field desorption (in mass spectrometry)
... identifying the resulting fragment ions. This information can then be pieced together to generate structural information regarding the intact molecule. Tandem mass spectrometry also enables specific compounds to be detected in complex mixtures on account of their specific and characteristic fragment ...
... identifying the resulting fragment ions. This information can then be pieced together to generate structural information regarding the intact molecule. Tandem mass spectrometry also enables specific compounds to be detected in complex mixtures on account of their specific and characteristic fragment ...
Mass Spectrometry and Organic
... question. Molecular weight is not ambiguous. A compound has a unique MW. Our ability to analyze compounds on this basis, depends completely on being able to generate ions from the compound. Specifically molecular ions*, whose weight is equal to the MW of the compound, are critical. Once produced, ou ...
... question. Molecular weight is not ambiguous. A compound has a unique MW. Our ability to analyze compounds on this basis, depends completely on being able to generate ions from the compound. Specifically molecular ions*, whose weight is equal to the MW of the compound, are critical. Once produced, ou ...
Mass Spectrometry of Peptides
... 1. Proteins digested w/ an enzyme to produce peptides 2. Peptides charged (ionized) and separated according to their different m/z ratios 3. Each peptide fragmented into ions and m/z values of fragment ions are measured ...
... 1. Proteins digested w/ an enzyme to produce peptides 2. Peptides charged (ionized) and separated according to their different m/z ratios 3. Each peptide fragmented into ions and m/z values of fragment ions are measured ...
Expected paths Observed paths Nuclear atom • Rutherford
... o Groups are numbered left to right, from 1 to 18. o Periods are numbered top to bottom, from 1 to 7. • Elements are separated into three types, metals and nonmetals (separated by a thick, stair-step line), and metalloids (or semimetals) which occur along the boundary line. • Certain Groups have fam ...
... o Groups are numbered left to right, from 1 to 18. o Periods are numbered top to bottom, from 1 to 7. • Elements are separated into three types, metals and nonmetals (separated by a thick, stair-step line), and metalloids (or semimetals) which occur along the boundary line. • Certain Groups have fam ...
Lecture 13
... Breuker, K.; McLafferty, F. W. Native electron capture dissociation for the structural characterization of noncovalent interactions in native cytochrome c Angewandte Chemie-International Edition 2003, 42, 4900-4904. ...
... Breuker, K.; McLafferty, F. W. Native electron capture dissociation for the structural characterization of noncovalent interactions in native cytochrome c Angewandte Chemie-International Edition 2003, 42, 4900-4904. ...
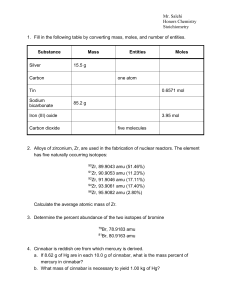
CHE1031 Chapter 2 Practice Quiz Key
... Isotopes & Average Atomic Weight 7. The element X has three isotopes whose masses and abundances are shown below. Calculate the average atomic mass of element X. ...
... Isotopes & Average Atomic Weight 7. The element X has three isotopes whose masses and abundances are shown below. Calculate the average atomic mass of element X. ...
Key
... 9. The element X has three isotopes whose masses and abundances are shown below. Calculate the average atomic mass of element X. isotope abundance (%) mass (amu) ...
... 9. The element X has three isotopes whose masses and abundances are shown below. Calculate the average atomic mass of element X. isotope abundance (%) mass (amu) ...
Stoichiometry - Cloudfront.net
... The substance formed 279.3 mg of CO2 and 66.46 mg H2O on combustion. The compound contains 4.680% N by mass. Is the white solid cocaine? 8. An unknown compound (molar mass = 176 g/mol) contains 68.2 mass % C, 6.86 mass % H, 15.9 mass % N, and 9.08 mass % O. What is the molecular formula? 9. Calcium ...
... The substance formed 279.3 mg of CO2 and 66.46 mg H2O on combustion. The compound contains 4.680% N by mass. Is the white solid cocaine? 8. An unknown compound (molar mass = 176 g/mol) contains 68.2 mass % C, 6.86 mass % H, 15.9 mass % N, and 9.08 mass % O. What is the molecular formula? 9. Calcium ...
