Esters amines and amides
... Carboxylic acids: • form hydrogen bonds with many water molecules. • with 1-4 carbon atoms are very soluble in water. • as carbon atoms increase in chains, solubility decreases. ...
... Carboxylic acids: • form hydrogen bonds with many water molecules. • with 1-4 carbon atoms are very soluble in water. • as carbon atoms increase in chains, solubility decreases. ...
CHAPTER 15 ACIDS AND BASES
... Table 15.2 of the text contains a list of important Brønsted acids and bases. (a) both (why?), (b) base, (c) acid, (d) base, (e) acid, (f) base, (g) base, (h) base, (i) acid, (j) acid. ...
... Table 15.2 of the text contains a list of important Brønsted acids and bases. (a) both (why?), (b) base, (c) acid, (d) base, (e) acid, (f) base, (g) base, (h) base, (i) acid, (j) acid. ...
1 Acids and Bases
... Arrhenius bases are easy to spot because basically all of them are salts where the cation is a metal (or ammonium) and the anion is hydroxide. Due to the way we name ionic compounds, these substances will also have “hydroxide” in the name. Acids can sometimes be a little trickier if all you have is ...
... Arrhenius bases are easy to spot because basically all of them are salts where the cation is a metal (or ammonium) and the anion is hydroxide. Due to the way we name ionic compounds, these substances will also have “hydroxide” in the name. Acids can sometimes be a little trickier if all you have is ...
Chapter 14 - faculty at Chemeketa
... naturally-derived ethanoic acid to remove keratin debris buildup from under the nail. With regular use, your nails are clear and healthy-looking – or your money back. Diabetic-friendly NonyX Nail gel is used by podiatrists and can be found in the footcare aisle of your favorite pharmacy or mass reta ...
... naturally-derived ethanoic acid to remove keratin debris buildup from under the nail. With regular use, your nails are clear and healthy-looking – or your money back. Diabetic-friendly NonyX Nail gel is used by podiatrists and can be found in the footcare aisle of your favorite pharmacy or mass reta ...
CHEMISTRY OF p-ELEMENTS - Львівський національний
... including proteins, nucleic acids, hydrocarbons, enzymes, vitamins. The study of life is known as biological chemistry or biochemistry. Oxygen atoms are present in water (H2O) and water is essential to all life. Oxygen is present in many organic compounds. Most organisms use oxygen for respiration. ...
... including proteins, nucleic acids, hydrocarbons, enzymes, vitamins. The study of life is known as biological chemistry or biochemistry. Oxygen atoms are present in water (H2O) and water is essential to all life. Oxygen is present in many organic compounds. Most organisms use oxygen for respiration. ...
Practical and Efficient Synthesis of -Aminophosphonic Acids
... rigid unusual secondary α-amino acids, where the nitrogen is involved in a ring, may result in significant consequences for the conformation of peptidomimetics as synthetic tools for drug discovery [1,2]. Some of the most important molecules are the 1,2,3,4-tetrahydroquinoline-2-carboxylic acid 1 [3 ...
... rigid unusual secondary α-amino acids, where the nitrogen is involved in a ring, may result in significant consequences for the conformation of peptidomimetics as synthetic tools for drug discovery [1,2]. Some of the most important molecules are the 1,2,3,4-tetrahydroquinoline-2-carboxylic acid 1 [3 ...
RSE on PVC «South Kazakhstan state pharmaceutical academy
... ~ For determination of admissible arsenic in medicines by Sanger-Black hinder connections of .... |antimony, phosphorus, heavy metals, sulphides and sulphites ions |chlorine, barium, potassium, sulfur, |sodium, calcium, magnesium, chlorine |magnesium, chloride, sodium, potassium, |sodium, potassium, ...
... ~ For determination of admissible arsenic in medicines by Sanger-Black hinder connections of .... |antimony, phosphorus, heavy metals, sulphides and sulphites ions |chlorine, barium, potassium, sulfur, |sodium, calcium, magnesium, chlorine |magnesium, chloride, sodium, potassium, |sodium, potassium, ...
