Latest Publication (still not complete)
... products. However, in addition to this, the structure and bonding of carbene complexes is also discussed. In particular, an analysis of information gained through computational analysis is provided. Such information has been used to rationalize most of the observed reaction pathways and hence is cru ...
... products. However, in addition to this, the structure and bonding of carbene complexes is also discussed. In particular, an analysis of information gained through computational analysis is provided. Such information has been used to rationalize most of the observed reaction pathways and hence is cru ...
A-Frame Complexes of Dirhodium Bridged by Dicarbene and
... properties like those of trialkylphosphines3 and may actually be better donors than phosphines.4 However, NHC’s are not merely phosphine substitutes; they offer a useful steric alternative to phosphines in having a very different shape. While phosphines are often described as conical,5 NHC ligands a ...
... properties like those of trialkylphosphines3 and may actually be better donors than phosphines.4 However, NHC’s are not merely phosphine substitutes; they offer a useful steric alternative to phosphines in having a very different shape. While phosphines are often described as conical,5 NHC ligands a ...
Helicene-grafted vinyl- and carbene-osmium complexes - HAL
... The first helicene-based carbene osmium complex has been prepared from a vinyl-osmium derivative and this system has been shown to behave as a potential acid-base triggered chiroptical switch. Helicene derivatives have recently shown potential as molecular materials due to their inherent chirality, ...
... The first helicene-based carbene osmium complex has been prepared from a vinyl-osmium derivative and this system has been shown to behave as a potential acid-base triggered chiroptical switch. Helicene derivatives have recently shown potential as molecular materials due to their inherent chirality, ...
Beyond Conventional N-Heterocyclic Carbenes
... canonical form. Clearly, the borderline between the two limiting representations is continuous, and the issue of whether a ligand is, in reality, a carbene or not may become semantic. In the case of the C2-bound imidazolylidenes, experimental and theoretical studies are in agreement with a relativel ...
... canonical form. Clearly, the borderline between the two limiting representations is continuous, and the issue of whether a ligand is, in reality, a carbene or not may become semantic. In the case of the C2-bound imidazolylidenes, experimental and theoretical studies are in agreement with a relativel ...
Get cached
... of both C r ( C N — C H ) (in C H C 1 , v :2070, 2012, 1965 cm" ) and N i ( C N — C H ) (in C H C 1 , *> :2050, 1990 cm" ) show more than one C N — band, is taken as evidence that, in this case at least, the V B approach can afford an adequate explanation of the observations. The I R spectra of Cr(O ...
... of both C r ( C N — C H ) (in C H C 1 , v :2070, 2012, 1965 cm" ) and N i ( C N — C H ) (in C H C 1 , *> :2050, 1990 cm" ) show more than one C N — band, is taken as evidence that, in this case at least, the V B approach can afford an adequate explanation of the observations. The I R spectra of Cr(O ...
Reactions of Alkenes and Alkynes
... carbon radical a) One electron from C=C bond pairs up with electron of benzoyloxy radical to form C-O bond b) Other electron remains on carbon (a carbon-centered radical) ...
... carbon radical a) One electron from C=C bond pairs up with electron of benzoyloxy radical to form C-O bond b) Other electron remains on carbon (a carbon-centered radical) ...
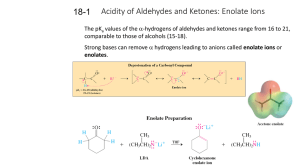
18-1 Enolates (PPT)
... The pKa values of the -hydrogens of aldehydes and ketones range from 16 to 21, comparable to those of alcohols (15-18). ...
... The pKa values of the -hydrogens of aldehydes and ketones range from 16 to 21, comparable to those of alcohols (15-18). ...
- White Rose Research Online
... predicts that the reaction between palladium acetate and one equivalent of imidazolium 1 is exergonic (Figure 4), with the most stable intermediate being 13. However, DFT calculations indicate that addition of the second equivalent of 1 occurs at 15, as the activation energy is less favourable start ...
... predicts that the reaction between palladium acetate and one equivalent of imidazolium 1 is exergonic (Figure 4), with the most stable intermediate being 13. However, DFT calculations indicate that addition of the second equivalent of 1 occurs at 15, as the activation energy is less favourable start ...
© Ravi Divakaran THE CHELATE EFFECT Replacement of
... pairs, we see that the log value for complex with ‘en’ ligand is greater than that with NH3 ligands, meaning that they are more stable. The reason for the extra stability may be attributed to both enthalpy and entropy effects. For example, in the replacement of NH3 ligands by ‘en’, 1. The basic na ...
... pairs, we see that the log value for complex with ‘en’ ligand is greater than that with NH3 ligands, meaning that they are more stable. The reason for the extra stability may be attributed to both enthalpy and entropy effects. For example, in the replacement of NH3 ligands by ‘en’, 1. The basic na ...
