Biogeochemical Cycles
... • Step 3: The nitrates and nitrites are used by plants to make amino acids which are then used to make plant proteins. Assimilation • Step 4: Plants are consumed by other organisms which use the plant amino acids to make their own. ...
... • Step 3: The nitrates and nitrites are used by plants to make amino acids which are then used to make plant proteins. Assimilation • Step 4: Plants are consumed by other organisms which use the plant amino acids to make their own. ...
Chapter27
... having such a limited discussion of life in the Universe was that I thought the subject was still very speculative. For example, at that time, only a little more than 10 years ago, we didn’t yet have any evidence for planetary systems orbiting ordinary stars other than the Sun. A lot has happened in ...
... having such a limited discussion of life in the Universe was that I thought the subject was still very speculative. For example, at that time, only a little more than 10 years ago, we didn’t yet have any evidence for planetary systems orbiting ordinary stars other than the Sun. A lot has happened in ...
File
... produced by vehicles Storing carbon in woodbased by-products (paper, lumber, furniture) Remove carbon through photosynthesis ...
... produced by vehicles Storing carbon in woodbased by-products (paper, lumber, furniture) Remove carbon through photosynthesis ...
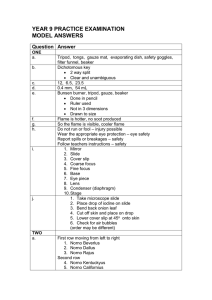
model answers year 9 practice examination
... and electricity, silver in colour, usually solid at room temperature Non-metals Dull, brittle, do not conduct heat or electricity, various states at room temperature, a variety of colours i. water ii. carbon dioxide iii. nitrogen gas iv. sulfuric acid Elements: Gold, sulfur, oxygen, hydrogen Compoun ...
... and electricity, silver in colour, usually solid at room temperature Non-metals Dull, brittle, do not conduct heat or electricity, various states at room temperature, a variety of colours i. water ii. carbon dioxide iii. nitrogen gas iv. sulfuric acid Elements: Gold, sulfur, oxygen, hydrogen Compoun ...
Chemical Basis of Life
... C. To determine the # of neutrons take the element’s ___________________ and subtract its’ _______________________ or number of ___________. If you add or remove neutrons you get ___________________. Why is the atomic mass for many elements not a whole number? ...
... C. To determine the # of neutrons take the element’s ___________________ and subtract its’ _______________________ or number of ___________. If you add or remove neutrons you get ___________________. Why is the atomic mass for many elements not a whole number? ...
Slides from the fourth lecture
... Climate on the Earth The Sun is getting brighter, and was 30% fainter in the beginning. We’d be frozen now without greenhouse gases (and really frozen then). Somehow the greenhouse effect has been regulated to keep liquid water on the surface. In less than a billion years, it will be hard to stop a ...
... Climate on the Earth The Sun is getting brighter, and was 30% fainter in the beginning. We’d be frozen now without greenhouse gases (and really frozen then). Somehow the greenhouse effect has been regulated to keep liquid water on the surface. In less than a billion years, it will be hard to stop a ...
Chemistry of Life PP
... Describe the process by which polymers in living things are formed from smaller molecules. Polymers form when one monomer loses an H+ ion and another loses an OH- to form water. A covalent bond forms between the monomers. ...
... Describe the process by which polymers in living things are formed from smaller molecules. Polymers form when one monomer loses an H+ ion and another loses an OH- to form water. A covalent bond forms between the monomers. ...
Space Review Packet
... 9. Evaluate the habitability of planets and moons g. The accumulation of CO2 in beyond Earth in terms of temperature, water, the atmosphere increases Earth’s atmosphere, nutrients, and energy (TWANE). greenhouse effect and causes ...
... 9. Evaluate the habitability of planets and moons g. The accumulation of CO2 in beyond Earth in terms of temperature, water, the atmosphere increases Earth’s atmosphere, nutrients, and energy (TWANE). greenhouse effect and causes ...
Carbon
... phosphate group Sugar is ribose in RNA, and deoxyribose in DNA (only difference is a single oxygen atom on the 2nd carbon) ...
