Practice Test 1 (Chapters 1-7)
... number in front of the substance in bold type? NBr3 + NaOH N2 + NaBr + HOBr a. b. c. d. e. ...
... number in front of the substance in bold type? NBr3 + NaOH N2 + NaBr + HOBr a. b. c. d. e. ...
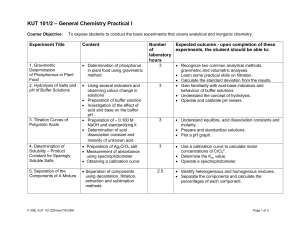
KUT 101/2 – General Chemistry Practical I
... • Determination of acid dissociation constant and molarity of unknown acid • Preparation of Ag2CrO4 salt • Measurement of absorbance using spectrophotometer • Obtaining a calibration curve ...
... • Determination of acid dissociation constant and molarity of unknown acid • Preparation of Ag2CrO4 salt • Measurement of absorbance using spectrophotometer • Obtaining a calibration curve ...
students - Teach-n-Learn-Chem
... Polymers and Monomers polymer: a large molecule (often a chain) made of many smaller molecules called monomers Polymers can be made more rigid if the chains are linked together by way of a crosslinking agent. ...
... Polymers and Monomers polymer: a large molecule (often a chain) made of many smaller molecules called monomers Polymers can be made more rigid if the chains are linked together by way of a crosslinking agent. ...
LAB – Observing Changes in Matter
... In this lab, you will carry out 6 different experiments and record the information into the data table found in the Data/Observations section. Record the properties of the reactants before doing the reaction. Remember, safety first! There is acid being used. Safety goggles and lab aprons are a must! ...
... In this lab, you will carry out 6 different experiments and record the information into the data table found in the Data/Observations section. Record the properties of the reactants before doing the reaction. Remember, safety first! There is acid being used. Safety goggles and lab aprons are a must! ...
Unit A Remediation Review
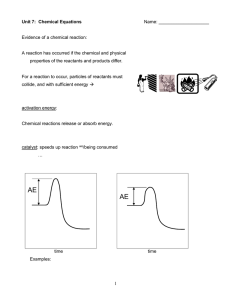
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
Hearthburn wonder labs balanced equations copy
... 2) Count the number of atoms on each side of the equation, keeping subscripts and parentheses into account 3) To balance the number of atoms on each side of the equation (LAW OF CONSERVATION OF MATTER!) we will add coefficients to compounds where we need more of a certain element. ...
... 2) Count the number of atoms on each side of the equation, keeping subscripts and parentheses into account 3) To balance the number of atoms on each side of the equation (LAW OF CONSERVATION OF MATTER!) we will add coefficients to compounds where we need more of a certain element. ...
Chemical Identity and Synonyms Physical Chemical Properties
... this chemical to employees, stakeholders, and the environment. This chemical is not released to the air. The packaged products are warehoused and any spills in the warehouse that are not immediately contained, naturally drain to our process water waste treatment. The spilled material is then segrega ...
... this chemical to employees, stakeholders, and the environment. This chemical is not released to the air. The packaged products are warehoused and any spills in the warehouse that are not immediately contained, naturally drain to our process water waste treatment. The spilled material is then segrega ...
1.5.16(Chem) - mrcarlsonschemistryclass
... Cations and Anions • Cations are ions with a POSITIVE charge. • Anions are ions with a NEGATIVE charge. • Draw the funny way to remember cations and anions: ...
... Cations and Anions • Cations are ions with a POSITIVE charge. • Anions are ions with a NEGATIVE charge. • Draw the funny way to remember cations and anions: ...
Sodium Nitrate Specifications
... Synonym : Nitrate of Soda; Nitric acid sodium salt (1:1); Nitric acid, sodium salt Formula : NaNO3 Class : Inorganic Chemicals: Nitrates, Phosphates, Oxides & more Drilling and Completion Fluids Industrial Chemicals Product Type : Hazard : 5.1 Oxidizer ...
... Synonym : Nitrate of Soda; Nitric acid sodium salt (1:1); Nitric acid, sodium salt Formula : NaNO3 Class : Inorganic Chemicals: Nitrates, Phosphates, Oxides & more Drilling and Completion Fluids Industrial Chemicals Product Type : Hazard : 5.1 Oxidizer ...
Chemical Reaction Powered Car
... • Department web site: www.cheg.uark.edu • My research web site: www.comp.uark.edu/~sservoss ...
... • Department web site: www.cheg.uark.edu • My research web site: www.comp.uark.edu/~sservoss ...
Sodium bicarbonate

Sodium bicarbonate (IUPAC name: sodium hydrogen carbonate) is a chemical compound with the formula NaHCO3. Sodium bicarbonate is a white solid that is crystalline but often appears as a fine powder. It has a slightly salty, alkaline taste resembling that of washing soda (sodium carbonate). The natural mineral form is nahcolite. It is a component of the mineral natron and is found dissolved in many mineral springs.It is among the food additives encoded by European Union, identified by the initials E 500.Since it has long been known and is widely used, the salt has many related names such as baking soda, bread soda, cooking soda, and bicarbonate of soda. The word saleratus, from Latin sal æratus meaning aerated salt, was widely used in the 19th century for both sodium bicarbonate and potassium bicarbonate.