Chemistry 1000 Lecture 6: Quantum mechanics and spectroscopy
... Chemistry 1000 Lecture 6: Quantum mechanics and spectroscopy Marc R. Roussel ...
... Chemistry 1000 Lecture 6: Quantum mechanics and spectroscopy Marc R. Roussel ...
Chapter 5 Electrons in Atoms
... piece at the center of the atom- “nucleus” Electrons would surround and move around it, like planets around the sun Atom is mostly empty space It did not explain the chemical properties of the elements – a better description of the electron behavior was needed ...
... piece at the center of the atom- “nucleus” Electrons would surround and move around it, like planets around the sun Atom is mostly empty space It did not explain the chemical properties of the elements – a better description of the electron behavior was needed ...
Lecture 15: The Hydrogen Atom
... Hydrogen Atom is Unstable? It is known that accelerating charges emit radiation Thus, electron should emit radiation, lose energy and eventually fall into the nucleus! Why doesn’t this happen? Shows that something was wrong with this model of the hydrogen atom ...
... Hydrogen Atom is Unstable? It is known that accelerating charges emit radiation Thus, electron should emit radiation, lose energy and eventually fall into the nucleus! Why doesn’t this happen? Shows that something was wrong with this model of the hydrogen atom ...
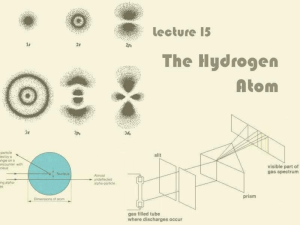
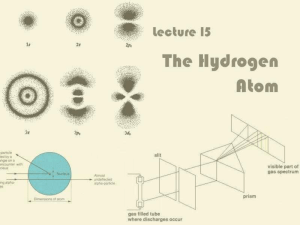
Lecture 15: The Hydrogen Atom
... Depending on which energy level it is in, the electron can take one of a number of stationary probability ...
... Depending on which energy level it is in, the electron can take one of a number of stationary probability ...
3.2 Bohr`s Model of the Atom
... The movement of an electron from one energy level to another is called a transition The ground state is the lowest energy state for an atom When an atom gains energy it moves into an excited state The transition from a lower energy level to a higher energy level requires a quantized amount of energy ...
... The movement of an electron from one energy level to another is called a transition The ground state is the lowest energy state for an atom When an atom gains energy it moves into an excited state The transition from a lower energy level to a higher energy level requires a quantized amount of energy ...
Discovery of the Electron, Models & Theories
... Schrodinger used mathematical equations to predict the likelihood of finding the electron and eventually found one that worked ...
... Schrodinger used mathematical equations to predict the likelihood of finding the electron and eventually found one that worked ...
Frank-Hertz Experiment with Argon
... elegantly supported Niels Bohr's model of the atom, with electrons orbiting the nucleus with specific, discrete energies. Franck and Hertz were awarded the Nobel Prize in Physics in 1925 for this work. In the early 20th century, experiments by Ernest Rutherford established that atoms consisted of a ...
... elegantly supported Niels Bohr's model of the atom, with electrons orbiting the nucleus with specific, discrete energies. Franck and Hertz were awarded the Nobel Prize in Physics in 1925 for this work. In the early 20th century, experiments by Ernest Rutherford established that atoms consisted of a ...
Set #4 - comsics
... electron typically spends about 10-8 s in an excited state before it drops to a lower state by emitting a photon. How many revolutions does an electron in an n = 2 Bohr orbit make in 10-8 s? ...
... electron typically spends about 10-8 s in an excited state before it drops to a lower state by emitting a photon. How many revolutions does an electron in an n = 2 Bohr orbit make in 10-8 s? ...
Atomic and Molecular Spectroscopy
... o Recommended Books: o The Physics of Atoms and Quanta: Introduction to experiement and theory Haken & Wolf (Springer) o Quantum Physics of Atoms, Molecules, Solids, Nuclei and Particles Eisberg & Resnick (Wiley) o Quantum Mechanics McMurry QuickTime™ and a TIFF (Uncompressed) decompressor are neede ...