... phosphate group Sugar is ribose in RNA, and deoxyribose in DNA (only difference is a single oxygen atom on the 2nd carbon) ...
Document
... • Lowell built a large observatory near Flagstaff, AZ (Incidentally, this enabled C. Tombaugh to find Pluto in 1930) ...
... • Lowell built a large observatory near Flagstaff, AZ (Incidentally, this enabled C. Tombaugh to find Pluto in 1930) ...
theory (casual usage of the word) vs. scientific theory
... galaxies are identified in 2D images (right), then have their distance determined from their spectrum to create a 2 billion light-years deep 3D map (left) where each galaxy is shown as a single point, the color representing the luminosity - this shows only those 66,976 our of 205,443 galaxies in th ...
... galaxies are identified in 2D images (right), then have their distance determined from their spectrum to create a 2 billion light-years deep 3D map (left) where each galaxy is shown as a single point, the color representing the luminosity - this shows only those 66,976 our of 205,443 galaxies in th ...
I. INTRODUCTION TO BIOLOGY (pg. 16)
... * ____________________ - come from within an organism (blood glucose level). 6. Living things maintain a __________________ internal environment. This is called _______________________ – “internal harmony”; maintaining a relatively stable internal environment. 7. Living things ____________________. ...
... * ____________________ - come from within an organism (blood glucose level). 6. Living things maintain a __________________ internal environment. This is called _______________________ – “internal harmony”; maintaining a relatively stable internal environment. 7. Living things ____________________. ...
1. For most of human history it was believed that Earth was at the
... 39. The size of our solar system (considered to be the size of Pluto’s orbit) is: 40. If a planet’s perihelion distance is 3 A.U. and aphelion distance is 5 A.U. What is the size of the semi-major axis of the orbit? 41. Approximately how many years would it take light to travel across the length of ...
... 39. The size of our solar system (considered to be the size of Pluto’s orbit) is: 40. If a planet’s perihelion distance is 3 A.U. and aphelion distance is 5 A.U. What is the size of the semi-major axis of the orbit? 41. Approximately how many years would it take light to travel across the length of ...
Matter and Chemical Change Quick Summary
... excessive plant nutrients. The chemical concentration must fall within government guidelines in parts per million. A concentration of 4ppm means 4 parts of chemical in 1,000,000 parts of chemical water solution. -Toxins or poisons are measured with a scale called LD50. LD stands for “lethal dose” an ...
... excessive plant nutrients. The chemical concentration must fall within government guidelines in parts per million. A concentration of 4ppm means 4 parts of chemical in 1,000,000 parts of chemical water solution. -Toxins or poisons are measured with a scale called LD50. LD stands for “lethal dose” an ...
Elements and Compounds power point
... Carbon is in the atmosphere in the form of carbon dioxide Dissolved in all natural water Component of many rocks like limestone Makes up fossil fuels It is found in abundance in the sun, stars, comets, and atmospheres of ...
... Carbon is in the atmosphere in the form of carbon dioxide Dissolved in all natural water Component of many rocks like limestone Makes up fossil fuels It is found in abundance in the sun, stars, comets, and atmospheres of ...
Slide 1
... Carbon is in the atmosphere in the form of carbon dioxide Dissolved in all natural water Component of many rocks like limestone Makes up fossil fuels It is found in abundance in the sun, stars, comets, and atmospheres of ...
... Carbon is in the atmosphere in the form of carbon dioxide Dissolved in all natural water Component of many rocks like limestone Makes up fossil fuels It is found in abundance in the sun, stars, comets, and atmospheres of ...
Water - WordPress.com
... ·It takes energy to break the H bonds and get water molecules to increase in kinetic energy ·e.g. the water in blood keeps your body temperature fairly stable ...
... ·It takes energy to break the H bonds and get water molecules to increase in kinetic energy ·e.g. the water in blood keeps your body temperature fairly stable ...
The Water Cycle - Fall River Public Schools
... Organisms need water, minerals, and other compounds too ...
