Chemistry Part - teko classes bhopal
... Within each layer of graphite, every carbon atom is joined to three others by strong covalent bonds. This forms a pattern of interlocking hexagonal rings. The carbon atoms are difficult to separate from one another. So graphite also has high melting point. However, the bonds between the layers are w ...
... Within each layer of graphite, every carbon atom is joined to three others by strong covalent bonds. This forms a pattern of interlocking hexagonal rings. The carbon atoms are difficult to separate from one another. So graphite also has high melting point. However, the bonds between the layers are w ...
Unit Six: Atomic structure
... • Nonmetals • Metalloids a. Metals are located on the left side of the periodic table b. Non metals are located on the right side of the periodic table. c. The six metalloids fall along the “steps”. They include: Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), Tellurium (Te) ...
... • Nonmetals • Metalloids a. Metals are located on the left side of the periodic table b. Non metals are located on the right side of the periodic table. c. The six metalloids fall along the “steps”. They include: Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), Tellurium (Te) ...
Discovering Elements
... From the late Middle Ages onwards, new elements had been discovered from time to time, sometimes during the investigation of metal ores, sometimes almost by accident, as in the case of Hennig Brand’s discovery of phosphorus. In the 19th century more sophisticated techniques, such as spectroscopy and ...
... From the late Middle Ages onwards, new elements had been discovered from time to time, sometimes during the investigation of metal ores, sometimes almost by accident, as in the case of Hennig Brand’s discovery of phosphorus. In the 19th century more sophisticated techniques, such as spectroscopy and ...
General Structure of the Periodic Table
... In the sixty-seven years from Dalton's formulation of atomic weight to Mendeleyev's periodic table many scientists had tried to create a working organizational structure for the elements. Mendeleyev succeeded where others failed because he realized that there existed a number of as yet unknown eleme ...
... In the sixty-seven years from Dalton's formulation of atomic weight to Mendeleyev's periodic table many scientists had tried to create a working organizational structure for the elements. Mendeleyev succeeded where others failed because he realized that there existed a number of as yet unknown eleme ...
Lesson 7.8 Basic Properties of the Main Group Elements Suggested
... We have already mentioned the distinct trend from nonmetal to metal going down the Group IVA elements. Carbon Exists in two well-known allotropic forms: graphite, a soft, black substance used in pencil leads and diamond, a very hard, clear, crystalline substance. Another allotrope a carbon is a mole ...
... We have already mentioned the distinct trend from nonmetal to metal going down the Group IVA elements. Carbon Exists in two well-known allotropic forms: graphite, a soft, black substance used in pencil leads and diamond, a very hard, clear, crystalline substance. Another allotrope a carbon is a mole ...
Full Chapter - CPO Science
... Remember, the atomic number is the number of protons all atoms of that element have in their nuclei. If the atom is neutral, it will have the same number of electrons as protons. ...
... Remember, the atomic number is the number of protons all atoms of that element have in their nuclei. If the atom is neutral, it will have the same number of electrons as protons. ...
Periodic Table - Marian High School
... Remember, the atomic number is the number of protons all atoms of that element have in their nuclei. If the atom is neutral, it will have the same number of electrons as protons. ...
... Remember, the atomic number is the number of protons all atoms of that element have in their nuclei. If the atom is neutral, it will have the same number of electrons as protons. ...
Periodic Table
... Earth’s crust. It is found in a mineral called bauxite. Most of our laboratory glassware contains boron, which helps prevent it from shattering when heated. ...
... Earth’s crust. It is found in a mineral called bauxite. Most of our laboratory glassware contains boron, which helps prevent it from shattering when heated. ...
the periodic table
... • - each column of the periodic table (called a group) has similar properties (periodic trends) The Periodic Law - chemical and physical properties of element vary in a periodic way with their atomic number Hands-On: elemental samples (Helen) ...
... • - each column of the periodic table (called a group) has similar properties (periodic trends) The Periodic Law - chemical and physical properties of element vary in a periodic way with their atomic number Hands-On: elemental samples (Helen) ...
The Periodic Table
... Mendeleev was the first scientist to notice the relationship between the elements ...
... Mendeleev was the first scientist to notice the relationship between the elements ...
Elements and the Periodic Table
... 20. What is the atomic number of an element? 21. Looking at the periodic table, which element would have similar chemical properties to Beryllium, Be? 22. Complete the following chart: ...
... 20. What is the atomic number of an element? 21. Looking at the periodic table, which element would have similar chemical properties to Beryllium, Be? 22. Complete the following chart: ...
Elements and the Periodic Table
... 1. Which of the following compounds contains the greatest number of elements? ...
... 1. Which of the following compounds contains the greatest number of elements? ...
