AQA Science GCSE Chemistry
... AQA recognizes the importance of good-quality teaching, learning and assessment resources to accompany their specification. That's why they've chosen to work exclusively with nelson Thornes. With AQA examiners providing content and quality control, you can be confident that this course is as closely ...
... AQA recognizes the importance of good-quality teaching, learning and assessment resources to accompany their specification. That's why they've chosen to work exclusively with nelson Thornes. With AQA examiners providing content and quality control, you can be confident that this course is as closely ...
Chapter 4
... • The transition metals constitute Groups 3 through 12 and are sometimes called the d-block elements because of their position in the periodic table. • A transition metal is one of the metals that can use the inner shell before using the outer shell to bond. • A transition metal may lose one, two, o ...
... • The transition metals constitute Groups 3 through 12 and are sometimes called the d-block elements because of their position in the periodic table. • A transition metal is one of the metals that can use the inner shell before using the outer shell to bond. • A transition metal may lose one, two, o ...
Chapter 4 - My Chemistry Site
... 1. Which group of elements is the most unreactive?Why? The noble gases ( group 18)because they have a full valence level of electrons 2. Why do groups among the main-group elements display similar chemical behavior? They have the same number of valence electrons 3. What properties do the halogens h ...
... 1. Which group of elements is the most unreactive?Why? The noble gases ( group 18)because they have a full valence level of electrons 2. Why do groups among the main-group elements display similar chemical behavior? They have the same number of valence electrons 3. What properties do the halogens h ...
Class XI worksheet - Indian School Muscat
... Arrange the following elements S,P,O,N in the increasing order of non-metallic character ...
... Arrange the following elements S,P,O,N in the increasing order of non-metallic character ...
Chapter 4 Ppt - s3.amazonaws.com
... • The transition metals constitute Groups 3 through 12 and are sometimes called the d-block elements because of their position in the periodic table. • A transition metal is one of the metals that can use the inner shell before using the outer shell to bond. • A transition metal may lose one, two, o ...
... • The transition metals constitute Groups 3 through 12 and are sometimes called the d-block elements because of their position in the periodic table. • A transition metal is one of the metals that can use the inner shell before using the outer shell to bond. • A transition metal may lose one, two, o ...
Chapter 1 Introduction to Forensic Chemistry
... oxygen (often with applied heat); carbon-containing compounds then produce carbon dioxide and hydrogen-containing compounds then produce water as a result. Neutralization reactions occur when an acid and a base react to form a salt and water. Redox reactions occur when one substance gains electrons ...
... oxygen (often with applied heat); carbon-containing compounds then produce carbon dioxide and hydrogen-containing compounds then produce water as a result. Neutralization reactions occur when an acid and a base react to form a salt and water. Redox reactions occur when one substance gains electrons ...
Chapter 5 PRACTICE TEST
... The electron configuration of an element is [Kr] 4d6 5s1. To what group does this element belong? a. Group 4 c. Group 7 b. Group 5 d. Group 9 Magnesium, atomic number 12, has the electron configuration [Ne] 3s2. To what group does magnesium belong? a. Group 2 c. Group 5 b. Group 3 d. Group 12 The mo ...
... The electron configuration of an element is [Kr] 4d6 5s1. To what group does this element belong? a. Group 4 c. Group 7 b. Group 5 d. Group 9 Magnesium, atomic number 12, has the electron configuration [Ne] 3s2. To what group does magnesium belong? a. Group 2 c. Group 5 b. Group 3 d. Group 12 The mo ...
Std 10th, Science and Technology, Maharashtra Board, English
... All ______ of the same elements have different masses but same atomic number. ...
... All ______ of the same elements have different masses but same atomic number. ...
Science - ExamResults.net
... All ______ of the same elements have different masses but same atomic number. ...
... All ______ of the same elements have different masses but same atomic number. ...
Classification and Periodic Properties of Elements
... atomic weights was strictly followed. He ignored the order of atomic weights and placed the elements with similar chemical properties together. For example, iodine having atomic weight 127 was placed after tellurium (atomic weight 128), together with fluorine, chlorine and bromine due to similaritie ...
... atomic weights was strictly followed. He ignored the order of atomic weights and placed the elements with similar chemical properties together. For example, iodine having atomic weight 127 was placed after tellurium (atomic weight 128), together with fluorine, chlorine and bromine due to similaritie ...
classification of elements and periodicity in properties
... group 1 elements (hydrogen, alkali metals). Elements with two electrons in their outer shells have the configuration of ns2. They belong to group 2 elements (alkaline earth metals). Elements with three electrons (two in ‘s’ orbital and one in ‘p’ orbital) have the configuration of ns2 np2. They belo ...
... group 1 elements (hydrogen, alkali metals). Elements with two electrons in their outer shells have the configuration of ns2. They belong to group 2 elements (alkaline earth metals). Elements with three electrons (two in ‘s’ orbital and one in ‘p’ orbital) have the configuration of ns2 np2. They belo ...
General and Inorganic Chemistry I.
... History of the periodic table About 330 B.C Aristotle proposed that everything is made up of a mixture of one or more of four ”roots”. The four elements were earth, water, air and fire. In 1661 Boyle defined an element as a substance that cannot be broken down into a simpler substance by a chemical ...
... History of the periodic table About 330 B.C Aristotle proposed that everything is made up of a mixture of one or more of four ”roots”. The four elements were earth, water, air and fire. In 1661 Boyle defined an element as a substance that cannot be broken down into a simpler substance by a chemical ...
Periodic Classification of Elements
... In the last two lessons you have studied about the structure of atoms and their electronic configurations. You have also studied that elements with similar electronic configurations show similar chemical properties. Electrons are filled in various shells and subshells in a fairly regular fashion. Th ...