Chapter 7 - Midway ISD
... element are present in the molecular formula Less electronegative element is listed first and is only given a prefix if there is more than one 2nd element is given a prefix, then the root of the element name, and then –ide ending “o” or “a” at end of prefix is dropped when element name starts with a ...
... element are present in the molecular formula Less electronegative element is listed first and is only given a prefix if there is more than one 2nd element is given a prefix, then the root of the element name, and then –ide ending “o” or “a” at end of prefix is dropped when element name starts with a ...
Word - ASDL Community
... While enzymes, such as trypsin, can be used to cleave proteins and peptides at specific amino acid linkages, we can also fragment peptides inside of a mass spectrometer to obtain additional information. These types of experiments are called tandem MS or MS-MS experiments. These experiments are parti ...
... While enzymes, such as trypsin, can be used to cleave proteins and peptides at specific amino acid linkages, we can also fragment peptides inside of a mass spectrometer to obtain additional information. These types of experiments are called tandem MS or MS-MS experiments. These experiments are parti ...
Abstract_Metabolomic_RFMF
... biological pathways: the Glucose-6-Phosphate is implicated in glycolysis that converts glucose to pyruvate. This metabolism permits the synthesis of ATP, which one has a critical role in energy transfer and metabolism. ATP is also implicated in the Krebs Cycle (or Citrate Cycle), which is another ex ...
... biological pathways: the Glucose-6-Phosphate is implicated in glycolysis that converts glucose to pyruvate. This metabolism permits the synthesis of ATP, which one has a critical role in energy transfer and metabolism. ATP is also implicated in the Krebs Cycle (or Citrate Cycle), which is another ex ...
Reacting Mass calculations
... 5.00 g of hydrated sodium sulphate crystals (Na2SO4.nH2O) gave 2.20 g of anhydrous sodium sulphate on heating to constant mass. Work out the relative molecular mass (Mr) of the hydrated sodium sulphate and the value of n. Na2SO4.nH2O Na2SO4 + n H2O ...
... 5.00 g of hydrated sodium sulphate crystals (Na2SO4.nH2O) gave 2.20 g of anhydrous sodium sulphate on heating to constant mass. Work out the relative molecular mass (Mr) of the hydrated sodium sulphate and the value of n. Na2SO4.nH2O Na2SO4 + n H2O ...
Proteomics and Mass Spectroscopy
... it overcomes the propensity of these molecules to fragment when ionized • Is the primary ion source used in liquid chromatography-mass spec because it's a liquid-gas interface that is capable of coupling liquid chomatography with mass spectrometry ...
... it overcomes the propensity of these molecules to fragment when ionized • Is the primary ion source used in liquid chromatography-mass spec because it's a liquid-gas interface that is capable of coupling liquid chomatography with mass spectrometry ...
Peptide Sequencing by Mass Spectrometry
... 1. Proteins digested w/ an enzyme to produce peptides 2. Peptides charged (ionized) and separated according to their different m/z ratios 3. Each peptide fragmented into ions and m/z values of fragment ions are measured ...
... 1. Proteins digested w/ an enzyme to produce peptides 2. Peptides charged (ionized) and separated according to their different m/z ratios 3. Each peptide fragmented into ions and m/z values of fragment ions are measured ...
Midterm Review Questions and Answers
... 19. A student determines the density of a piece of aluminum to be 2.2 g/cm3. What is his percent error? ...
... 19. A student determines the density of a piece of aluminum to be 2.2 g/cm3. What is his percent error? ...
South Pasadena · AP Chemistry
... percentage abundance. Calculate the average atomic mass of an element using the percent abundance and mass of each isotope. Calculate the percent abundance of isotopes given the average atomic mass and isotopic masses of an element. ...
... percentage abundance. Calculate the average atomic mass of an element using the percent abundance and mass of each isotope. Calculate the percent abundance of isotopes given the average atomic mass and isotopic masses of an element. ...
