Atoms, Elements, and Compounds
... graduate research under the guidance of the man who discovered the electron, J. J. Thomson. Through his research with Thomson, Rutherford became interested in studying radioactivity. In 1898 he described two kinds of particles emitted from radioactive atoms, calling them alpha and beta particles. He ...
... graduate research under the guidance of the man who discovered the electron, J. J. Thomson. Through his research with Thomson, Rutherford became interested in studying radioactivity. In 1898 he described two kinds of particles emitted from radioactive atoms, calling them alpha and beta particles. He ...
Masses of Atoms
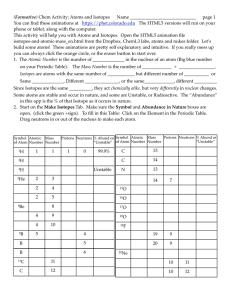
... 80% of Boron in nature have 5 protons, 6 neutrons ~ 11 amu 20% of Boron in nature have 5 protons, 5 neutrons ~ 10 amu (.8 • 11 amu) + (.2 • 10 amu) = 8.8 amu + 2 amu = 10.8 amu There are a few extra isotopes out there that we did not include. ...
... 80% of Boron in nature have 5 protons, 6 neutrons ~ 11 amu 20% of Boron in nature have 5 protons, 5 neutrons ~ 10 amu (.8 • 11 amu) + (.2 • 10 amu) = 8.8 amu + 2 amu = 10.8 amu There are a few extra isotopes out there that we did not include. ...
ch03 - earthjay science
... geochronology (29): The study of time as applied to Earth and planetary history. half-life (39): The time in which one-half of an original amount of a radioactive atoms decays to daughter products. Holocene Series (34): A term sometimes used to designate the period of time since the last major episo ...
... geochronology (29): The study of time as applied to Earth and planetary history. half-life (39): The time in which one-half of an original amount of a radioactive atoms decays to daughter products. Holocene Series (34): A term sometimes used to designate the period of time since the last major episo ...
Physical Science Chapter 1
... 5. The elements located on the stair-step dividing line are metalloids. ...
... 5. The elements located on the stair-step dividing line are metalloids. ...
Build An Atom - ChemConnections
... Neutral atoms have ________________________________ protons and electrons. Positive ions have ________________________________ protons than electrons. Negative ions have _______________________________ protons than electrons. ...
... Neutral atoms have ________________________________ protons and electrons. Positive ions have ________________________________ protons than electrons. Negative ions have _______________________________ protons than electrons. ...
Chapter 3—Time and Geology
... geochronology (27): The study of time as applied to Earth and planetary history. half-life (38): The time in which one-half of an original amount of a radioactive atoms decays to daughter products. Holocene Series (32): A term sometimes used to designate the period of time since the last major episo ...
... geochronology (27): The study of time as applied to Earth and planetary history. half-life (38): The time in which one-half of an original amount of a radioactive atoms decays to daughter products. Holocene Series (32): A term sometimes used to designate the period of time since the last major episo ...
Topic 2 Microscopic World I
... of the two statements is true or false; if both are true, then decide whether or not the second statement is a correct explanation of the first statement. Then select one option from A to D according to the following table : ...
... of the two statements is true or false; if both are true, then decide whether or not the second statement is a correct explanation of the first statement. Then select one option from A to D according to the following table : ...
Sub Unit Plan 1 Chem Periodic Table
... II.1 The placement or location of elements on the Periodic Table gives an indication of physical and chemical properties of that element. The elements on the Periodic Table are arranged in order of increasing atomic number. (3.1y) II.2 The number of protons in an atom (atomic number) identifies the ...
... II.1 The placement or location of elements on the Periodic Table gives an indication of physical and chemical properties of that element. The elements on the Periodic Table are arranged in order of increasing atomic number. (3.1y) II.2 The number of protons in an atom (atomic number) identifies the ...
1b Atomic Structure
... the non-metals of the periodic table (with the exception of the noble gasses of Group 18) form negatively charged ions. Atoms can also become positively charged ions. This is typical of the metallic elements. Sodium (Na), for example has a single electron in its outermost (second) shell. In order to ...
