Lithium Iodide Original Commentary - Groupe Charette
... LiI as an Additive for Organometallic-mediated Transformations. Diastereoselectivity in the cyclization of 5-hexenyllithiums was shown to be influenced by LiI.34 As an additive in the reduction of α,β-unsaturated ketones by Bu2 SnH2 /Bu2 SnF2 , LiI has a dramatic effect on the selectivity for 1,2- ve ...
... LiI as an Additive for Organometallic-mediated Transformations. Diastereoselectivity in the cyclization of 5-hexenyllithiums was shown to be influenced by LiI.34 As an additive in the reduction of α,β-unsaturated ketones by Bu2 SnH2 /Bu2 SnF2 , LiI has a dramatic effect on the selectivity for 1,2- ve ...
Purification and characterization of a novel caffeine oxidase from
... beverages and wastewaters because of potential chronic ingestion of caffeine and/or its byproduct 8chlorocaffeine (produced during chlorination) can have adverse effects on the physiological systems (Gould and Hay, 1982; White and Rasmussen, 1998). Decaffeination is usually performed by physico-chem ...
... beverages and wastewaters because of potential chronic ingestion of caffeine and/or its byproduct 8chlorocaffeine (produced during chlorination) can have adverse effects on the physiological systems (Gould and Hay, 1982; White and Rasmussen, 1998). Decaffeination is usually performed by physico-chem ...
1H-Imidazol-4(5H)-ones and thiazol-4(5H)
... enolate or equivalent is fixed due to their cyclic nature, thus facilitating the control of the stereoselectivity; iii) they are substituted at the α-position of the carbonyl and therefore, after reaction with an electrophile, a tetrasubstituted stereocenter is created and, iv) the resulting adducts ...
... enolate or equivalent is fixed due to their cyclic nature, thus facilitating the control of the stereoselectivity; iii) they are substituted at the α-position of the carbonyl and therefore, after reaction with an electrophile, a tetrasubstituted stereocenter is created and, iv) the resulting adducts ...
Proofs to - Research Explorer
... monosubstituted vinylidene [Fe(C=CHPh)(dppe)Cp][PF6] with refluxing MeOH have been unsuccessful although mass spectrometry of the isolated product revealed a weak peak with m/z = 653, consistent with formation of the known complex [Fe{C(OMe)Ph}(dppe)Cp]+, [28]. An alternative strategy to facilitate ...
... monosubstituted vinylidene [Fe(C=CHPh)(dppe)Cp][PF6] with refluxing MeOH have been unsuccessful although mass spectrometry of the isolated product revealed a weak peak with m/z = 653, consistent with formation of the known complex [Fe{C(OMe)Ph}(dppe)Cp]+, [28]. An alternative strategy to facilitate ...
Efficient hydrogenation of organic carbonates, carbamates and
... complexes 1–4 as catalysts. Reaction of methyl formate and dihydrogen (7 atm) catalysed by 1 (0.1 mol%) at 145 8C in 1,4dioxane yielded 71% of methanol after 30 h (Table 3, entry 1). Performing the reaction under 9 atm of H2 resulted in complete conversion of methyl formate to methanol after 36 h wi ...
... complexes 1–4 as catalysts. Reaction of methyl formate and dihydrogen (7 atm) catalysed by 1 (0.1 mol%) at 145 8C in 1,4dioxane yielded 71% of methanol after 30 h (Table 3, entry 1). Performing the reaction under 9 atm of H2 resulted in complete conversion of methyl formate to methanol after 36 h wi ...
Polimeri - Polymer Technology Group
... made from crude oil, a nonrenewable raw material. For example, nylon 6,6 is prepared industrially from adipic acid and 1,6-diaminohexane, both of which originate from benzene, a product of petroleum refining. Figure 30.8 Synthesis of adipic acid and ...
... made from crude oil, a nonrenewable raw material. For example, nylon 6,6 is prepared industrially from adipic acid and 1,6-diaminohexane, both of which originate from benzene, a product of petroleum refining. Figure 30.8 Synthesis of adipic acid and ...
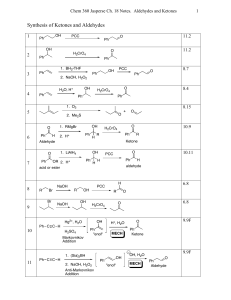
Synthesis of Ketones and Aldehydes
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
Synthesis of Ketones and Aldehydes
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
Derivatization reactions for the determination of amines by gas
... inherent problems related to the difficulty in handling low-molecular-mass amines because of their high water solubility and high volatility. Therefore, these amines are difficult to extract from water and are not easily chromatographed owing to their polarity. Furthermore, amines tend to be adsorbe ...