Chapter 2: Target/decoy search strategy for increased
... derived from set numbers of correct and incorrect PSMs. This program exploited the target-decoy principle and therefore relied on the same assumptions explained previously, namely that all decoy hits are incorrect, and that there are equal numbers of incorrect target and decoy hits. The program too ...
... derived from set numbers of correct and incorrect PSMs. This program exploited the target-decoy principle and therefore relied on the same assumptions explained previously, namely that all decoy hits are incorrect, and that there are equal numbers of incorrect target and decoy hits. The program too ...
name - cloudfront.net
... carbon to give aluminum metal and carbon monoxide. If the yield of this reaction is 75%, what mass of aluminum metal can be produced from the reaction of 1.65 106 g of aluminum oxide with 1.50 106 g of carbon? (6.6 105 g) 15. Which of the following compounds is a strong electrolyte? A. H2O B. ...
... carbon to give aluminum metal and carbon monoxide. If the yield of this reaction is 75%, what mass of aluminum metal can be produced from the reaction of 1.65 106 g of aluminum oxide with 1.50 106 g of carbon? (6.6 105 g) 15. Which of the following compounds is a strong electrolyte? A. H2O B. ...
Mass Spectrometry - University of Exeter
... molecule by measuring the mass-tocharge ratio (m/z) of its ion ...
... molecule by measuring the mass-tocharge ratio (m/z) of its ion ...
answers - van Maarseveen
... 3. Do the following calculations making sure that you show all the necessary steps. i) ii) iii) ...
... 3. Do the following calculations making sure that you show all the necessary steps. i) ii) iii) ...
Proteomics - OpenWetWare
... Step 4: Separate chains (2D gel, gas or liquid chromatography) Step 5: Analyze separated protein parts by mass spectrometry ...
... Step 4: Separate chains (2D gel, gas or liquid chromatography) Step 5: Analyze separated protein parts by mass spectrometry ...
Document
... Applications of LC/MS • Forensic toxicology / Drugs of abuse • Clinical toxicology/TDM • Clinical endocrinology • Clinical chemistry • Inborn errors of metabolism ...
... Applications of LC/MS • Forensic toxicology / Drugs of abuse • Clinical toxicology/TDM • Clinical endocrinology • Clinical chemistry • Inborn errors of metabolism ...
A Database of Peak Annotations of Empirically Derived Mass Spectra
... industry standard supported by HUPO. The IBG-MSP also supports MS/MS annotations, where peaks may be annotated using terminology conventionally used in describing MS/MS fragment ion series. This is accomplished through the implementation of algorithms based on the fragmentation rules of Collisionall ...
... industry standard supported by HUPO. The IBG-MSP also supports MS/MS annotations, where peaks may be annotated using terminology conventionally used in describing MS/MS fragment ion series. This is accomplished through the implementation of algorithms based on the fragmentation rules of Collisionall ...
Mass spectrometry
Mass spectrometry (MS) is an analytical chemistry technique that helps identify the amount and type of chemicals present in a sample by measuring the mass-to-charge ratio and abundance of gas-phase ions.A mass spectrum (plural spectra) is a plot of the ion signal as a function of the mass-to-charge ratio. The spectra are used to determine the elemental or isotopic signature of a sample, the masses of particles and of molecules, and to elucidate the chemical structures of molecules, such as peptides and other chemical compounds. Mass spectrometry works by ionizing chemical compounds to generate charged molecules or molecule fragments and measuring their mass-to-charge ratios.In a typical MS procedure, a sample, which may be solid, liquid, or gas, is ionized, for example by bombarding it with electrons. This may cause some of the sample's molecules to break into charged fragments. These ions are then separated according to their mass-to-charge ratio, typically by accelerating them and subjecting them to an electric or magnetic field: ions of the same mass-to-charge ratio will undergo the same amount of deflection. The ions are detected by a mechanism capable of detecting charged particles, such as an electron multiplier. Results are displayed as spectra of the relative abundance of detected ions as a function of the mass-to-charge ratio. The atoms or molecules in the sample can be identified by correlating known masses to the identified masses or through a characteristic fragmentation pattern.