1. Kathy Zabrocka - Sickle Cell Anemia
... Novel Diagnostics & Therapies SCA is a disease caused by a single point mutation in the HBB gene (11p15.5). Hemoglobin normally consists of a chain of four subunits – two alpha-globin and two beta-globin. This SNP is simply a replacement of one Adenine with a Thymine in the HBB gene. which thus ...
... Novel Diagnostics & Therapies SCA is a disease caused by a single point mutation in the HBB gene (11p15.5). Hemoglobin normally consists of a chain of four subunits – two alpha-globin and two beta-globin. This SNP is simply a replacement of one Adenine with a Thymine in the HBB gene. which thus ...
Hemoglobin
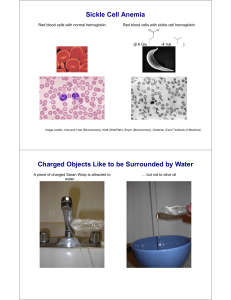
... The substitution forms a protrusion on the β-globin chain of hemoglobin Protrusion fits into a complementary site on the α chain of another hemoglobin molecule At low oxygen, deoxyhemoglobin S polymerizes into a network of fibres that distort RBCs sickled cells block the flow of blood in capillaries ...
... The substitution forms a protrusion on the β-globin chain of hemoglobin Protrusion fits into a complementary site on the α chain of another hemoglobin molecule At low oxygen, deoxyhemoglobin S polymerizes into a network of fibres that distort RBCs sickled cells block the flow of blood in capillaries ...
HgB - idexx.eu
... Hemoglobin may be measured by either a modified hemoglobin cyanide method (a blood sample is lysed and hemoglobin is released from the red blood cells, the hemoglobin is then treated with a cyanide compound, and concentration then evaluated by spectrophotometry) or directly within the red blood cell ...
... Hemoglobin may be measured by either a modified hemoglobin cyanide method (a blood sample is lysed and hemoglobin is released from the red blood cells, the hemoglobin is then treated with a cyanide compound, and concentration then evaluated by spectrophotometry) or directly within the red blood cell ...
Hemoglobin /he·mo·glo·bin/ (he´mo
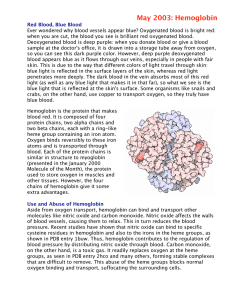
... the liver. The iron is transported to the bone marrow to be incorporated in the hemoglobin of newly formed erythrocytes. The hemoglobin concentration of blood varies with the hematocrit. The normal values for the blood hemoglobin concentration are 13.5 to 18.0 g/100 ml in males and 12.0 to 16.0 g/10 ...
... the liver. The iron is transported to the bone marrow to be incorporated in the hemoglobin of newly formed erythrocytes. The hemoglobin concentration of blood varies with the hematocrit. The normal values for the blood hemoglobin concentration are 13.5 to 18.0 g/100 ml in males and 12.0 to 16.0 g/10 ...
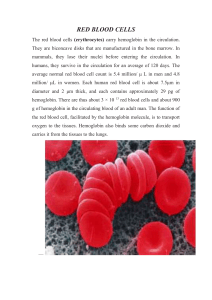
RED BLOOD CELLS The red blood cells (erythrocytes) carry
... The γ chains also contain 146 amino acid residues but have 37 that differ from those in the β chain. Fetal hemoglobin is normally replaced by adult hemoglobin soon after birth. In certain individuals, it fails to disappear and persists throughout life. In the body, its O2 content at a given PO2 is g ...
... The γ chains also contain 146 amino acid residues but have 37 that differ from those in the β chain. Fetal hemoglobin is normally replaced by adult hemoglobin soon after birth. In certain individuals, it fails to disappear and persists throughout life. In the body, its O2 content at a given PO2 is g ...
Sickle Cell Anemia
... Consequences of Red Blood Cell Sickling • Occlusion of small blood vessels, causing tissue damage ...
... Consequences of Red Blood Cell Sickling • Occlusion of small blood vessels, causing tissue damage ...
Heme group
... -Myoglobin is O2 binding protein found in almost all mammals mainly in muscles and heart -its main function is to store O2 for periods where energy demands is high, it also increases the rate of transport of oxygen within the muscle cells. - Compact structure, 80% of its polypeptide chain is α- ...
... -Myoglobin is O2 binding protein found in almost all mammals mainly in muscles and heart -its main function is to store O2 for periods where energy demands is high, it also increases the rate of transport of oxygen within the muscle cells. - Compact structure, 80% of its polypeptide chain is α- ...
Hemoglobinopathies
... members. DNA testing is not routinely done but can be used to help diagnose hemoglobin variants, thalassemia, and to determine carrier ...
... members. DNA testing is not routinely done but can be used to help diagnose hemoglobin variants, thalassemia, and to determine carrier ...
BIOLOGY LAB: How can a MUTATION in DNA affect
... INTRODUCTION: Sometimes the DNA code that makes up a gene has an error in it. This error is called a MUTATION. When the DNA contains an error, the mRNA it makes will copy that error. When the mRNA contains an error, it will code for incorrect amino acids and produce an incorrect protein. Sickle-cell ...
