LAP5 and LAP6 encode anther-specific proteins with similarity to
... layers: (i) an outer exine, itself a multilayered structure, primarily made of sporopollenin, (ii) an inner intine, made primarily of cellulose, and (iii) a lipid- and protein-rich pollen coat in the crevices of exine. The exine is morphologically diverse, provides protection against environmental s ...
... layers: (i) an outer exine, itself a multilayered structure, primarily made of sporopollenin, (ii) an inner intine, made primarily of cellulose, and (iii) a lipid- and protein-rich pollen coat in the crevices of exine. The exine is morphologically diverse, provides protection against environmental s ...
biolsci.org - International Journal of Biological Sciences
... examples (Figure 1). In E. coli, THZ-P is derived from an oxidative condensation of tyrosine, cysteine, and 1-deoxy-D-xylulose 5-phosphate (DXP). Seven genes (thiF, thiS, thiG, thiH, thiI, iscS, and Dxs) are involved in this process [28-35]. B. subtilis THZ-P biosynthesis is different from E. coli s ...
... examples (Figure 1). In E. coli, THZ-P is derived from an oxidative condensation of tyrosine, cysteine, and 1-deoxy-D-xylulose 5-phosphate (DXP). Seven genes (thiF, thiS, thiG, thiH, thiI, iscS, and Dxs) are involved in this process [28-35]. B. subtilis THZ-P biosynthesis is different from E. coli s ...
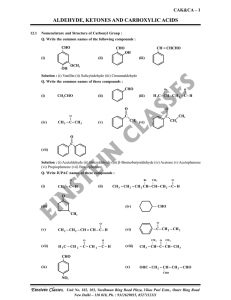
aldehyde, ketones and carboxylic acids
... of carbon changes from sp2 to sp3 in this process, and a tetrahedral alkoxide intermediate is produced. This intermediate captures a proton from the reaction medium to give the electrically neutral product. The net result is addition of Nu– and H+ across the carbon oxygen double bond. Q. Why Aldehyd ...
... of carbon changes from sp2 to sp3 in this process, and a tetrahedral alkoxide intermediate is produced. This intermediate captures a proton from the reaction medium to give the electrically neutral product. The net result is addition of Nu– and H+ across the carbon oxygen double bond. Q. Why Aldehyd ...
Lipoteichoic Acid Synthesis and Function in Gram
... hydroxyl groups at the C2 position of the GroP repeating units are, to varying degrees, modified with D-alanyl or glycosyl groups. The key enzymes involved in type I LTA synthesis have been identified and are depicted in Figure 2 for S. aureus. The glycolipid anchor is produced in the cytoplasm of the ...
... hydroxyl groups at the C2 position of the GroP repeating units are, to varying degrees, modified with D-alanyl or glycosyl groups. The key enzymes involved in type I LTA synthesis have been identified and are depicted in Figure 2 for S. aureus. The glycolipid anchor is produced in the cytoplasm of the ...
A Metabolic Node in Action: Chorismate
... The native AroQt.TyrA enzyme is a dimer and is retro-inhibited by Tyr (45% for the CM and 95% for the dehydrogenase activity).44,45 It is now evident that the mutase and the dehydrogenase active sites are distinct, but display some similar chemical properties.46-50 Kinetic studies have shown that T ...
... The native AroQt.TyrA enzyme is a dimer and is retro-inhibited by Tyr (45% for the CM and 95% for the dehydrogenase activity).44,45 It is now evident that the mutase and the dehydrogenase active sites are distinct, but display some similar chemical properties.46-50 Kinetic studies have shown that T ...
NPTEL-Module-1: Introduction to Bioorganic Chemistry Dr. S. S. Bag
... biradical specis via Bergman cyclization. In addition to that neocarzinostatin (NCS) chromophore which does not contain the classical conjugated enediyne system also demonstrated very similar DNA cleavage mechanism via the generation biradical species through the MyersSaito cyclization. The enediyne ...
