or fallout resulting from a nuclear explosion. Many such shelters were constructed as civil defense measures during the Cold War.During a nuclear explosion, matter vaporized in the resulting fireball is exposed to neutrons from the explosion, absorbs them, and becomes radioactive. When this material condenses in the rain, it forms dust and light sandy materials that resembles ground pumice. The fallout emits alpha and beta particles, as well as gamma rays.Much of this highly radioactive material falls to earth, subjecting anything within the line of sight to radiation, becoming a significant hazard. A fallout shelter is designed to allow its occupants to minimize exposure to harmful fallout until radioactivity has decayed to a safer level.