... Organisms need water, minerals, and other compounds too ...
Molecules of Life
... Composed of CHON (carbon, hydrogen, oxygen, nitrogen) Some types of proteins can contain other elements like S, P, Fe, and Cu (Sulfur, Phosphorous, Iron, Copper) Made of small units (monomers) called ____________ __________. Examples: – Proteins make up muscle, __________ and hair – Hemoglobin (____ ...
... Composed of CHON (carbon, hydrogen, oxygen, nitrogen) Some types of proteins can contain other elements like S, P, Fe, and Cu (Sulfur, Phosphorous, Iron, Copper) Made of small units (monomers) called ____________ __________. Examples: – Proteins make up muscle, __________ and hair – Hemoglobin (____ ...
the Note
... Proteins are organic compound containing carbon, hydrogen, oxygen and nitrogen. Sulphur and phosphorus are sometimes present. A molecule of protein is made up of large number of subunits called amino acids. Proteins are needed for growth and the repair of body tissues. They are also needed for forma ...
... Proteins are organic compound containing carbon, hydrogen, oxygen and nitrogen. Sulphur and phosphorus are sometimes present. A molecule of protein is made up of large number of subunits called amino acids. Proteins are needed for growth and the repair of body tissues. They are also needed for forma ...
Basic Atomic Structure
... -hydroen bonds in ice are more “ordered”, making ice less dense -If ice sank, all bodies of water would eventually freeze solid, making life impossible on Earth. ...
... -hydroen bonds in ice are more “ordered”, making ice less dense -If ice sank, all bodies of water would eventually freeze solid, making life impossible on Earth. ...
File
... Oxygen makes hydrogen peroxide, which is used as an industrial and cosmetic Bleach and disinfectant Is used as a liquid rocket fuel Fluorine: Is added to many materials to lower the melting point. Used to enrich U for use In nuclear power plants. Is added to water supplies and toothpaste to prevent ...
... Oxygen makes hydrogen peroxide, which is used as an industrial and cosmetic Bleach and disinfectant Is used as a liquid rocket fuel Fluorine: Is added to many materials to lower the melting point. Used to enrich U for use In nuclear power plants. Is added to water supplies and toothpaste to prevent ...
File
... substances in a new material with different properties. • By combining the useful properties of two or more substances in a composite, chemists can make a new material that works better than either one alone. • Many composite materials include one or more polymers. ...
... substances in a new material with different properties. • By combining the useful properties of two or more substances in a composite, chemists can make a new material that works better than either one alone. • Many composite materials include one or more polymers. ...
File
... electrons available to form covalent chemical bonds. Carbon is the 15th most abundant elements in the Earth's crust, and the fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. It is present in all known lifeforms, and in the human body carbon is the second most ...
... electrons available to form covalent chemical bonds. Carbon is the 15th most abundant elements in the Earth's crust, and the fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. It is present in all known lifeforms, and in the human body carbon is the second most ...
Hypothetical types of biochemistry

Hypothetical types of biochemistry are forms of biochemistry speculated to be scientifically viable but not proven to exist at this time. The kinds of living beings currently known on Earth all use carbon compounds for basic structural and metabolic functions, water as a solvent and DNA or RNA to define and control their form. If life exists on other planets or moons, it may be chemically similar; it is also possible that there are organisms with quite different chemistries—for instance involving other classes of carbon compounds, compounds of another element, or another solvent in place of water.The possibility of life-forms being based on ""alternative"" biochemistries is the topic of an ongoing scientific discussion, informed by what is known about extraterrestrial environments and about the chemical behaviour of various elements and compounds. It is also a common subject in science fiction.The element silicon has been much discussed as a hypothetical alternative to carbon. Silicon is in the same group as carbon in the periodic table, and like carbon is tetravalent, although the silicon analogs of organic compounds are generally less stable. Hypothetical alternatives to water include ammonia, which, like water, is a polar molecule, and cosmically abundant; and non-polar hydrocarbon solvents such as methane and ethane, which are known to exist in liquid form on the surface of Titan.