BC1 Atoms Unit Standards
... 2c Identify the number of neutrons in an atom from atomic number and mass number 2d Identify the number of electrons in an atom when given the number of protons and charge 2e Identify the charge of an ion from the number of protons and electrons 2f Identify the mass of an atom from the number of pro ...
... 2c Identify the number of neutrons in an atom from atomic number and mass number 2d Identify the number of electrons in an atom when given the number of protons and charge 2e Identify the charge of an ion from the number of protons and electrons 2f Identify the mass of an atom from the number of pro ...
The study of biology can help you better understand human
... Which subatomic particle determines chemical properties of an element? ...
... Which subatomic particle determines chemical properties of an element? ...
C2- Topic 1: Atomic structure and the periodic table. Assessable
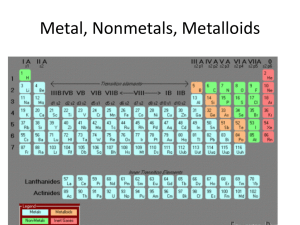
... Explain how Mendeleev: - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
... Explain how Mendeleev: - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
Section 1 Review
... or broken down into simpler substances by chemical means isotope one of two or more atoms that have the same number of protons (atomic number) but different numbers of neutrons (atomic mass) ...
... or broken down into simpler substances by chemical means isotope one of two or more atoms that have the same number of protons (atomic number) but different numbers of neutrons (atomic mass) ...
Atomic Structure and the Periodic Table Vocabulary
... __________________________________________________________________ 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __ ...
... __________________________________________________________________ 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __ ...
1 - cloudfront.net
... How is the number of neutrons in the nucleus of an atom calculated? All atoms are neutral, with the number of protons equaling the ___. Isotopes of the same element have different _____. Using the periodic table, determine the number of neutrons in 16O. What does the number 84 represent in the name ...
... How is the number of neutrons in the nucleus of an atom calculated? All atoms are neutral, with the number of protons equaling the ___. Isotopes of the same element have different _____. Using the periodic table, determine the number of neutrons in 16O. What does the number 84 represent in the name ...
atomic structure - IGCSE STUDY BANK
... It is the proton number that determines the specific identity of a particular element and its electron structure. The mass number (A) is also known as the nucleon number (N), that is the number of particles in the nucleus of a particular isotope. Protons and neutrons are the nucleons present in the ...
... It is the proton number that determines the specific identity of a particular element and its electron structure. The mass number (A) is also known as the nucleon number (N), that is the number of particles in the nucleus of a particular isotope. Protons and neutrons are the nucleons present in the ...
atoms - Trinity Regional School
... John Dalton (aka Jack) 1. Atoms are the smallest form of matter 2. Atoms make up all matter 3. Atoms of the same element are the same 4. Atoms cannot be broken down 5. Atoms combine in specific ratios ...
... John Dalton (aka Jack) 1. Atoms are the smallest form of matter 2. Atoms make up all matter 3. Atoms of the same element are the same 4. Atoms cannot be broken down 5. Atoms combine in specific ratios ...
IPC Atoms and Periodic Table
... of the naturally occurring isotopes of an element • Reported as atomic mass on the periodic ...
... of the naturally occurring isotopes of an element • Reported as atomic mass on the periodic ...
Pre-Knowledge: Chemistry and Physics Vocabulary Atomic Number
... become more stable by emitting radiation. Radioactive decay is the process in which an unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. This radiation can be emitted in the form of a positively charged alpha particle, a negatively charged ...
... become more stable by emitting radiation. Radioactive decay is the process in which an unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. This radiation can be emitted in the form of a positively charged alpha particle, a negatively charged ...
The New Alchemy
... Protons – one of the parts of an atom. Protons have a (+) charge and are found in the nucleus. Neutrons – one of the parts of an atom. Neutrons have no charge and are found in the nucleus. Nucleus – found in the center of an atom. It contains protons and neutrons. Nuclei is the plural of nucleus. Nu ...
... Protons – one of the parts of an atom. Protons have a (+) charge and are found in the nucleus. Neutrons – one of the parts of an atom. Neutrons have no charge and are found in the nucleus. Nucleus – found in the center of an atom. It contains protons and neutrons. Nuclei is the plural of nucleus. Nu ...
Chapter 6 Review“The Periodic Table”
... 15. About how many more times massive is a proton than electron? 16. How many neutrons are present in an isotope of uranium-235? 17. What is the atomic number for an element with 41 neutrons and a mass number of 80? 18. What is the maximum number of electrons in the second principal energy level? 19 ...
... 15. About how many more times massive is a proton than electron? 16. How many neutrons are present in an isotope of uranium-235? 17. What is the atomic number for an element with 41 neutrons and a mass number of 80? 18. What is the maximum number of electrons in the second principal energy level? 19 ...
Physical Science Chapter 6 Study Guide Every element consists of
... o Every element consists of tiny particles called ____________ o All atoms of a particular element have the same ____________________ o Different elements have different properties because their atoms are different o Atoms of different elements can combine in specific ways to form ____________ o Che ...
... o Every element consists of tiny particles called ____________ o All atoms of a particular element have the same ____________________ o Different elements have different properties because their atoms are different o Atoms of different elements can combine in specific ways to form ____________ o Che ...
Chapter 3 – Atomic Structure - Mercer Island School District
... • Each element is composed of tiny atoms • Atoms of an element are identical but differ from those of other elements. • Atoms are neither created nor destroyed. • A given compound always has the same relative numbers and kinds of atoms. ...
... • Each element is composed of tiny atoms • Atoms of an element are identical but differ from those of other elements. • Atoms are neither created nor destroyed. • A given compound always has the same relative numbers and kinds of atoms. ...
Atomic Theory- 1. Matter is composed of tiny, indivisible particles
... called atoms. 2. An element is composed of one type of atom. Properties of atoms are identical to each other. 3. A compound contains two or more different elements. The relative number of atoms of each element in a compound is the same. 4. Atoms don’t change their identities in chemical reactions.-j ...
... called atoms. 2. An element is composed of one type of atom. Properties of atoms are identical to each other. 3. A compound contains two or more different elements. The relative number of atoms of each element in a compound is the same. 4. Atoms don’t change their identities in chemical reactions.-j ...
Physical Science Chapter 6 Study Guide Atomic Theory of Matter
... Nuclear reactor—a type of controlled reaction used to harness useful energy ...
... Nuclear reactor—a type of controlled reaction used to harness useful energy ...
atomic number - Thomas C. Cario Middle School
... The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of atom. The atomic number of an atom is equal to the number of protons in its nucleus. The number of electrons surrounding the nucleus of an atom is equal ...
... The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of atom. The atomic number of an atom is equal to the number of protons in its nucleus. The number of electrons surrounding the nucleus of an atom is equal ...

![Atomic Structure [PowerPoint]](http://s1.studyres.com/store/data/000122096_1-1d100da6540d2f26db122fc51f672fe5-300x300.png)