... biradical specis via Bergman cyclization. In addition to that neocarzinostatin (NCS) chromophore which does not contain the classical conjugated enediyne system also demonstrated very similar DNA cleavage mechanism via the generation biradical species through the MyersSaito cyclization. The enediyne ...
Fragmentation pathway for glutamine identification: Loss of 73 da
... to initiate cleavage. Scheme 1 illustrates a two-step process for the loss of 73: i) the migration of a hydrogen from the butyl ester to the°adjacent°carbonyl°oxygen°via°a°six-membered°ring structure which, together with the loss of isobutene (56 Da) leads to the dimethylformamidine glutamine struct ...
... to initiate cleavage. Scheme 1 illustrates a two-step process for the loss of 73: i) the migration of a hydrogen from the butyl ester to the°adjacent°carbonyl°oxygen°via°a°six-membered°ring structure which, together with the loss of isobutene (56 Da) leads to the dimethylformamidine glutamine struct ...
Terpene Biosynthesis
... hmg1 gene in tobacco (Nicotiana tabacum) was found to decrease general isoprenoid levels.15 The crystal structures of two non-productive ternary complexes of HMGR from Pseudomonas mevalonii with HMGCoA/NAD⫹ and with MVA/NADH have been determined.16 In the structure of the apoenzyme reported earlier, ...
... hmg1 gene in tobacco (Nicotiana tabacum) was found to decrease general isoprenoid levels.15 The crystal structures of two non-productive ternary complexes of HMGR from Pseudomonas mevalonii with HMGCoA/NAD⫹ and with MVA/NADH have been determined.16 In the structure of the apoenzyme reported earlier, ...
Sugar Amino Acids - The Krasavin research group
... Ag2 O, followed by esterification with diazomethane, and final catalytic hydrogenation over Pd/C catalysis to give the final furanoid α-amino acid 5, as the α- or β-anomer, depending on the stereochemistry of starting thiazolyl ketol acetate. This approach was also reported for a galacto-derived pyr ...
... Ag2 O, followed by esterification with diazomethane, and final catalytic hydrogenation over Pd/C catalysis to give the final furanoid α-amino acid 5, as the α- or β-anomer, depending on the stereochemistry of starting thiazolyl ketol acetate. This approach was also reported for a galacto-derived pyr ...
Side Chain Chemistry Mediates Backbone Fragmentation in
... A crown ether based, photolabile radical precursor which forms noncovalent complexes with peptides has been prepared. The peptide/precursor complexes can be electrosprayed, isolated in an ion trap, and then subjected to laser photolysis and collision induced dissociation to generate hydrogen deficie ...
... A crown ether based, photolabile radical precursor which forms noncovalent complexes with peptides has been prepared. The peptide/precursor complexes can be electrosprayed, isolated in an ion trap, and then subjected to laser photolysis and collision induced dissociation to generate hydrogen deficie ...
Fatty Acids :biosynthesis
... ^ A new malonyl CoA binds ACP on other subunit. • The cycle (condendation, reduction, dehydration, reduction) repeats until 16 carbon palmitate is formed (not shown). • Palmitate is released by TE (see slide 14). ...
... ^ A new malonyl CoA binds ACP on other subunit. • The cycle (condendation, reduction, dehydration, reduction) repeats until 16 carbon palmitate is formed (not shown). • Palmitate is released by TE (see slide 14). ...
LipidMetabolism
... Prostaglandin H synthase (PGHS) 132 kDa dimer binds on inner surface of ER Monomer shown ...
... Prostaglandin H synthase (PGHS) 132 kDa dimer binds on inner surface of ER Monomer shown ...
Novel targets for tuberculosis drug discovery
... enzymes are closely related and share a common cofactor, S-adenosyl methionine. Analogues of S-adenosyl methionine have been successfully synthesized and are effective inhibitors of bacterial and fungal methyltransferases [24]. Pks13, the enzyme that catalyzes the final condensation step in mycolic ...
... enzymes are closely related and share a common cofactor, S-adenosyl methionine. Analogues of S-adenosyl methionine have been successfully synthesized and are effective inhibitors of bacterial and fungal methyltransferases [24]. Pks13, the enzyme that catalyzes the final condensation step in mycolic ...
