Protein Structure Function Relationship
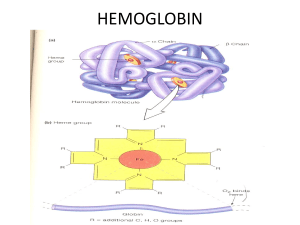
... The ability of myoglobin or hemoglobin to bind oxygen depends on the presence of a heme prosthetic group. It is the heme that gives these proteins their characterisitc red color. All four chains of hemoglbin contain one identical heme prostetic groups. All of the heme in both myoglobin and hemoglob ...
... The ability of myoglobin or hemoglobin to bind oxygen depends on the presence of a heme prosthetic group. It is the heme that gives these proteins their characterisitc red color. All four chains of hemoglbin contain one identical heme prostetic groups. All of the heme in both myoglobin and hemoglob ...
MStage861092014Competency208.5.3
... Myoglobin Structure Cont. The iron atom is always open at the end of each heme group and can form two additional bonds at the end or each heme group The bonding sites that these create are what is essentially referred to as coordination sites. These coordination sites are recurrent and go to ...
... Myoglobin Structure Cont. The iron atom is always open at the end of each heme group and can form two additional bonds at the end or each heme group The bonding sites that these create are what is essentially referred to as coordination sites. These coordination sites are recurrent and go to ...
Hemoglobin - Medico Tutorials
... Combine with O2 Combine with CO2 Combine with CO Combine with NO Combine with H Ions ...
... Combine with O2 Combine with CO2 Combine with CO Combine with NO Combine with H Ions ...
Lung Capacity - SPatriotsSBI3U
... Once the venous blood that is carrying carbon dioxide in all of its forms has reached the lungs, the oxygen dislodges the hydrogen ions so that they can combine with the Bicarbonate ions to form Carbon dioxide and water The highly concentrated carbon dioxide diffuses from the blood into the lungs __ ...
... Once the venous blood that is carrying carbon dioxide in all of its forms has reached the lungs, the oxygen dislodges the hydrogen ions so that they can combine with the Bicarbonate ions to form Carbon dioxide and water The highly concentrated carbon dioxide diffuses from the blood into the lungs __ ...
Red Blood Cells - Amazon Web Services
... Improve oxygen carrying capacity in critical cases, as well as treatment for symptomatic anemia. Contraindications Red-cell-containing components should not be used to treat anemias that can be corrected with specific hematinic medications such as iron, vitamin B12, folic acid, or erythropoietin. RB ...
... Improve oxygen carrying capacity in critical cases, as well as treatment for symptomatic anemia. Contraindications Red-cell-containing components should not be used to treat anemias that can be corrected with specific hematinic medications such as iron, vitamin B12, folic acid, or erythropoietin. RB ...
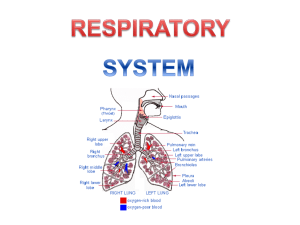
Respiratory System2010_2
... they are in an environment where oxygen is always at a higher concentration outside the cell than inside the cell so oxygen will continue to diffuse into cell. • Humans bodies are to thick and only our outside cells are exposed to air. Diffusion wouldn’t be efficient. A ventilation system is needed ...
... they are in an environment where oxygen is always at a higher concentration outside the cell than inside the cell so oxygen will continue to diffuse into cell. • Humans bodies are to thick and only our outside cells are exposed to air. Diffusion wouldn’t be efficient. A ventilation system is needed ...
transport of oxygen and oxygen-hemoglobin dissociation curve
... • Carboxy hemoglobin – contains CO instead of O2 (cherry red in color) • Sulfa – hemoglobin- contains sulfur – green in color ...
... • Carboxy hemoglobin – contains CO instead of O2 (cherry red in color) • Sulfa – hemoglobin- contains sulfur – green in color ...
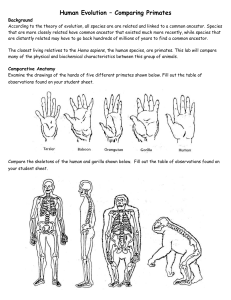
Human Evolution – Comparing Primates Background According to
... Animals with red blood each have a large protein molecule called hemoglobin in their blood cells. Hemoglobin is needed to transport oxygen from the lungs to the rest of the body. Hemoglobin, like all proteins, is made of amino acids. Below is a summary of some of the amino acids of hemoglobin. Parti ...
... Animals with red blood each have a large protein molecule called hemoglobin in their blood cells. Hemoglobin is needed to transport oxygen from the lungs to the rest of the body. Hemoglobin, like all proteins, is made of amino acids. Below is a summary of some of the amino acids of hemoglobin. Parti ...