... o Recommended Books: o The Physics of Atoms and Quanta: Introduction to experiement and theory Haken & Wolf (Springer) o Quantum Physics of Atoms, Molecules, Solids, Nuclei and Particles Eisberg & Resnick (Wiley) o Quantum Mechanics McMurry QuickTime™ and a TIFF (Uncompressed) decompressor are neede ...
Problem-set-6
... Consider a hydrogen atom in a state with principal quantum number n=45. These states with high-n quantum numbers are usually called Rydberg states. a) What is the binding energy of this state, calculated in eV and in units of cm-1. b) What is the absorption wavelength for an atom in n=45 making a tr ...
... Consider a hydrogen atom in a state with principal quantum number n=45. These states with high-n quantum numbers are usually called Rydberg states. a) What is the binding energy of this state, calculated in eV and in units of cm-1. b) What is the absorption wavelength for an atom in n=45 making a tr ...
Models of the Atom
... Could not explain why emission lines are double, triple or more Could not explain why some lines brighter than others Could not explain how atoms bond Mixed classical and quantum ideas ...
... Could not explain why emission lines are double, triple or more Could not explain why some lines brighter than others Could not explain how atoms bond Mixed classical and quantum ideas ...
The Atom
... What are the similarities and differences of the atomic models of Democritus, Aristotle, and Dalton? How was Dalton’s theory used to explain the conservation of mass? What is an atom? How can the subatomic particles be distinguished in terms of relative charge and mass? What is an isotope? Giv ...
... What are the similarities and differences of the atomic models of Democritus, Aristotle, and Dalton? How was Dalton’s theory used to explain the conservation of mass? What is an atom? How can the subatomic particles be distinguished in terms of relative charge and mass? What is an isotope? Giv ...
Simple Harmonic Oscillator
... Never express yourself more clearly than you are able to think. Prediction is very difficult, especially about the future. - Niels Bohr ...
... Never express yourself more clearly than you are able to think. Prediction is very difficult, especially about the future. - Niels Bohr ...
Atom is a basic unit of matter that consists of a nucleus
... tiny masses proportional to the volume implied by these dimensions. Atoms in solid states (or, to be precise, their electron clouds) can be observed individually using special instruments such as the scanning tunneling microscope. Hydrogen-1 (one proton + one electron) is the simplest form of atoms, ...
... tiny masses proportional to the volume implied by these dimensions. Atoms in solid states (or, to be precise, their electron clouds) can be observed individually using special instruments such as the scanning tunneling microscope. Hydrogen-1 (one proton + one electron) is the simplest form of atoms, ...
Physical Chemistry
... of matter --planetary motion, fluid flow, elasticity, etc. • Thermodynamics had its first two laws and most of their consequences • Basic statistical mechanics had been applied to chemical systems • Light was explained as an electromagnetic wave ...
... of matter --planetary motion, fluid flow, elasticity, etc. • Thermodynamics had its first two laws and most of their consequences • Basic statistical mechanics had been applied to chemical systems • Light was explained as an electromagnetic wave ...
5.1.03-15 Franck-Hertz experiment with Ne
... among those states producing radiation whose frequency is determined by the energy differences between states can be derived from the quantum mechanics which replaced classical mechanics when dealing with structures as small as atoms. It seems reasonable from the Bohr model that just as electrons ma ...
... among those states producing radiation whose frequency is determined by the energy differences between states can be derived from the quantum mechanics which replaced classical mechanics when dealing with structures as small as atoms. It seems reasonable from the Bohr model that just as electrons ma ...
the Bohr`s atom model - Latin-American Journal of Physics Education
... Palabras clave: Educación en Física, Historia de la Ciencia, Libros de Texto. PACS: 01.30.M. 01.40.-d, 01.65.+g ...
... Palabras clave: Educación en Física, Historia de la Ciencia, Libros de Texto. PACS: 01.30.M. 01.40.-d, 01.65.+g ...
R - University of St Andrews
... Elliptical orbits (Bohr-Sommerfeld) When viewed at high resolution, transitions split. Transitions between any two Bohr energy states involve several spectral lines. This is known as fine structure. Explanation: each energy level actually consists of several distinct states with almost the same ene ...