... the non-metals of the periodic table (with the exception of the noble gasses of Group 18) form negatively charged ions. Atoms can also become positively charged ions. This is typical of the metallic elements. Sodium (Na), for example has a single electron in its outermost (second) shell. In order to ...
Atoms and nukes packet 2016
... But objects of different ages need isotopes with different length half lives. After about 6 half lives, pretty much all of the original is decayed, and the technique isn’t useful. Or, at the very start, not enough has decayed to give an accurate result. Can you select the correct isotope to use to d ...
... But objects of different ages need isotopes with different length half lives. After about 6 half lives, pretty much all of the original is decayed, and the technique isn’t useful. Or, at the very start, not enough has decayed to give an accurate result. Can you select the correct isotope to use to d ...
Reading 2.1 A Return to Isotopes
... Notice that because the lithium atom always has protons, the atomic number for lithium is always Z = 3. The mass number, however, is A = 6 for the isotope with 3 neutrons, and A = 7 for the isotope with 4 neutrons. In nature, only certain isotopes exist. For instance, stable lithium exists as an iso ...
... Notice that because the lithium atom always has protons, the atomic number for lithium is always Z = 3. The mass number, however, is A = 6 for the isotope with 3 neutrons, and A = 7 for the isotope with 4 neutrons. In nature, only certain isotopes exist. For instance, stable lithium exists as an iso ...
The Periodic Table of the Elements
... Page 104 . . . . # 4.7 through #4.12 Page 108 . . . . # 4.15 - #4.16 Page 111 . . . . # 4.23 - #4.30 Page 114 . . . . # 4.31 - #4.34 Page 115 . . . . # 4.35 - #4.36 Page 119 . . . . # 4.43 - #4.46 Page 130 . . . . # 4.87 - #4.90 Page 131 . . . . # 4.91 - #4.100 ...
... Page 104 . . . . # 4.7 through #4.12 Page 108 . . . . # 4.15 - #4.16 Page 111 . . . . # 4.23 - #4.30 Page 114 . . . . # 4.31 - #4.34 Page 115 . . . . # 4.35 - #4.36 Page 119 . . . . # 4.43 - #4.46 Page 130 . . . . # 4.87 - #4.90 Page 131 . . . . # 4.91 - #4.100 ...
Radiometric Dating - Tulane University
... Prior to 1905 the best and most accepted age of the Earth was that proposed by Lord Kelvin based on the amount of time necessary for the Earth to cool to its present temperature from a completely liquid state. Although we now recognize lots of problems with that calculation, the age of 25 my was acc ...
... Prior to 1905 the best and most accepted age of the Earth was that proposed by Lord Kelvin based on the amount of time necessary for the Earth to cool to its present temperature from a completely liquid state. Although we now recognize lots of problems with that calculation, the age of 25 my was acc ...
Atomic structure and periodic table review questions What is an
... 3. Another name for the two sub-atomic particles found in the nucleus is ____________. 4. What are the sub-atomic particles found outside of the nucleus? 5. Which particle has a positive charge? 6. Which particle has a neutral charge? 7. Which particle has a negative charge? 8. An element’s atomic n ...
... 3. Another name for the two sub-atomic particles found in the nucleus is ____________. 4. What are the sub-atomic particles found outside of the nucleus? 5. Which particle has a positive charge? 6. Which particle has a neutral charge? 7. Which particle has a negative charge? 8. An element’s atomic n ...
Introduction to Atoms
... • Atoms are the building blocks of all materials • An atom is made of 3 parts: – Protons and Neutrons are in the nucleus (center) – Electrons orbit around the nucleus ...
... • Atoms are the building blocks of all materials • An atom is made of 3 parts: – Protons and Neutrons are in the nucleus (center) – Electrons orbit around the nucleus ...
Henry Moseley, the Atomic Number, and Synthesis
... Introduce Henry Moseley who was a researcher at Rutherford’s laboratory. In 1913 Moseley used Rutherford’s work to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alig ...
... Introduce Henry Moseley who was a researcher at Rutherford’s laboratory. In 1913 Moseley used Rutherford’s work to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alig ...
Henry Moseley, the Atomic Number, and Synthesis
... Introduce Henry Moseley who was a researcher at Rutherford’s laboratory. In 1913 Moseley used Rutherford’s work to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alig ...
