MSE 221 Quantum Physics of Materials
... Elementary quantum concepts, wave mechanics, energy states, bonding, transitions, electronic properties Prerequisite: PHYS 151 14 weeks, lectures (3 times a week), recitation (once a week) Introduction Old Quantum Theory, Bohr model, De Broglie, duality Interference and Diffraction—Young’s experimen ...
... Elementary quantum concepts, wave mechanics, energy states, bonding, transitions, electronic properties Prerequisite: PHYS 151 14 weeks, lectures (3 times a week), recitation (once a week) Introduction Old Quantum Theory, Bohr model, De Broglie, duality Interference and Diffraction—Young’s experimen ...
Concepts of Modern Physics Presentations
... You will choose a topic in modern physics from the list below. Each topic is a basic concept or idea in modern physics (related to quantum mechanics, general relativity, particle physics, or theories of everything) You will be allotted 10 minutes to present a summary of your topic, highlighting the ...
... You will choose a topic in modern physics from the list below. Each topic is a basic concept or idea in modern physics (related to quantum mechanics, general relativity, particle physics, or theories of everything) You will be allotted 10 minutes to present a summary of your topic, highlighting the ...
Spherical harmonics: • The quantum numbers n, l, m determine the
... have zeros for several values. These result in nodal surfaces where the probability of finding the electron vanishes. This is because bound particles are standing waves! ...
... have zeros for several values. These result in nodal surfaces where the probability of finding the electron vanishes. This is because bound particles are standing waves! ...
Quantum Mechanics
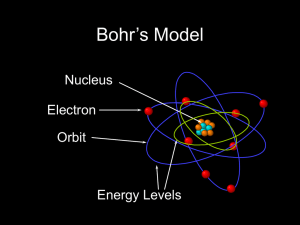
... model of the atom The Bohr model of the atom explains the equations for the atomic spectrum of hydrogen, but does not work for any other element Even though his theory was wrong, it provided an excellent starting point for quantum mechanics ...
... model of the atom The Bohr model of the atom explains the equations for the atomic spectrum of hydrogen, but does not work for any other element Even though his theory was wrong, it provided an excellent starting point for quantum mechanics ...
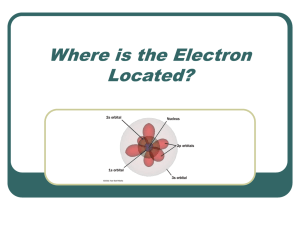
Complete nomenclature for electron orbitals
... l The previous picture is still a little too classical, though l It pictures the electrons as orbiting the nucleus in circular (or elliptical orbitals) l But in fact the only reality is |y|2, the square of the wavefunction, which gives the probability of the electron to be in a given place at a give ...
... l The previous picture is still a little too classical, though l It pictures the electrons as orbiting the nucleus in circular (or elliptical orbitals) l But in fact the only reality is |y|2, the square of the wavefunction, which gives the probability of the electron to be in a given place at a give ...
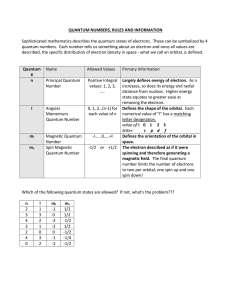
Quantum Number Table
... Sophisticated mathematics describes the quantum states of electrons. These can be symbolized by 4 quantum numbers. Each number tells us something about an electron and once all values are described, the specific distribution of electron density in space - what we call an orbital, is defined. ...
... Sophisticated mathematics describes the quantum states of electrons. These can be symbolized by 4 quantum numbers. Each number tells us something about an electron and once all values are described, the specific distribution of electron density in space - what we call an orbital, is defined. ...
The Quantum Mechanical Behavior of Light and Matter
... classical: matter behaves like particles, light behaves like waves quantum mechanics: both matter and light behave like both particles and waves ...
... classical: matter behaves like particles, light behaves like waves quantum mechanics: both matter and light behave like both particles and waves ...
File
... the photoelectric effect in terms of discretized electromagnetic radiation. The second formed the foundation for modern statistical physics by explaining the seemingly-random motion of particles in a fluid, a behavior called Brownian motion. The third reconciled Maxwellian electrodynamics with class ...
... the photoelectric effect in terms of discretized electromagnetic radiation. The second formed the foundation for modern statistical physics by explaining the seemingly-random motion of particles in a fluid, a behavior called Brownian motion. The third reconciled Maxwellian electrodynamics with class ...
Two-electron Interference
... possible. For a single particle, the interference is between the amplitudes of the particle’s wave function, whereas the interference between two particles is a direct result of quantum exchange statistics. Such interference is observed only in the joint probability of finding the particles in tw ...
... possible. For a single particle, the interference is between the amplitudes of the particle’s wave function, whereas the interference between two particles is a direct result of quantum exchange statistics. Such interference is observed only in the joint probability of finding the particles in tw ...
Quantum Numbers Practice Problems Name: AP Physics Period: 1
... Quantum Numbers Practice Problems AP Physics ...
... Quantum Numbers Practice Problems AP Physics ...
File - SPHS Devil Physics
... a. The electron microscope and the tunnelling electron microscope rely on the findings from studies in quantum physics b. Probability is treated in a mathematical sense in Mathematical studies SL sub-topics 3.6–3.7 9. Aims: a. Aim 1: study of quantum phenomena introduces students to an exciting new ...
... a. The electron microscope and the tunnelling electron microscope rely on the findings from studies in quantum physics b. Probability is treated in a mathematical sense in Mathematical studies SL sub-topics 3.6–3.7 9. Aims: a. Aim 1: study of quantum phenomena introduces students to an exciting new ...
Quantum electrodynamics

In particle physics, quantum electrodynamics (QED) is the relativistic quantum field theory of electrodynamics. In essence, it describes how light and matter interact and is the first theory where full agreement between quantum mechanics and special relativity is achieved. QED mathematically describes all phenomena involving electrically charged particles interacting by means of exchange of photons and represents the quantum counterpart of classical electromagnetism giving a complete account of matter and light interaction.In technical terms, QED can be described as a perturbation theory of the electromagnetic quantum vacuum. Richard Feynman called it ""the jewel of physics"" for its extremely accurate predictions of quantities like the anomalous magnetic moment of the electron and the Lamb shift of the energy levels of hydrogen.