... In the last two lessons you have studied about the structure of atoms and their electronic configurations. You have also studied that elements with similar electronic configurations show similar chemical properties. Electrons are filled in various shells and subshells in a fairly regular fashion. Th ...
Lorna Merklinger
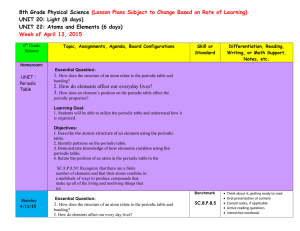
... 2. How do elements affect our everyday lives? 3. How does an element’s position on the periodic table affect the periodic properties? Learning Goal: 1. Students will be able to utilize the periodic table and understand how it is organized. Objectives: 1. Describe the atomic structure of an element u ...
... 2. How do elements affect our everyday lives? 3. How does an element’s position on the periodic table affect the periodic properties? Learning Goal: 1. Students will be able to utilize the periodic table and understand how it is organized. Objectives: 1. Describe the atomic structure of an element u ...
File
... The three types of hydrides are ionic, covalent, and metallic (or interstitial). The ionic and covalent hydrides are true compounds obeying the law of definite proportions and differ from each other in the type of bonding. The interstitial hydrides are more like solid solutions of hydrogen with a tr ...
... The three types of hydrides are ionic, covalent, and metallic (or interstitial). The ionic and covalent hydrides are true compounds obeying the law of definite proportions and differ from each other in the type of bonding. The interstitial hydrides are more like solid solutions of hydrogen with a tr ...
ch 3 classification of elements and periodic properties
... 1. in group 2 → filled ns subshells 2. in group 15 → half-filled np subshells 3. in group 18 → fully filled subshells These elec config are relatively stable & hence these have +ve or very low –ve EGEs. ...
... 1. in group 2 → filled ns subshells 2. in group 15 → half-filled np subshells 3. in group 18 → fully filled subshells These elec config are relatively stable & hence these have +ve or very low –ve EGEs. ...
Chapter 6: The Periodic Table and Periodic Law
... who is shown in Figure 6-2, proposed an organization scheme for the elements. Newlands noticed that when the elements were arranged by increasing atomic mass, their properties repeated every eighth element. In other words, the first and eighth elements had similar properties, the second and ninth el ...
... who is shown in Figure 6-2, proposed an organization scheme for the elements. Newlands noticed that when the elements were arranged by increasing atomic mass, their properties repeated every eighth element. In other words, the first and eighth elements had similar properties, the second and ninth el ...
Now
... ‘d block’ elements are called outer transition elements because they contain at most two electrons in their outer shell. The elements for which f sub shells are filling are called the inner transition elements. Based on electronic configuration, all elements are grouped into four categories. They ar ...
... ‘d block’ elements are called outer transition elements because they contain at most two electrons in their outer shell. The elements for which f sub shells are filling are called the inner transition elements. Based on electronic configuration, all elements are grouped into four categories. They ar ...
Chapter 6: The Periodic Table and Periodic Law
... shown in Figure 6.2, became widely accepted because he predicted the existence and properties of undiscovered elements that were later found. Mendeleev left blank spaces in the table where he thought the undiscovered elements should go. By noting trends in the properties of known elements, he was ab ...
... shown in Figure 6.2, became widely accepted because he predicted the existence and properties of undiscovered elements that were later found. Mendeleev left blank spaces in the table where he thought the undiscovered elements should go. By noting trends in the properties of known elements, he was ab ...
Periods and Blocks of the Periodic Table
... • The number of the highest occupied energy level is 5, so the element is in the fifth period. • There are five electrons in the d sublevel, which means that it is incompletely filled. The d sublevel can hold 10 electrons. Therefore, the element is in the d block. • For d-block elements, the number ...
... • The number of the highest occupied energy level is 5, so the element is in the fifth period. • There are five electrons in the d sublevel, which means that it is incompletely filled. The d sublevel can hold 10 electrons. Therefore, the element is in the d block. • For d-block elements, the number ...
Chemistry Part - teko classes bhopal
... compounds are low. Forces of attraction between these molecules of organic compounds are not very strong/ As these compound are largely non conductors of electricity hence the bonding in these compound does not give rise to any ions. The reactivity of elements if explained at their tendency to attai ...
... compounds are low. Forces of attraction between these molecules of organic compounds are not very strong/ As these compound are largely non conductors of electricity hence the bonding in these compound does not give rise to any ions. The reactivity of elements if explained at their tendency to attai ...
Concerning Electronegativity as a Basic Elemental Property and
... This paper is not about the epistemology of quantum mechanics. Indeed, the four quantum numbers and the associated four rules are held to be a priori. As Richard Feynman was quoted as saying on many occasions (Feynman 1965): “Nobody understands quantum mechanics”, in the sense that while we can obse ...
... This paper is not about the epistemology of quantum mechanics. Indeed, the four quantum numbers and the associated four rules are held to be a priori. As Richard Feynman was quoted as saying on many occasions (Feynman 1965): “Nobody understands quantum mechanics”, in the sense that while we can obse ...
The Periodic Table and Periodic Law
... and properties of undiscovered elements that were later found. Mendeleev left blank spaces in the table where he thought the undiscovered elements should go. By noting trends in the properties of known elements, he was able to predict the properties of the yet-to-be-discovered elements scandium, gal ...
... and properties of undiscovered elements that were later found. Mendeleev left blank spaces in the table where he thought the undiscovered elements should go. By noting trends in the properties of known elements, he was able to predict the properties of the yet-to-be-discovered elements scandium, gal ...