10B Atoms and Isotopes
... This is because you add one proton at a time for each element. The atomic mass however, increases by amounts greater than one. This difference is due to the neutrons in the nucleus. The value of the atomic mass reflects the abundance of the stable isotopes for an element that exist in the universe. ...
... This is because you add one proton at a time for each element. The atomic mass however, increases by amounts greater than one. This difference is due to the neutrons in the nucleus. The value of the atomic mass reflects the abundance of the stable isotopes for an element that exist in the universe. ...
Isotopes, Ions Worksheet
... b) Do different isotopes have different half-lifes (t ½ )? YES Different isotopes have a different neutron number which results in different half-life 21. List THREE Nuclear Applications 1. _______________________ 2. ______________________ 3. _______________________ 22. Why is it important to know t ...
... b) Do different isotopes have different half-lifes (t ½ )? YES Different isotopes have a different neutron number which results in different half-life 21. List THREE Nuclear Applications 1. _______________________ 2. ______________________ 3. _______________________ 22. Why is it important to know t ...
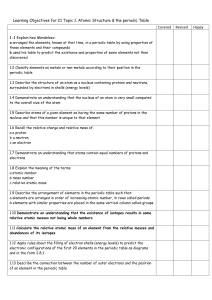
C2- Topic 1: Atomic structure and the periodic table. Assessable
... - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
... - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
Atomic Number, Mass Number, and Isotopes
... have the same number of neutrons. Isotopes are atoms of the same element that have different masses (resulting from the different numbers of neutrons). An analogy is the flavors of ice cream (even ones to which no specific flavor is added). Identifying an isotope requires knowing both the atomic num ...
... have the same number of neutrons. Isotopes are atoms of the same element that have different masses (resulting from the different numbers of neutrons). An analogy is the flavors of ice cream (even ones to which no specific flavor is added). Identifying an isotope requires knowing both the atomic num ...
Vocabulary for Periodic Table
... 12) Period: a horizontal row in the periodic table of elements that have varying properties. 13) Reactive: indicates how likely an element is to undergo a chemical change. 14) Metal: an element that tends to be shiny, easily shaped, and a good conductor of electricity and heat. 15) Nonmetal: an elem ...
... 12) Period: a horizontal row in the periodic table of elements that have varying properties. 13) Reactive: indicates how likely an element is to undergo a chemical change. 14) Metal: an element that tends to be shiny, easily shaped, and a good conductor of electricity and heat. 15) Nonmetal: an elem ...
Honors Review Unit 2 answers
... Conducted experiments with cathode rays. ____Thomson_________ Know the 5 points of Dalton’s atomic theory and how two of those points have been changed! 1. All matter is made of atoms. 2. All atoms of the same element are exactly the same. Atoms of different elements are different. CHANGE – ATOMS OF ...
... Conducted experiments with cathode rays. ____Thomson_________ Know the 5 points of Dalton’s atomic theory and how two of those points have been changed! 1. All matter is made of atoms. 2. All atoms of the same element are exactly the same. Atoms of different elements are different. CHANGE – ATOMS OF ...
Practice Test #2 - smhs
... for Ag as 107.868 amu. Find the percent abundance of the lighter isotope of Ag. 107.868 = 106.9041 (X) + 108.9047 (1.000 - X) -1.0367 = -2.0006 X X = 0.51819 ...
... for Ag as 107.868 amu. Find the percent abundance of the lighter isotope of Ag. 107.868 = 106.9041 (X) + 108.9047 (1.000 - X) -1.0367 = -2.0006 X X = 0.51819 ...
Periodic Table Vocabulary Periodic Table – a chart that organizes
... Inert – elements and/or compounds that when put together are unable to react chemically. The Law of Conservation of Matter – a scientific law that states that during a chemical reaction, matter cannot be created or destroyed but can be changed into a different form. Period law- The chemical properti ...
... Inert – elements and/or compounds that when put together are unable to react chemically. The Law of Conservation of Matter – a scientific law that states that during a chemical reaction, matter cannot be created or destroyed but can be changed into a different form. Period law- The chemical properti ...
Lesson Outline - WordPress.com
... up 80.22%. What is the average atomic mass of Boron? 2. Silver has two isotopes, the first has a mass of 106.9 u and an abundance of 51.8%, the second has a mass of 108.9 u and an abundance of 48.2. What is the average atomic mass of ...