New Reactions in the Crotonase Superfamily: Structure of
... similar active sites have evolved to catalyze different overall reactions. These investigations complement and contrast ongoing studies of members of the enolase superfamily (19). In the enolase superfamily, the active sites are found at the interfaces between two domains: the catalytic groups are l ...
... similar active sites have evolved to catalyze different overall reactions. These investigations complement and contrast ongoing studies of members of the enolase superfamily (19). In the enolase superfamily, the active sites are found at the interfaces between two domains: the catalytic groups are l ...
3 ON THE THERMODYNAMICS OF FATTY ACID OXIDATION
... and so, combined with (3), the limiting yield of ATP is about 0.5 mol g-1. The oxidation of the FAs from human fat or rattite or seed oils yields about 0.41 mol ATP g-1 (9) despite quite different FA compositions (Table 1). Naturally, the ATP yield increases with n and decreases only slightly with ...
... and so, combined with (3), the limiting yield of ATP is about 0.5 mol g-1. The oxidation of the FAs from human fat or rattite or seed oils yields about 0.41 mol ATP g-1 (9) despite quite different FA compositions (Table 1). Naturally, the ATP yield increases with n and decreases only slightly with ...
Enzyme screening of dikaryotic cultures from lignocellulolitic
... as expected. The drop enzyme tests showed tyrosinase was present in Phellinus flavomarginatus, Rigidoporus lineatus and Antrodia albida. Other enzymes used with this drop test were laccase and peroxidases, which were detected in 15 strains, the exception being Antrodia albida. In liquid media, howev ...
... as expected. The drop enzyme tests showed tyrosinase was present in Phellinus flavomarginatus, Rigidoporus lineatus and Antrodia albida. Other enzymes used with this drop test were laccase and peroxidases, which were detected in 15 strains, the exception being Antrodia albida. In liquid media, howev ...
Cytochrome P450-enzymes involved in the biosynthesis of mono
... of Tennessee Health Science Center, named over 18,000 genes attributed to several hundred families (Nelson 2012). Generally, a common overall topology and tridimensional fold of all cytochromes P450 have been observed (Graham and Peterson 1999) despite their relative low sequence similarity on amino ...
... of Tennessee Health Science Center, named over 18,000 genes attributed to several hundred families (Nelson 2012). Generally, a common overall topology and tridimensional fold of all cytochromes P450 have been observed (Graham and Peterson 1999) despite their relative low sequence similarity on amino ...
MCAD - MSOE Center for BioMolecular Modeling
... Enzymes catalyzes the rate of reaction Multiple Intermolecular Interactions ...
... Enzymes catalyzes the rate of reaction Multiple Intermolecular Interactions ...
Nikolai N. Semenov - Nobel Lecture
... 700 to 800ºC), and in the experiments of N. M. Emanuel18, who was concerned with the slow reaction of hydrogen sulfide oxidation (the reaction took some minutes). In both cases, the presence of radicals (H, OH, SO) was observed directly, the number of H atoms rising up by 10% compared with the start ...
... 700 to 800ºC), and in the experiments of N. M. Emanuel18, who was concerned with the slow reaction of hydrogen sulfide oxidation (the reaction took some minutes). In both cases, the presence of radicals (H, OH, SO) was observed directly, the number of H atoms rising up by 10% compared with the start ...
Cloning and sequencing of the kedarcidin biosynthetic
... (Fig. S1, ESI†), and the solution structure solved by NMR spectroscopy.3 The structure of the KED chromophore was first established on the basis of an extensive spectroscopic analysis in 1992.4,5 It has since been revised twice according to total syntheses6,7 with the final revised structure shown i ...
... (Fig. S1, ESI†), and the solution structure solved by NMR spectroscopy.3 The structure of the KED chromophore was first established on the basis of an extensive spectroscopic analysis in 1992.4,5 It has since been revised twice according to total syntheses6,7 with the final revised structure shown i ...