ON THE ROAD TO CARBENE AND CARBYNE COMPLEXES
... a metal atom, we get, as before, the σ compounds, for example, the vinyl lithium derivatives. In the second type, only the π electrons of the double bond are used for binding the organic molecule to the metal atom. In this way, we obtain π complexes (5,6) (Fig. 1b), the first representative of this ...
... a metal atom, we get, as before, the σ compounds, for example, the vinyl lithium derivatives. In the second type, only the π electrons of the double bond are used for binding the organic molecule to the metal atom. In this way, we obtain π complexes (5,6) (Fig. 1b), the first representative of this ...
Hydrogenation of lipid mixtures catalysed by palladium(II)
... applicable in the hydrogenation of lipids under mild conditions tolerable even by live cells. In the case of live systems only catalysts having considerable activities at low temperatures can be used. The solubility of the catalysts in water is extremely important, since the application of other sol ...
... applicable in the hydrogenation of lipids under mild conditions tolerable even by live cells. In the case of live systems only catalysts having considerable activities at low temperatures can be used. The solubility of the catalysts in water is extremely important, since the application of other sol ...
Ruthenium(II) Complexes Bearing a Pyridyl-Supported Pyrazolyl
... obtained by Claisen condensation of 4 with 3,3-dimethyl-2butone followed by reacting with hydrazine hydrate. The ligand precursor 6 was efficiently prepared by the reaction of 5 with 1-iodobutane in refluxing acetonitrile. When the tert-butyl group in 5 was substituted by a methyl group, the corresp ...
... obtained by Claisen condensation of 4 with 3,3-dimethyl-2butone followed by reacting with hydrazine hydrate. The ligand precursor 6 was efficiently prepared by the reaction of 5 with 1-iodobutane in refluxing acetonitrile. When the tert-butyl group in 5 was substituted by a methyl group, the corresp ...
Organometallic Chemistry
... the metabolism of every cell of the body, especially affecting DNA synthesis and regulation, but also fatty acid synthesis and energy production. Vitamin B-12 is the name for a class of chemically-related compounds, all of which have vitamin activity. It is structurally the most complicated vitamin ...
... the metabolism of every cell of the body, especially affecting DNA synthesis and regulation, but also fatty acid synthesis and energy production. Vitamin B-12 is the name for a class of chemically-related compounds, all of which have vitamin activity. It is structurally the most complicated vitamin ...
Document
... • Conversion of p Bond to 2 s Bonds Typically Energy Favored • Two s Bonds Higher Energy than One p + One s • Overall Process is thus Typically Exothermic ...
... • Conversion of p Bond to 2 s Bonds Typically Energy Favored • Two s Bonds Higher Energy than One p + One s • Overall Process is thus Typically Exothermic ...
Normal and abnormal carbene complexes derived
... S–C5–C4 angle. The normal thiazolylidene complex features, however, the smallest angle at the metal bound C2-atom. The compression of the S–C2–N bond angle upon metallation is accompanied by a distinct elongation of the C2–S1 and, to a slighter extent, of the C2–N3 bond. Identical modifications have ...
... S–C5–C4 angle. The normal thiazolylidene complex features, however, the smallest angle at the metal bound C2-atom. The compression of the S–C2–N bond angle upon metallation is accompanied by a distinct elongation of the C2–S1 and, to a slighter extent, of the C2–N3 bond. Identical modifications have ...
Chapter 1: General Introduction
... A free carbene molecule is comprised of a carbon with two substituents. One of the four carbon valence electrons is involved in σ–bonding to each of the substituents. The remaining two valence electrons are available for bonding. The carbene carbon atom therefore represents a six–electron species an ...
... A free carbene molecule is comprised of a carbon with two substituents. One of the four carbon valence electrons is involved in σ–bonding to each of the substituents. The remaining two valence electrons are available for bonding. The carbene carbon atom therefore represents a six–electron species an ...
Density Functional Study on the Preactivation Scenario of the
... of organic synthesis by means of organometallic catalysts is still rather scarce, but hopefully, recent advances in the methodology of computational chemistry, combined with the increasing performance of computer hardware, are starting to widen the range of molecules which can be calculated.15,16 Th ...
... of organic synthesis by means of organometallic catalysts is still rather scarce, but hopefully, recent advances in the methodology of computational chemistry, combined with the increasing performance of computer hardware, are starting to widen the range of molecules which can be calculated.15,16 Th ...
Ch 26 C-C bond formation
... Organoboranes in Suzuki Reaction • Two types of organoboranes can be used in the Suzuki reaction: vinylboranes and arylboranes. • Vinylboranes, which have a boron atom bonded to a carbon– carbon double bond, are prepared by hydroboration using catecholborane, a commercially available reagent. • Hyd ...
... Organoboranes in Suzuki Reaction • Two types of organoboranes can be used in the Suzuki reaction: vinylboranes and arylboranes. • Vinylboranes, which have a boron atom bonded to a carbon– carbon double bond, are prepared by hydroboration using catecholborane, a commercially available reagent. • Hyd ...