Chapter 5 student
... elements in the modern periodic table. • Explain how the atomic mass of an element is determined and how atomic mass units are defined. • Identify general properties of metals, nonmetals, and metalloids. ...
... elements in the modern periodic table. • Explain how the atomic mass of an element is determined and how atomic mass units are defined. • Identify general properties of metals, nonmetals, and metalloids. ...
Chapter 5
... • Atomic mass is a value that depends on the distribution of an element’s isotopes in nature and the masses of ...
... • Atomic mass is a value that depends on the distribution of an element’s isotopes in nature and the masses of ...
E/F Physical Science Learning Targets ALL Name: Date: Hour
... 6. Circle the letter of each sentence that is true about a carbon-12 atom. a. It has 6 protons and 6 neutrons b. Scientists assigned a mass of 6 atomic mass units to the atom c. It is used as a standard for comparing the masses of all atoms d. An atomic mass unit is defined as one twelfth the mass o ...
... 6. Circle the letter of each sentence that is true about a carbon-12 atom. a. It has 6 protons and 6 neutrons b. Scientists assigned a mass of 6 atomic mass units to the atom c. It is used as a standard for comparing the masses of all atoms d. An atomic mass unit is defined as one twelfth the mass o ...
noble gases
... electrons) were later discovered, it became clear that the atomic number (number of protons in the nucleus) held the key to the identity of an element. ...
... electrons) were later discovered, it became clear that the atomic number (number of protons in the nucleus) held the key to the identity of an element. ...
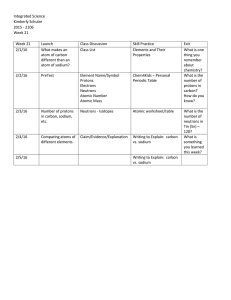
Week 21 Lessons - Highline Public Schools
... - # of protons = Atomic Number - # of electrons = # of protons (this is a neutral atom.) - Mass number = # of protons + # of neutrons - # of neutrons = mass number - # of protons (Mass number must be a whole number!!!! You can’t have half a neutron. Use atomic mass and round up.) Exit Ticket: The nu ...
... - # of protons = Atomic Number - # of electrons = # of protons (this is a neutral atom.) - Mass number = # of protons + # of neutrons - # of neutrons = mass number - # of protons (Mass number must be a whole number!!!! You can’t have half a neutron. Use atomic mass and round up.) Exit Ticket: The nu ...
Chapter 5
... Similarity of the two • Eka-aluminum and Gallium were similar in properties, so they were determined to be the ________ • The close match between Mendeleev’s _________ and the actual properties of new elements showed how ________ his periodic table could be ...
... Similarity of the two • Eka-aluminum and Gallium were similar in properties, so they were determined to be the ________ • The close match between Mendeleev’s _________ and the actual properties of new elements showed how ________ his periodic table could be ...
periodictrendsss - rlsciencecurriculum
... When elements are listed in order by their atomic numbers and grouped according to similar properties, they form seven horizontal rows called periods. Each vertical column in the table contains elements with similar properties. These are called groups or families. The alkali metal family for example ...
... When elements are listed in order by their atomic numbers and grouped according to similar properties, they form seven horizontal rows called periods. Each vertical column in the table contains elements with similar properties. These are called groups or families. The alkali metal family for example ...
Elements of the Periodic Table
... but its high cost has kept it from becoming commercially viable allowing it to compete with nitrous oxide. It is 44% more potent as an anesthetic than nitrous oxide. A very common noble gas, as a matter of fact the second most abundant element in the universe is Helium. Helium represents about 24% o ...
... but its high cost has kept it from becoming commercially viable allowing it to compete with nitrous oxide. It is 44% more potent as an anesthetic than nitrous oxide. A very common noble gas, as a matter of fact the second most abundant element in the universe is Helium. Helium represents about 24% o ...
The Periodic Table
... • Mendeleev wasn’t too far off. • Now the elements are put in rows by increasing ...
... • Mendeleev wasn’t too far off. • Now the elements are put in rows by increasing ...
PART 1: ATOMIC NUMBER - hrsbstaff.ednet.ns.ca
... Using the periodic table, answer the following questions in the spaces provided. 1. Write the name and symbol of the element that is located in: ...
... Using the periodic table, answer the following questions in the spaces provided. 1. Write the name and symbol of the element that is located in: ...
Who`s in this family?
... Element Profile--Magnesium • Magnesium is the lightest of all structural metals • It is used to build some airplanes • It activates many of the enzymes that speed up processes in the human body • It combines with many other elements to form useful compounds such as, milk of magnesia & ...
... Element Profile--Magnesium • Magnesium is the lightest of all structural metals • It is used to build some airplanes • It activates many of the enzymes that speed up processes in the human body • It combines with many other elements to form useful compounds such as, milk of magnesia & ...