... Introduce Henry Moseley who was a researcher at Rutherford’s laboratory. In 1913 Moseley used Rutherford’s work to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alig ...
Lesson 1 & 2 Periodic table trends and formation
... An element is a substance made of only one kind of atom. 2. What is the atomic mass of an element? The atomic mass is the mass of an atom of a particular element. It is the total number of protons and neutrons in the nucleus of an atom of a particular element, averaged over all the isotopes of the e ...
... An element is a substance made of only one kind of atom. 2. What is the atomic mass of an element? The atomic mass is the mass of an atom of a particular element. It is the total number of protons and neutrons in the nucleus of an atom of a particular element, averaged over all the isotopes of the e ...
OCR AS LEVEL CHEMISTRY A 1.1.1 ATOMS 1.2.1 ELECTRON
... The first ionisation energies of the elements Li to Na are shown in the figure below. ...
... The first ionisation energies of the elements Li to Na are shown in the figure below. ...
Properties of Atoms and the Periodic Table 16
... Not all atoms of an element have the same number of neutrons. Atoms of the same element that have different numbers of neutrons are called isotopes. For example, boron atoms can have mass numbers of 10 or 11. To find the number of neutrons in an isotope, you can use the formula above. Look at the ta ...
... Not all atoms of an element have the same number of neutrons. Atoms of the same element that have different numbers of neutrons are called isotopes. For example, boron atoms can have mass numbers of 10 or 11. To find the number of neutrons in an isotope, you can use the formula above. Look at the ta ...
mystery elements
... Who discovered the mass of an electron? __________________________ Who discovered the nucleus? _________________________________ What are the forces called in the nucleus that hold the protons and neutrons together, even though like charges should repel? ______________________________ Define atomic ...
... Who discovered the mass of an electron? __________________________ Who discovered the nucleus? _________________________________ What are the forces called in the nucleus that hold the protons and neutrons together, even though like charges should repel? ______________________________ Define atomic ...
atom - geraldinescience
... • Atoms that contain too many or two few neutrons are unstable and lose energy through radioactive decay to form a stable nucleus. • Few exist in nature—most have already decayed to stable forms. ...
... • Atoms that contain too many or two few neutrons are unstable and lose energy through radioactive decay to form a stable nucleus. • Few exist in nature—most have already decayed to stable forms. ...
An atom is the small unit of which all matter is made. It consists of
... the electrons. Therefore, almost all the mass of an atom is in its nucleus. Each proton in an atom’s nucleus has a positive electric charge, and each electron circling around the nucleus has a negative charge. The neutrons carry no charge at all – that is, they are electrically neutral. When an atom ...
... the electrons. Therefore, almost all the mass of an atom is in its nucleus. Each proton in an atom’s nucleus has a positive electric charge, and each electron circling around the nucleus has a negative charge. The neutrons carry no charge at all – that is, they are electrically neutral. When an atom ...
Chapter 4, Lesson 2: The Periodic Table
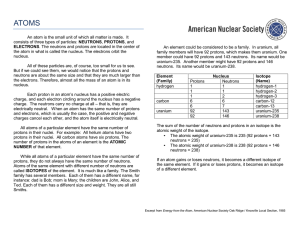
... The atomic mass of an element is based on the mass of the protons, neutrons, and electrons of the atoms of that element. The mass of the proton and neutron are about the same, but the mass of the electron is much smaller (about 1/2000 the mass of the proton or neutron). The majority of the atomic ma ...
... The atomic mass of an element is based on the mass of the protons, neutrons, and electrons of the atoms of that element. The mass of the proton and neutron are about the same, but the mass of the electron is much smaller (about 1/2000 the mass of the proton or neutron). The majority of the atomic ma ...
Chapter 4, Lesson 2: The Periodic Table
... Each student should find and present some basic information about their element to the class. The presentation can be in the form of a poster, pamphlet, PowerPoint presentation or other form. The presentations should be short and can include: atom name, atomic number, derivation of name, when and wh ...
... Each student should find and present some basic information about their element to the class. The presentation can be in the form of a poster, pamphlet, PowerPoint presentation or other form. The presentations should be short and can include: atom name, atomic number, derivation of name, when and wh ...