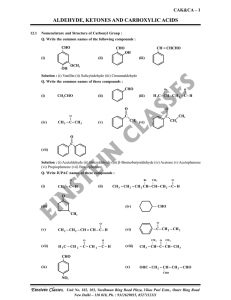
aldehyde, ketones and carboxylic acids
... Solution : A nucleophile attacks the electrophilic carbon atom of the polar carbonyl group from a direction approximately perpendicular to the plane of sp2 hybridised orbitals of carbonyl carbon. The hybridisation of carbon changes from sp2 to sp3 in this process, and a tetrahedral alkoxide intermed ...
... Solution : A nucleophile attacks the electrophilic carbon atom of the polar carbonyl group from a direction approximately perpendicular to the plane of sp2 hybridised orbitals of carbonyl carbon. The hybridisation of carbon changes from sp2 to sp3 in this process, and a tetrahedral alkoxide intermed ...
Chlorine
... Furthermore, electrolysis of fused chloride salts ( Downs process ) also enables chlorine to be produced, in this case as a by product of the manufacture of metallic sodium or magnesium. 5 . 2 . Other methods Before electrolytic methods were used for chlorine production, the direct oxidation of hydr ...
... Furthermore, electrolysis of fused chloride salts ( Downs process ) also enables chlorine to be produced, in this case as a by product of the manufacture of metallic sodium or magnesium. 5 . 2 . Other methods Before electrolytic methods were used for chlorine production, the direct oxidation of hydr ...
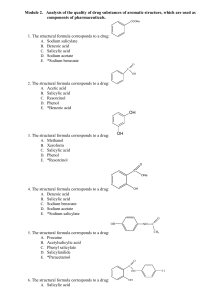
Module 2. Drug substances of aromatic structure
... E. *Bismuth tribromophenol basic with bismuth oxide 82. For synthesis of thymol it is possible to use such initial substance: A. Phenylsalicylate B. Phthalic acid C. Benzol D. Phenol E. *3-Metylphenol (m-cresol) 83. For assay of resorcinol, according to Pharmacopoeia, use method: A. Cerymetry, direc ...
... E. *Bismuth tribromophenol basic with bismuth oxide 82. For synthesis of thymol it is possible to use such initial substance: A. Phenylsalicylate B. Phthalic acid C. Benzol D. Phenol E. *3-Metylphenol (m-cresol) 83. For assay of resorcinol, according to Pharmacopoeia, use method: A. Cerymetry, direc ...
Lecture 13 11-20-02
... Since the analyte is diprotic there are two equivalence points, each requiring the same volume of titrant. Before the first equivalence point the pH is controlled by a buffer consisting of H 2A and HA-, and the HA-/A2- buffer determines the pH between the two equivalence points. After the second equ ...
... Since the analyte is diprotic there are two equivalence points, each requiring the same volume of titrant. Before the first equivalence point the pH is controlled by a buffer consisting of H 2A and HA-, and the HA-/A2- buffer determines the pH between the two equivalence points. After the second equ ...
chapter 15 acids and bases
... This problem could be solved more easily if we could assume that (0.10 − x) ≈ 0.10. If the assumption is mathematically valid, then it would not be necessary to solve a quadratic equation, as we did above. Re-solve the problem above, making the assumption. Was the assumption valid? What is our crite ...
... This problem could be solved more easily if we could assume that (0.10 − x) ≈ 0.10. If the assumption is mathematically valid, then it would not be necessary to solve a quadratic equation, as we did above. Re-solve the problem above, making the assumption. Was the assumption valid? What is our crite ...
Chemistry 217 Problem Set 3 Recommended Problems from the Book
... of the carbons are attached to four groups, which should make them sp3-hybridized. However, the bond angles of the three-membered ring must be 60°, an angle more consistent with an sp2 hybridized carbon than an sp3, which has bond angles of 109.5°. 5. IR spectroscopy is commonly used by crime labs t ...