... Elliptical orbits (Bohr-Sommerfeld) When viewed at high resolution, transitions split. Transitions between any two Bohr energy states involve several spectral lines. This is known as fine structure. Explanation: each energy level actually consists of several distinct states with almost the same ene ...
Academic Chemistry Atomic History Study Guide 1. Identify and
... 10. A young engineer named ________________ ________________, who was killed in World War I, contributed greatly to science prior to his death by discovering the atomic number. 11. According to the laws of classical physics, a charged particle traveling on a curved path will lose energy. This would ...
... 10. A young engineer named ________________ ________________, who was killed in World War I, contributed greatly to science prior to his death by discovering the atomic number. 11. According to the laws of classical physics, a charged particle traveling on a curved path will lose energy. This would ...
Chem20u2(5.2) - Mr. Searcy Chemistry 20
... 5. Identify the relationships among a hydrogen atom’s energy levels, sublevels, and atomic orbitals. II. The following questions will help to cover these objectives as you read through the section. Briefly answer each question. ...
... 5. Identify the relationships among a hydrogen atom’s energy levels, sublevels, and atomic orbitals. II. The following questions will help to cover these objectives as you read through the section. Briefly answer each question. ...
Franck-Hertz experiment with Ne-tube Related Topics
... discrete states with transitions among those states producing radiation whose frequency is determined by the energy differences between states can be derived from the quantum mechanics which replaced classical mechanics when dealing with Fig. 3: Principle of the measurement. structures as small as a ...
... discrete states with transitions among those states producing radiation whose frequency is determined by the energy differences between states can be derived from the quantum mechanics which replaced classical mechanics when dealing with Fig. 3: Principle of the measurement. structures as small as a ...
The Quantum Atom (section 18)
... Rutherford, Geiger and Marsden experiment (Muncaster p762) fired alpha particles at a thin gold foil. A few alpha particles are deflected through a large angle. Conclusion – there is a small, dense, positively charged nucleus (otherwise the electric field strength would never be strong enough to pro ...
... Rutherford, Geiger and Marsden experiment (Muncaster p762) fired alpha particles at a thin gold foil. A few alpha particles are deflected through a large angle. Conclusion – there is a small, dense, positively charged nucleus (otherwise the electric field strength would never be strong enough to pro ...
James Franck

James Franck (26 August 1882 – 21 May 1964) was a German physicist who won the 1925 Nobel Prize for Physics with Gustav Hertz ""for their discovery of the laws governing the impact of an electron upon an atom"". He completed his doctorate in 1906 and his habilitation in 1911 at the Frederick William University in Berlin, where he lectured and taught until 1918, having reached the position of professor extraordinarius. He served as a volunteer in the German Army during World War I. He was seriously injured in 1917 in a gas attack and was awarded the Iron Cross 1st Class.Franck became the Head of the Physics Division of the Kaiser Wilhelm Gesellschaft for Physical Chemistry. In 1920, Franck became professor ordinarius of experimental physics and Director of the Second Institute for Experimental Physics at the University of Göttingen. While there he worked on quantum physics with Max Born, who was Director of the Institute of Theoretical Physics. His work included the Franck–Hertz experiment, an important confirmation of the Bohr model of the atom. He promoted the careers of women in physics, notably Lise Meitner, Hertha Sponer and Hilde Levi.After the NSDAP came to power in Germany in 1933, Franck resigned his post in protest against the dismissal of fellow academics. He assisted Frederick Lindemann in helping dismissed Jewish scientists find work overseas, before he left Germany in November 1933. After a year at the Niels Bohr Institute in Denmark, he moved to the United States, where he worked at Johns Hopkins University in Baltimore and then the University of Chicago. During this period he became interested in photosynthesis.Franck participated in the Manhattan Project during World War II as Director of the Chemistry Division of the Metallurgical Laboratory. He was also the chairman of the Committee on Political and Social Problems regarding the atomic bomb, which is best known for the compilation of the Franck Report, which recommended that the atomic bombs not be used on the Japanese cities without warning.