... up 80.22%. What is the average atomic mass of Boron? 2. Silver has two isotopes, the first has a mass of 106.9 u and an abundance of 51.8%, the second has a mass of 108.9 u and an abundance of 48.2. What is the average atomic mass of ...
Practice Test #2 - smhs
... Answer the following questions based on the -2 anion of an isotopic form of sulfur: S-35 24.________ What is the A number for this nuclide? 25.________ What is the Z number for this nuclide? 26.________ What is the number of protons in the anion form of this nonmetal? 27.________ What is the number ...
... Answer the following questions based on the -2 anion of an isotopic form of sulfur: S-35 24.________ What is the A number for this nuclide? 25.________ What is the Z number for this nuclide? 26.________ What is the number of protons in the anion form of this nonmetal? 27.________ What is the number ...
The Nuclear Atom
... Democritus (460 B.C. – 370 B.C.) • first to suggest the existence of “atoms” ...
... Democritus (460 B.C. – 370 B.C.) • first to suggest the existence of “atoms” ...
answers
... c.) Rutherford – discovered positively charged nucleus d.) Bohr – solar system model of atoms, energy levels at increasing distance from nucleus ...
... c.) Rutherford – discovered positively charged nucleus d.) Bohr – solar system model of atoms, energy levels at increasing distance from nucleus ...
PP - myndrs.com
... be made or destroyed All atoms of the same element are identical Different elements have different types of atoms Chemical reactions occur when atoms are rearranged Compounds are formed from atoms of the different elements coming together. ...
... be made or destroyed All atoms of the same element are identical Different elements have different types of atoms Chemical reactions occur when atoms are rearranged Compounds are formed from atoms of the different elements coming together. ...
Structure of an Atom structure_of_atom
... be made or destroyed All atoms of the same element are identical Different elements have different types of atoms Chemical reactions occur when atoms are rearranged Compounds are formed from atoms of the different elements coming together. ...
... be made or destroyed All atoms of the same element are identical Different elements have different types of atoms Chemical reactions occur when atoms are rearranged Compounds are formed from atoms of the different elements coming together. ...
Exam III Review
... 15. Rutherford’s gold foil experiment indicated that a. The nucleus of an atom occupies most of an atom’s volume. b. Positive charges are dispersed throughout the atom. c. Positive charges are concentrated in a very small core at the atom’s center. d. Protons and neutrons are located in the nucleus. ...
... 15. Rutherford’s gold foil experiment indicated that a. The nucleus of an atom occupies most of an atom’s volume. b. Positive charges are dispersed throughout the atom. c. Positive charges are concentrated in a very small core at the atom’s center. d. Protons and neutrons are located in the nucleus. ...
Chapter 2
... • Nonmetals – right side; poor conductors; gain electrons (- ions) • Metalloids – stair-step line • Groups – columns; grouped by similar properties • 18 – each has a name ...
... • Nonmetals – right side; poor conductors; gain electrons (- ions) • Metalloids – stair-step line • Groups – columns; grouped by similar properties • 18 – each has a name ...
Lawrencium

Lawrencium is a synthetic chemical element with chemical symbol Lr (formerly Lw) and atomic number 103. It is named in honor of Ernest Lawrence, inventor of the cyclotron, a device that was used to discover many artificial radioactive elements. A radioactive metal, lawrencium is the eleventh transuranic element and is also the final member of the actinide series. Like all elements with atomic number over 100, lawrencium can only be produced in particle accelerators by bombarding lighter elements with charged particles. Twelve isotopes of lawrencium are currently known; the most stable is 266Lr with a half-life of 11 hours, but the shorter-lived 260Lr (half-life 2.7 minutes) is most commonly used in chemistry because it can be produced on a larger scale.Chemistry experiments have confirmed that lawrencium indeed behaves as a heavier homolog to lutetium in the periodic table, and is a trivalent element. It thus could also be classified as the first of the 7th-period transition metals: however, its electron configuration is anomalous for its position in the periodic table, having an s2p configuration instead of the s2d configuration of its homolog lutetium. This means that lawrencium may be less volatile than expected for its position in the periodic table and have a volatility comparable to that of lead.In the 1950s, 1960s, and 1970s, many claims of the synthesis of lawrencium of varying quality were made from laboratories in the Soviet Union and the United States. The priority of the discovery and therefore the naming of the element was disputed between Soviet and American scientists, and while the International Union of Pure and Applied Chemistry (IUPAC) established lawrencium as the official name for the element and gave the American team credit for the discovery, this was reevaluated in 1997, giving both teams shared credit for the discovery but not changing the element's name.