... of the carbons are attached to four groups, which should make them sp3-hybridized. However, the bond angles of the three-membered ring must be 60°, an angle more consistent with an sp2 hybridized carbon than an sp3, which has bond angles of 109.5°. 5. IR spectroscopy is commonly used by crime labs t ...
PRACTICAL ORGANIC CHEMISTRY
... III-1-5-Test for Fructose: Upon heating a little fructose with dilute CoCl2 solution, cooling and treat with a little ammonia solution. ...
... III-1-5-Test for Fructose: Upon heating a little fructose with dilute CoCl2 solution, cooling and treat with a little ammonia solution. ...
Anhydrides, Esters and Amides
... 19 Hydrolysis of Esters • Esters hydrolyze only very slowly, even in boiling water. • Hydrolysis becomes considerably more rapid, however, when the ester is heated in aqueous acid or base. • Hydrolysis of esters in aqueous acid is the reverse of Fischer esterification. • A large excess of water dri ...
... 19 Hydrolysis of Esters • Esters hydrolyze only very slowly, even in boiling water. • Hydrolysis becomes considerably more rapid, however, when the ester is heated in aqueous acid or base. • Hydrolysis of esters in aqueous acid is the reverse of Fischer esterification. • A large excess of water dri ...
Acids and Bases
... respectively. However, there are plenty of compounds that contain oxygen and hydrogen atoms that are not acids and bases. What other clues can we look for? Arrhenius bases are easy to spot because basically all of them are salts where the cation is a metal (or ammonium) and the anion is hydroxide. D ...
... respectively. However, there are plenty of compounds that contain oxygen and hydrogen atoms that are not acids and bases. What other clues can we look for? Arrhenius bases are easy to spot because basically all of them are salts where the cation is a metal (or ammonium) and the anion is hydroxide. D ...
H/ t = W/m
... constant of the thermocouple was found by placing the thermocouple in three consecutive beakers of water that varied in temperature. It was held in the first beaker for 20 seconds then removed for 20 seconds before being placed in the next beaker. LabView was used to record voltage as a function of ...
... constant of the thermocouple was found by placing the thermocouple in three consecutive beakers of water that varied in temperature. It was held in the first beaker for 20 seconds then removed for 20 seconds before being placed in the next beaker. LabView was used to record voltage as a function of ...
Lecture 22 - Chemistry Courses
... For a weak acid, the extent of proton transfer is so small that it is usually assumed that the molar concentration of HA is unchanged from its nominal value. Also, the molar amounts of H3O+ and A– formed from the reaction are equal (we amount from the autoprotolysis of water is considered minimal), ...
... For a weak acid, the extent of proton transfer is so small that it is usually assumed that the molar concentration of HA is unchanged from its nominal value. Also, the molar amounts of H3O+ and A– formed from the reaction are equal (we amount from the autoprotolysis of water is considered minimal), ...
PAGE PROOFS
... What can send a shiver down your spine, bring tears to your eyes and make your mouth pucker? Simply the taste of a freshly cut lemon or unripe pineapple. The vinegar in salad dressing, though less stringent, has the same effect. Even a cola or a glass of sparkling wine may leave a slightly sour tast ...
... What can send a shiver down your spine, bring tears to your eyes and make your mouth pucker? Simply the taste of a freshly cut lemon or unripe pineapple. The vinegar in salad dressing, though less stringent, has the same effect. Even a cola or a glass of sparkling wine may leave a slightly sour tast ...
Leaching of Sphalerite with Hydrogen Peroxide and Nitric Acid
... The leaching experiments were carried out in an all glass reactor (250ml) equipped with a mechanical stirrer. It was heated at constant temperature in a water bath within a temperature in the range of ± 1oC. A mixture of 50cm3 of hydrogen peroxide and 50cm3 of the nitric acid was charged into the re ...
... The leaching experiments were carried out in an all glass reactor (250ml) equipped with a mechanical stirrer. It was heated at constant temperature in a water bath within a temperature in the range of ± 1oC. A mixture of 50cm3 of hydrogen peroxide and 50cm3 of the nitric acid was charged into the re ...