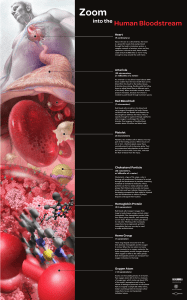
Zoom into the Human Bloodstream Annotated
... four heme group subunits, each containing an iron atom. These iron atoms give blood its red color. Working at the nanoscale, researchers have engineered synthetic hemoglobin that may one day be used to make artificial blood. ...
... four heme group subunits, each containing an iron atom. These iron atoms give blood its red color. Working at the nanoscale, researchers have engineered synthetic hemoglobin that may one day be used to make artificial blood. ...
p(O 2 )
... The oxygen affinity of unloaded hemoglobin is smaller than that of individual subunits. Oxygen binding to one subunit of hemoglobin favors the planar form at neighboring subunits fully loaded hemoglobin has an affinity similar to that of an individual subunit. http://www.chemistry.wustl.edu/~edud ...
... The oxygen affinity of unloaded hemoglobin is smaller than that of individual subunits. Oxygen binding to one subunit of hemoglobin favors the planar form at neighboring subunits fully loaded hemoglobin has an affinity similar to that of an individual subunit. http://www.chemistry.wustl.edu/~edud ...
Blood Disorders
... caused when the mechanisms that defend against oxidative stress within the red blood cell are overwhelmed and the oxygen carrying ferrous ion (Fe2+) of the heme group of the hemoglobin molecule is oxidized to the ferric state (Fe3+). • This converts hemoglobin to methemoglobin, resulting in a reduce ...
... caused when the mechanisms that defend against oxidative stress within the red blood cell are overwhelmed and the oxygen carrying ferrous ion (Fe2+) of the heme group of the hemoglobin molecule is oxidized to the ferric state (Fe3+). • This converts hemoglobin to methemoglobin, resulting in a reduce ...
Sickle Cell Anemia
... Sickle red blood cells become hard, sticky and shaped like sickles used to cut wheat. When these hard and pointed red cells go through the small blood tube, they clog the flow and break apart. This can cause pain, damage and a low blood count, or anemia. ...
... Sickle red blood cells become hard, sticky and shaped like sickles used to cut wheat. When these hard and pointed red cells go through the small blood tube, they clog the flow and break apart. This can cause pain, damage and a low blood count, or anemia. ...
Hemoglobin Hb
... The hemoglobin protein is one of the most important proteins in our body as it is responsible for the binding and transport of oxygen in the blood. Various scientists in the 1800s began to slowly discover the properties of the protein, its interaction with heme and its presence in blood cells. Howev ...
... The hemoglobin protein is one of the most important proteins in our body as it is responsible for the binding and transport of oxygen in the blood. Various scientists in the 1800s began to slowly discover the properties of the protein, its interaction with heme and its presence in blood cells. Howev ...
Hemoglobin

Hemoglobin (/ˈhiːmɵˌɡloʊbɨn/); also spelled haemoglobin and abbreviated Hb or Hgb, is the iron-containing oxygen-transport metalloprotein in the red blood cells of all vertebrates (with the exception of the fish family Channichthyidae) as well as the tissues of some invertebrates. Hemoglobin in the blood carries oxygen from the respiratory organs (lungs or gills) to the rest of the body (i.e. the tissues). There it releases the oxygen to permit aerobic respiration to provide energy to power the functions of the organism in the process called metabolism.In mammals, the protein makes up about 96% of the red blood cells' dry content (by weight), and around 35% of the total content (including water). Hemoglobin has an oxygen-binding capacity of 1.34 mL O2 per gram, which increases the total blood oxygen capacity seventy-fold compared to dissolved oxygen in blood. The mammalian hemoglobin molecule can bind (carry) up to four oxygen molecules.Hemoglobin is involved in the transport of other gases: It carries some of the body's respiratory carbon dioxide (about 10% of the total) as carbaminohemoglobin, in which CO2 is bound to the globin protein. The molecule also carries the important regulatory molecule nitric oxide bound to a globin protein thiol group, releasing it at the same time as oxygen.Hemoglobin is also found outside red blood cells and their progenitor lines. Other cells that contain hemoglobin include the A9 dopaminergic neurons in the substantia nigra, macrophages, alveolar cells, and mesangial cells in the kidney. In these tissues, hemoglobin has a non-oxygen-carrying function as an antioxidant and a regulator of iron metabolism.Hemoglobin and hemoglobin-like molecules are also found in many invertebrates, fungi, and plants. In these organisms, hemoglobins may carry oxygen, or they may act to transport and regulate other things such as carbon dioxide, nitric oxide, hydrogen sulfide and sulfide. A variant of the molecule, called leghemoglobin, is used to scavenge oxygen away from anaerobic systems, such as the nitrogen-fixing nodules of leguminous plants, before the oxygen can poison the system.