Word - icho39.chem.msu.ru
... Let nH 2 = nH2 H2 , where H 2 is the number of moles of hydrogen added to the system. Since H 2 is small, H2 ...
... Let nH 2 = nH2 H2 , where H 2 is the number of moles of hydrogen added to the system. Since H 2 is small, H2 ...
Structural and Biochemical Characterization of a Bifunctional
... of various Gram-negative bacteria and in the S-layers glycans of some Gram-positive bacteria. The biosynthetic pathway for its production in Aneurinibacillus thermoaerophilus L420-91T was elucidated in 2003 and is shown in Scheme 1.1 The first step, catalyzed by a thymidylyltransferase (RmlA), involv ...
... of various Gram-negative bacteria and in the S-layers glycans of some Gram-positive bacteria. The biosynthetic pathway for its production in Aneurinibacillus thermoaerophilus L420-91T was elucidated in 2003 and is shown in Scheme 1.1 The first step, catalyzed by a thymidylyltransferase (RmlA), involv ...
fulltext
... is a result of unfolding and subsequent aggregation of the protein chains. Looking at proteins in micro scale also yields fascinating findings. An enzyme is able to bind one or more distinct molecules or atoms, its substrates, and provide an environment which is more favourable for a chemical reacti ...
... is a result of unfolding and subsequent aggregation of the protein chains. Looking at proteins in micro scale also yields fascinating findings. An enzyme is able to bind one or more distinct molecules or atoms, its substrates, and provide an environment which is more favourable for a chemical reacti ...
ANTIBIOTICS
... Our knowledge of the biosynthesis of peptide antibiotics comes mostly from the study of the gramicidin S and bacitracin synthetases. Gramicidin S synthetase consists of two complementary enzymes having molecular weights of 100 kD and 280 kD while bacitracin synthetase consists of three subunits (Rol ...
... Our knowledge of the biosynthesis of peptide antibiotics comes mostly from the study of the gramicidin S and bacitracin synthetases. Gramicidin S synthetase consists of two complementary enzymes having molecular weights of 100 kD and 280 kD while bacitracin synthetase consists of three subunits (Rol ...
Biosynthesis of doxorubicin
Doxorubicin (DXR) is a 14-hydroxylated version of daunorubicin, the immediate precursor of DXR in its biosynthetic pathway. Daunorubicin is more abundantly found as a natural product because it is produced by a number of different wild type strains of streptomyces. In contrast, only one known non-wild type species, streptomyces peucetius subspecies cesius ATCC 27952, was initially found to be capable of producing the more widely used doxorubicin. This strain was created by Arcamone et al. in 1969 by mutating a strain producing daunorubicin, but not DXR, at least in detectable quantities. Subsequently, Hutchinson's group showed that under special environmental conditions, or by the introduction of genetic modifications, other strains of streptomyces can produce doxorubicin. His group has also cloned many of the genes required for DXR production, although not all of them have been fully characterized. In 1996, Strohl's group discovered, isolated and characterized dox A, the gene encoding the enzyme that converts daunorubicin into DXR. By 1999, they produced recombinant Dox A, a Cytochrome P450 oxidase, and found that it catalyzes multiple steps in DXR biosynthesis, including steps leading to daunorubicin. This was significant because it became clear that all daunorubicin producing strains have the necessary genes to produce DXR, the much more therapeutically important of the two. Hutchinson's group went on to develop methods to improve the yield of DXR, from the fermentation process used in its commercial production, not only by introducing Dox A encoding plasmids, but also by introducing mutations to deactivate enzymes that shunt DXR precursors to less useful products, for example baumycin-like glycosides. Some triple mutants, that also over-expressed Dox A, were able to double the yield of DXR. This is of more than academic interest because at that time DXR cost about $1.37 million per kg and current production in 1999 was 225 kg per annum. More efficient production techniques have brought the price down to $1.1 million per kg for the non-liposomal formulation. Although DXR can be produced semi-synthetically from daunorubicin, the process involves electrophilic bromination and multiple steps and the yield is poor. Since daunorubicin is produced by fermentation, it would be ideal if the bacteria could complete DXR synthesis more effectively.