* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download The Quantum Mechanical Behavior of Light and Matter
Interpretations of quantum mechanics wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Elementary particle wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Probability amplitude wikipedia , lookup
Copenhagen interpretation wikipedia , lookup
Delayed choice quantum eraser wikipedia , lookup
History of quantum field theory wikipedia , lookup
Renormalization wikipedia , lookup
Quantum teleportation wikipedia , lookup
Quantum state wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Quantum key distribution wikipedia , lookup
EPR paradox wikipedia , lookup
Canonical quantization wikipedia , lookup
Hidden variable theory wikipedia , lookup
Atomic orbital wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Particle in a box wikipedia , lookup
Electron scattering wikipedia , lookup
Electron configuration wikipedia , lookup
Double-slit experiment wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Matter wave wikipedia , lookup
Wave–particle duality wikipedia , lookup
Hydrogen atom wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
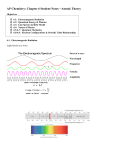
2 1/r Laws gravity, electric force, light let’s look at a radiating source same amount of energy/time, or luminosity (L) crosses successive spheres at larger r energy/area/time (energy flux) at distance r = L/4πr or, luminosity/surface area standard candles --> distances 2 Lecture 3: Quantum Mechanical Behavior of Light and Matter wave-particle duality (eg, photoelectric effect vs. refraction) classical: matter behaves like particles, light behaves like waves quantum mechanics: both matter and light behave like both particles and waves any formula with Planck’s constant h = 6.63 × 10−27 gm cm2 s−1 cannot be derived from classical mechanics Quantum Mechanical Behavior (cont.) Photoelectric Effect (Einstein 1905) light comes in discrete packets (photons) of energy E related to wavelength, not intensity = hc λ Quantum Mechanical Behavior (cont.) shorter wavelength photons have more energy, momentum --> generate faster moving electrons higher intensity light generates more electrons, not higher velocity electrons = p = h/λ hc λ Quantum Mechanical Behavior of Light Heisenberg Uncertainty Principle position, momentum of free particle cannot be measured to arbitrary accuracy at same time (∆x)(∆px ) > h on microscopic scales, nature is fuzzy Quantum Mechanical Behavior of Matter p = h/λ de Broglie (thesis!) every particle with momentum p has associated probability wave solutions to “particle in a box” problem Apply to Hydrogen Atom (Bohr’s Model) size of electron’s orbit not arbitrary, must be integral number of wavelengths 2πr = nλe where λe = h/pe = h/me v http://www.walter-fendt.de/ph11e/bohrh.htm What are the allowable values of r? what is v? 2 F = ma = e /r 2 2 and a = v /r for circular motion therefore, r = n2 (h̄/me e2 ) where h̄ = h/2π, n ≡ principal quantum number , h̄/me e2 ≡ Bohr radius smallest value of r obtained from n=1 (ground state of atomic hydrogen) for circular orbits n = 2 has radius that is four times larger than ground state (even though we really mean fuzzy probability shells) even in ground state, hydrogen atom ~ 105 bigger than its −13 nucleus (mostly empty space) 10 electrons cannot spiral inward (in discreet shells) huge electric forces support matter against gravity! we really mean fuzzy probability shells and there are other quantum numbers http://www.falstad.com/qmatom/ Spectrum of Hydrogen Atom allowable values for orbital energy of electron E = KE + electric potential energy 2 2 = me v /2 + (−e /r) must be quantized! using Bohr radius 2 4 2 E = −n (me e /2h̄ ) E < 0 for bound states, E--> 0 as n--> ∞ , ionization limit consider hydrogen atom at n’=5 electron jumps spontaneously to n=3 energy released (photon) hc = E n! − E n E= λ photon energy fixed, characteristic Spectrum of Hydrogen Atom (cont.) in addition to discrete line transitions, H atom can scatter radiation (propagation direction changes, but E, wavelength unaffected) make transitions to, from continuum at n= 1, λ = 912 Å (Lyman limit) required for ionization photons with λ > 912 Å absorbed only at discrete wavelengths at other wavelengths, scattered (cause of reflection and refraction)