A Bonding Model for Gold(I) Carbene Complexes
... yield, 11:1 cis:trans). Phosphine ligands fell in between these two extremes. Finally, AuCl was unreactive under these conditions. Charge distribution. In order to provide a better and more general view of the bonding in gold vinyl carbene species, we calculated natural charge distributions for the ...
... yield, 11:1 cis:trans). Phosphine ligands fell in between these two extremes. Finally, AuCl was unreactive under these conditions. Charge distribution. In order to provide a better and more general view of the bonding in gold vinyl carbene species, we calculated natural charge distributions for the ...
Reactivity of Transition Metal Organometallics L. J. Farrugia MSc
... At the very lowest temperature measured, the 1H NMR spectrum shows a singlet for the two (equivalent) η5-C5H5 groups and an AA'BB'C multiplet for the two (equivalent) η1-C5H5 groups (three different H environments). This compound shows two fluxional processes (i) migration of metal atom around the η ...
... At the very lowest temperature measured, the 1H NMR spectrum shows a singlet for the two (equivalent) η5-C5H5 groups and an AA'BB'C multiplet for the two (equivalent) η1-C5H5 groups (three different H environments). This compound shows two fluxional processes (i) migration of metal atom around the η ...
Organometallic Compounds
... – This includes interactions between the d-orbitals and the donor/-acceptor orbitals of the six ligands. – Understand this diagram in terms and strengths of the different types of interactions. – 18-electron is the most stable for this type of complex. Assuming the d-orbitals to be at similar ener ...
... – This includes interactions between the d-orbitals and the donor/-acceptor orbitals of the six ligands. – Understand this diagram in terms and strengths of the different types of interactions. – 18-electron is the most stable for this type of complex. Assuming the d-orbitals to be at similar ener ...
Recall
... Important Synthetic Technique: protecting groups. Using Silyl ethers to Protect Alcohols Protecting groups are used to temporarily deactivate a functional group while reactions are done on another part of the molecule. The group is then restored. Example: ROH can react with either acid or base. We w ...
... Important Synthetic Technique: protecting groups. Using Silyl ethers to Protect Alcohols Protecting groups are used to temporarily deactivate a functional group while reactions are done on another part of the molecule. The group is then restored. Example: ROH can react with either acid or base. We w ...
Synthesis and Characterisation of N
... I hereby certify that I am the sole author of this thesis and that no part of this thesis has been published or submitted for publication. I certify that, to the best of my knowledge, my thesis does not infringe upon anyone’s copyright nor violate any proprietary rights and that any ideas, technique ...
... I hereby certify that I am the sole author of this thesis and that no part of this thesis has been published or submitted for publication. I certify that, to the best of my knowledge, my thesis does not infringe upon anyone’s copyright nor violate any proprietary rights and that any ideas, technique ...
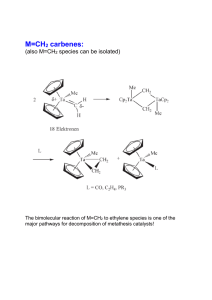
Steric protection of alkylidene is not needed:
... The bimolecular reaction of M=CH2 to ethylene species is one of the major pathways for decomposition of metathesis catalysts! ...
... The bimolecular reaction of M=CH2 to ethylene species is one of the major pathways for decomposition of metathesis catalysts! ...
Diverse Chemical Applications of N
... These materials have been demonstrated to undergo photochromic reactions that may provide opportunities for materials and catalysts with photoswitchable functions. N-Heterocyclic carbene ligands are useful in many complexes due to the steric protection they offer while maintaining a low coordination ...
... These materials have been demonstrated to undergo photochromic reactions that may provide opportunities for materials and catalysts with photoswitchable functions. N-Heterocyclic carbene ligands are useful in many complexes due to the steric protection they offer while maintaining a low coordination ...
Persistent carbene

A persistent carbene (also known as stable carbene) is a type of carbene demonstrating particular stability. The best-known examples and by far largest subgroup are the N-heterocyclic carbenes (sometimes called Arduengo carbenes), for example diaminocarbenes with the general formula (R2N)2C:, where the 'R's are typically alkyl and aryl groups. The groups can be linked to give heterocyclic carbenes, such as those derived from imidazole, imidazoline, thiazole or triazole.Traditionally carbenes are viewed as so reactive that were only studied indirectly, e.g. by trapping reactions. This situation has changed dramatically with the emergence of persistent carbenes. Although they are fairly reactive substances, i.e., undergoing dimerization, many can be isolated as pure substances.Persistent carbenes can exist in the singlet or the triplet states with the singlet state carbenes being more stable. The relative stability of these compounds is only partly due to steric hindrance by bulky groups. Some singlet carbenes are thermodynamically stable in the absence of moisture and (in most cases) oxygen, and can be isolated and indefinitely stored. Others are not thermodynamically stable and will dimerise slowly over days. The less stable triplet state carbenes have half-lives measured in seconds, and therefore can be observed but not stored.