... INTRODUCTION: Sometimes the DNA code that makes up a gene has an error in it. This error is called a MUTATION. When the DNA contains an error, the mRNA it makes will copy that error. When the mRNA contains an error, it will code for incorrect amino acids and produce an incorrect protein. Sickle-cell ...
Ch. 12 note packet
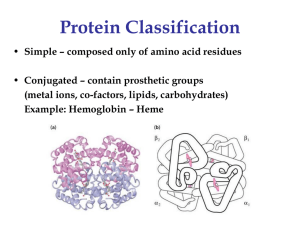
... • prosthetic group, heme, held in cavity inside by hydrophobic attractive forces • oxygen binds at Fe2+ site Hemoglobin – • 4 polypeptide chains (2 and 2 ) • -helices separated by -turns • hydrophobic residues where myoglobin has hydrophilic which serve to ...
... • prosthetic group, heme, held in cavity inside by hydrophobic attractive forces • oxygen binds at Fe2+ site Hemoglobin – • 4 polypeptide chains (2 and 2 ) • -helices separated by -turns • hydrophobic residues where myoglobin has hydrophilic which serve to ...
Oxygen concentration in plasma and tissue
... 1.725(mM) x 3.47% = 0.06 mM = 60 M The hemoglobin concentration in gray matter is given by 1.725(mM) x 4.73% = 0.082 mM = 82 M We usually use 75 M for baseline Hbt concentration. This seems reasonable. Note on solubility coefficient of oxygen in plasma In Valabregue et al 2003, the solubility coe ...
... 1.725(mM) x 3.47% = 0.06 mM = 60 M The hemoglobin concentration in gray matter is given by 1.725(mM) x 4.73% = 0.082 mM = 82 M We usually use 75 M for baseline Hbt concentration. This seems reasonable. Note on solubility coefficient of oxygen in plasma In Valabregue et al 2003, the solubility coe ...
ppt
... The figures are found at http://www.virtuallaboratory.net/Biofundamentals/lectureNotes/AllGraphics/myoglobinSurface.jpg and http://courses.washington.edu/conj/protein/hemo.gif (March 2007) ...
... The figures are found at http://www.virtuallaboratory.net/Biofundamentals/lectureNotes/AllGraphics/myoglobinSurface.jpg and http://courses.washington.edu/conj/protein/hemo.gif (March 2007) ...
circumlocutions
... The author ‘s finis at the end of Of Mice and Men left the audience in tears. ...
... The author ‘s finis at the end of Of Mice and Men left the audience in tears. ...
Myoglobin/Hemoglobin O2 Binding and Allosteric
... 1) In active tissues respiration, (glycolysis) results in lactic acid formation. These tissues need more O2. Without the H+ effect Hb would hold on to more of the O2. The increase [H+] induces Hb to dump 10% more of it's O2. 2) CO2 reversibly binds to N term (carbamate) to remove remaining CO2 R - ...
... 1) In active tissues respiration, (glycolysis) results in lactic acid formation. These tissues need more O2. Without the H+ effect Hb would hold on to more of the O2. The increase [H+] induces Hb to dump 10% more of it's O2. 2) CO2 reversibly binds to N term (carbamate) to remove remaining CO2 R - ...
Globular protein slides
... the hemoglobin iron, forming carboxyhemoglobin. • When carbon monoxide binds to one or more of the four heme sites, hemoglobin shifts to the relaxed conformation (R-form), causing the remaining heme sites to bind oxygen with high affinity. • As a result, the affected hemoglobin is unable to release ...
... the hemoglobin iron, forming carboxyhemoglobin. • When carbon monoxide binds to one or more of the four heme sites, hemoglobin shifts to the relaxed conformation (R-form), causing the remaining heme sites to bind oxygen with high affinity. • As a result, the affected hemoglobin is unable to release ...
Option H. Gaseous Exchange
... gas that dissolves in the water is proportional to its partial pressure in the air and its solubility in water. An equilibrium is reach when gas molecules enter and leave the solution at the same rate At this point the gas is said to have the same partial pressure in solution as it does in the ...
... gas that dissolves in the water is proportional to its partial pressure in the air and its solubility in water. An equilibrium is reach when gas molecules enter and leave the solution at the same rate At this point the gas is said to have the same partial pressure in solution as it does in the ...
Paper
... When an oxygen molecule binds to hemoglobin, a conformational change occurs in the shape of hemoglobin, and makes it much more likely to bind oxygen. This phenomenon is called cooperative binding. The importance of the sigmoidal curve is that when the PO2 is in range between 25 and 50 mmHg, a relat ...
... When an oxygen molecule binds to hemoglobin, a conformational change occurs in the shape of hemoglobin, and makes it much more likely to bind oxygen. This phenomenon is called cooperative binding. The importance of the sigmoidal curve is that when the PO2 is in range between 25 and 50 mmHg, a relat ...
hemogram? - HealthEast
... Red blood cells carry oxygen to all parts of the body. A decrease in red blood cells may cause anemia, fatigue and shortness of breath. Hemoglobin Hemoglobin is a protein that helps the RBCs carry the oxygen. ...
