Dephosphorylation of the Calcium Pump Coupled to Counterion
... supported by the fact that alkaline pH reduces the rate of dephosphorylation. The transition state (E2-P) has a low affinity to Ca2+, which contrasts with E2. Lumenal Ca 2+ is electrostatically repelled by the ...
... supported by the fact that alkaline pH reduces the rate of dephosphorylation. The transition state (E2-P) has a low affinity to Ca2+, which contrasts with E2. Lumenal Ca 2+ is electrostatically repelled by the ...
Javan Kilango Kisaka
... nutrient that Plasmodium falciparum needs in order to colonize its host. Its cellular levels are carefully regulated by a copper efflux protein (PfCuP-ATPase) to reduce its toxicity. PfCuP-ATPase is the largest copper transporter of the PIB P-type ATPase family made up of 2563 amino acid residues wi ...
... nutrient that Plasmodium falciparum needs in order to colonize its host. Its cellular levels are carefully regulated by a copper efflux protein (PfCuP-ATPase) to reduce its toxicity. PfCuP-ATPase is the largest copper transporter of the PIB P-type ATPase family made up of 2563 amino acid residues wi ...
MD simulering
... Model the dynamics of domain motion in large proteins Incorporate experimental data such as point mutation results in a protein domain model ...
... Model the dynamics of domain motion in large proteins Incorporate experimental data such as point mutation results in a protein domain model ...
Membrane Transport
... Fig. 1. (A) ABC transporter MetNI consists of four subunits: two membrane-spanning MetI subunits (green and pink) and two MetN ABC subunits (purple and tan) Structure B is A in a 90o Turn TMD – transmembrane domain, NBD – Nucleotide binding domain, C-2 Methionine binding domain ...
... Fig. 1. (A) ABC transporter MetNI consists of four subunits: two membrane-spanning MetI subunits (green and pink) and two MetN ABC subunits (purple and tan) Structure B is A in a 90o Turn TMD – transmembrane domain, NBD – Nucleotide binding domain, C-2 Methionine binding domain ...
Slide ()
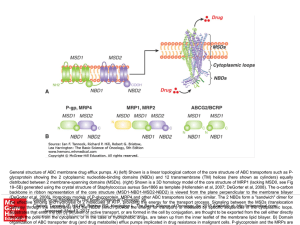
... General structure of ABC membrane drug efflux pumps. A) (left) Shown is a linear topological cartoon of the core structure of ABC transporters such as Pglycoprotein showing the 2 cytoplasmic nucleotide-binding domains (NBDs) and 12 transmembrane (TM) helices (here shown as cylinders) equally distrib ...
... General structure of ABC membrane drug efflux pumps. A) (left) Shown is a linear topological cartoon of the core structure of ABC transporters such as Pglycoprotein showing the 2 cytoplasmic nucleotide-binding domains (NBDs) and 12 transmembrane (TM) helices (here shown as cylinders) equally distrib ...
Presentation Abstract - UMBC Chemistry and Biochemistry
... The P1B-ATPases, which couple cation transport across membranes to ATP hydrolysis, are central to metal homeostasis in all organisms. Prior studies have focused chiefly on the involvement of these enzymes in copper and zinc transport. However, recent bioinformatic data reveal that these enzymes may ...
... The P1B-ATPases, which couple cation transport across membranes to ATP hydrolysis, are central to metal homeostasis in all organisms. Prior studies have focused chiefly on the involvement of these enzymes in copper and zinc transport. However, recent bioinformatic data reveal that these enzymes may ...
ATP-driven Pumps
... In addition there is a change in binding site affinity: allows ion pick-up at low concn on one side allows ion release at high concn on other side These are the two ways in which free energy from ATP hydrolysis is expended. ...
... In addition there is a change in binding site affinity: allows ion pick-up at low concn on one side allows ion release at high concn on other side These are the two ways in which free energy from ATP hydrolysis is expended. ...
Gail`s powerpoint
... • Na pump does not need to be active in order to reach PM • All 3 glycosylation Asn replaced with Glu on b-subunit – Proper assembly and trafficking to PM with wild-type a-subunit – Catalytically active, but increased susceptibility to degradation ...
... • Na pump does not need to be active in order to reach PM • All 3 glycosylation Asn replaced with Glu on b-subunit – Proper assembly and trafficking to PM with wild-type a-subunit – Catalytically active, but increased susceptibility to degradation ...
P-type ATPase

The P-type ATPases, also known as E1-E2 ATPases, are a large group of evolutionarily related ion and lipid pumps that are found in bacteria, archaea, and eukaryotes. They are α-helical bundle primary transporters referred to as P-type ATPases because they catalyze auto- (or self-) phosphorylation of a key conserved aspartate residue within the pump. In addition, they all appear to interconvert between at least two different conformations, denoted by E1 and E2.Most members of this transporter family are specific for the pumping of a large array of cations, however one subfamily is involved in flipping phospholipids to maintain the asymmetric nature of the biomembrane.Prominent examples of P-type ATPases are the sodium-potassium pump (Na+,K+-ATPase), the plasma membrane proton pump (H+-ATPase), the proton-potassium pump (H+,K+-ATPase), and the calcium pump (Ca2+-ATPase).