Journal of Molecular Catalysis A, 302
... Table 1 summarizes the results of scoping experiments in which some components of the reaction mixture were omitted in order to determine their importance. Entries 1 and 2 show standard reaction conditions for Rh- and Pd-catalyzed reactions, respectively. The observed products in all cases were 4-tr ...
... Table 1 summarizes the results of scoping experiments in which some components of the reaction mixture were omitted in order to determine their importance. Entries 1 and 2 show standard reaction conditions for Rh- and Pd-catalyzed reactions, respectively. The observed products in all cases were 4-tr ...
- Wiley Online Library
... molecular-sized clusters. It is well known that CNT cannot describe the thermodynamics of the smallest clusters accurately, and this deviation in the smallest scale can lead to very large errors in the particle formation rate calculations. Furthermore, the standard CNT for ion-induced binary system ...
... molecular-sized clusters. It is well known that CNT cannot describe the thermodynamics of the smallest clusters accurately, and this deviation in the smallest scale can lead to very large errors in the particle formation rate calculations. Furthermore, the standard CNT for ion-induced binary system ...
factors affecting strength of acids
... - Completely or nearly completely ionize in aqueous solution - Strong electrolytes Hydroxides of Groups IA and IIA are strong bases LiOH, CsOH, Ba(OH)2, Ca(OH)2 most common in lab: NaOH and KOH Weak bases - produce small amounts of OH- ions in aqueous solution Organic bases, methylamine, cocaine, mo ...
... - Completely or nearly completely ionize in aqueous solution - Strong electrolytes Hydroxides of Groups IA and IIA are strong bases LiOH, CsOH, Ba(OH)2, Ca(OH)2 most common in lab: NaOH and KOH Weak bases - produce small amounts of OH- ions in aqueous solution Organic bases, methylamine, cocaine, mo ...
Caboxylic acid Derivatives
... Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are esters, amides and nitriles, although acid halides and anhydrides are also derivatives (really activated forms of a carboxyl ...
... Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are esters, amides and nitriles, although acid halides and anhydrides are also derivatives (really activated forms of a carboxyl ...
word document
... Anhydride is a compound formed by the abstraction of a molecule of water, H2O, from a substance. The term acid anhydride is restricted sometime to the anhydride formed especially from an acid by dehydration or one that revert to the original substance upon hydration. In case of bimolecular, it can b ...
... Anhydride is a compound formed by the abstraction of a molecule of water, H2O, from a substance. The term acid anhydride is restricted sometime to the anhydride formed especially from an acid by dehydration or one that revert to the original substance upon hydration. In case of bimolecular, it can b ...
Sulfuric acid
Sulfuric acid (alternative spelling sulphuric acid) is a highly corrosive strong mineral acid with the molecular formula H2SO4 and molecular weight 98.079 g/mol. It is a pungent-ethereal, colorless to slightly yellow viscous liquid which is soluble in water at all concentrations. Sometimes, it is dyed dark brown during production to alert people to its hazards. The historical name of this acid is oil of vitriol.Sulfuric acid is a diprotic acid and shows different properties depending upon its concentration. Its corrosiveness on other materials, like metals, living tissues or even stones, can be mainly ascribed to its strong acidic nature and, if concentrated, strong dehydrating and oxidizing properties. Sulfuric acid at a high concentration can cause very serious damage upon contact, since not only does it cause chemical burns via hydrolysis, but also secondary thermal burns through dehydration. It can lead to permanent blindness if splashed onto eyes and irreversible damage if swallowed. Accordingly, safety precautions should be strictly observed when handling it. Moreover, it is hygroscopic, readily absorbing water vapour from the air.Sulfuric acid has a wide range of applications including domestic acidic drain cleaner, electrolyte in lead-acid batteries and various cleaning agents. It is also a central substance in the chemical industry. Principal uses include mineral processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. It is widely produced with different methods, such as contact process, wet sulfuric acid process and some other methods.