... Red blood cells carry oxygen to all parts of the body. A decrease in red blood cells may cause anemia, fatigue and shortness of breath. Hemoglobin Hemoglobin is a protein that helps the RBCs carry the oxygen. ...
Hemoglobin and Iron 4 6
... nuts and grain products. When you eat the two together, the non-heme iron is more easily absorbed. Foods high in vitamin C, like tomatoes, citrus fruits and red, yellow and orange peppers, can also help with the absorption of non-heme iron. ...
... nuts and grain products. When you eat the two together, the non-heme iron is more easily absorbed. Foods high in vitamin C, like tomatoes, citrus fruits and red, yellow and orange peppers, can also help with the absorption of non-heme iron. ...
Molecule of the Month on Hemoglobin
... regulate its action. Oxygen binding at the four heme sites in hemoglobin does not happen simultaneously. Once the first heme binds oxygen, it introduces small changes in the structure of the corresponding protein chain. These changes nudge the neighboring chains into a different shape, making them b ...
... regulate its action. Oxygen binding at the four heme sites in hemoglobin does not happen simultaneously. Once the first heme binds oxygen, it introduces small changes in the structure of the corresponding protein chain. These changes nudge the neighboring chains into a different shape, making them b ...
Physiology Objectives 43
... Haldane effect: as PO2 goes up, the CO2 dissociation curve shifts downward, meaning that less CO2 is carried in the blood Role of carbonic anhydrase in the chloride shift: carbonic anhydrase catalyzes the conversion of carbon H2CO2 to H+ and HCO3-; in the tissue, where HCO3- is being taken up, H+ is ...
... Haldane effect: as PO2 goes up, the CO2 dissociation curve shifts downward, meaning that less CO2 is carried in the blood Role of carbonic anhydrase in the chloride shift: carbonic anhydrase catalyzes the conversion of carbon H2CO2 to H+ and HCO3-; in the tissue, where HCO3- is being taken up, H+ is ...
Molecule of the Month extension
... penetrates more deeply. The dark blood in the vein absorbs most of this red light (as well as any blue light that makes it in that far), so what we see is the blue light that is reflected at the skin's surface. Some organisms like snails and crabs, on the other hand, use copper to transport oxygen, ...
... penetrates more deeply. The dark blood in the vein absorbs most of this red light (as well as any blue light that makes it in that far), so what we see is the blue light that is reflected at the skin's surface. Some organisms like snails and crabs, on the other hand, use copper to transport oxygen, ...
Transport of Oxygen
... On this curve, the x axis lists the PO2 of the blood (whether it is in the lungs or other body tissues), and the y axis lists the percent saturation of the hemoglobin with oxygen. This graph has a sigmoidal ‘S’ shape, such that changes in partial pressures above 80 mmHg do not have a major effect o ...
... On this curve, the x axis lists the PO2 of the blood (whether it is in the lungs or other body tissues), and the y axis lists the percent saturation of the hemoglobin with oxygen. This graph has a sigmoidal ‘S’ shape, such that changes in partial pressures above 80 mmHg do not have a major effect o ...
Hemoglobin

Hemoglobin (/ˈhiːmɵˌɡloʊbɨn/); also spelled haemoglobin and abbreviated Hb or Hgb, is the iron-containing oxygen-transport metalloprotein in the red blood cells of all vertebrates (with the exception of the fish family Channichthyidae) as well as the tissues of some invertebrates. Hemoglobin in the blood carries oxygen from the respiratory organs (lungs or gills) to the rest of the body (i.e. the tissues). There it releases the oxygen to permit aerobic respiration to provide energy to power the functions of the organism in the process called metabolism.In mammals, the protein makes up about 96% of the red blood cells' dry content (by weight), and around 35% of the total content (including water). Hemoglobin has an oxygen-binding capacity of 1.34 mL O2 per gram, which increases the total blood oxygen capacity seventy-fold compared to dissolved oxygen in blood. The mammalian hemoglobin molecule can bind (carry) up to four oxygen molecules.Hemoglobin is involved in the transport of other gases: It carries some of the body's respiratory carbon dioxide (about 10% of the total) as carbaminohemoglobin, in which CO2 is bound to the globin protein. The molecule also carries the important regulatory molecule nitric oxide bound to a globin protein thiol group, releasing it at the same time as oxygen.Hemoglobin is also found outside red blood cells and their progenitor lines. Other cells that contain hemoglobin include the A9 dopaminergic neurons in the substantia nigra, macrophages, alveolar cells, and mesangial cells in the kidney. In these tissues, hemoglobin has a non-oxygen-carrying function as an antioxidant and a regulator of iron metabolism.Hemoglobin and hemoglobin-like molecules are also found in many invertebrates, fungi, and plants. In these organisms, hemoglobins may carry oxygen, or they may act to transport and regulate other things such as carbon dioxide, nitric oxide, hydrogen sulfide and sulfide. A variant of the molecule, called leghemoglobin, is used to scavenge oxygen away from anaerobic systems, such as the nitrogen-fixing nodules of leguminous plants, before the oxygen can poison the system.