... inherent problems related to the difficulty in handling low-molecular-mass amines because of their high water solubility and high volatility. Therefore, these amines are difficult to extract from water and are not easily chromatographed owing to their polarity. Furthermore, amines tend to be adsorbe ...
Class Notes
... 1. After reaction, the original carbonyl carbon will have one and only one C-O single bond 2. For formaldehyde, aldehydes, and ketones, one R group adds (reactions 4-6) 3. For esters or carbonyl chlorides (“acid chlorides”), two R groups add o Replace not only the carbonyl p-bond, but also the “extr ...
... 1. After reaction, the original carbonyl carbon will have one and only one C-O single bond 2. For formaldehyde, aldehydes, and ketones, one R group adds (reactions 4-6) 3. For esters or carbonyl chlorides (“acid chlorides”), two R groups add o Replace not only the carbonyl p-bond, but also the “extr ...
aa-2005-38-71-negishi - University of Windsor
... electrophiles lacking proximal π bonds had been considered to be categorically very difficult, and the task of Pd-catalyzed alkylation had been achieved by using alkylmetals. The latter is still of much broader synthetic applicability. However, some recent developments suggest that this generalizati ...
... electrophiles lacking proximal π bonds had been considered to be categorically very difficult, and the task of Pd-catalyzed alkylation had been achieved by using alkylmetals. The latter is still of much broader synthetic applicability. However, some recent developments suggest that this generalizati ...
Document
... oxygen atom of nitroarene radical anions led to an indole synthesis now named “Bartoli indole synthesis” in many textbooks monographs and papers. This de novo construction of the indole ring is now one of the most widely used syntheses of 7-substituted indoles. A laboratory accident moved Prof. Bart ...
... oxygen atom of nitroarene radical anions led to an indole synthesis now named “Bartoli indole synthesis” in many textbooks monographs and papers. This de novo construction of the indole ring is now one of the most widely used syntheses of 7-substituted indoles. A laboratory accident moved Prof. Bart ...
[Ru(Triphos)H2(CO)] Characterisation - Durham e
... Phosphines are two-electron a-donor, neutral L-type ligands that are able to act as 71acceptor ligands. The bond formed between the metal and the phosphine from the adonation involves the overlap of orbitals that are located directly between them, with two electrons formally donated by the phosphine ...
... Phosphines are two-electron a-donor, neutral L-type ligands that are able to act as 71acceptor ligands. The bond formed between the metal and the phosphine from the adonation involves the overlap of orbitals that are located directly between them, with two electrons formally donated by the phosphine ...
Investigation of the steric and electronic properties of 3
... Activated dienes (such as conjugated 1,3-dienes and allenes) and alkynes are more readily hydroaminated than alkenes and unactivated dienes, which is reflected in the number of precatalysts published for the catalysis of each of these respective transformations. At first glance, the greater electron ...
... Activated dienes (such as conjugated 1,3-dienes and allenes) and alkynes are more readily hydroaminated than alkenes and unactivated dienes, which is reflected in the number of precatalysts published for the catalysis of each of these respective transformations. At first glance, the greater electron ...
endoglucanase in cellulose biosynthesis is not very clear
... Cellulose biosynthesis in Acetobacter xylinum. The bacterium A. xylinum produces pure cellulose. A single cell may polymerize up to 200,000 glucose residues per second into β-1,4-glucan chains. Advantages of using a bacterial system for production of cellulose is that the bacterium grows rapidly und ...
... Cellulose biosynthesis in Acetobacter xylinum. The bacterium A. xylinum produces pure cellulose. A single cell may polymerize up to 200,000 glucose residues per second into β-1,4-glucan chains. Advantages of using a bacterial system for production of cellulose is that the bacterium grows rapidly und ...
Lectins from Edible Mushrooms
... E. Boa (2004) on wild edible fungi [19], we have identified about 144 lectins from edible mushrooms, 38 lectins from reported poisonous mushrooms, and 30 lectins from mushrooms which can be used as medicine and/or as food. Along with this, we try to highlight the available data from the literature p ...
... E. Boa (2004) on wild edible fungi [19], we have identified about 144 lectins from edible mushrooms, 38 lectins from reported poisonous mushrooms, and 30 lectins from mushrooms which can be used as medicine and/or as food. Along with this, we try to highlight the available data from the literature p ...
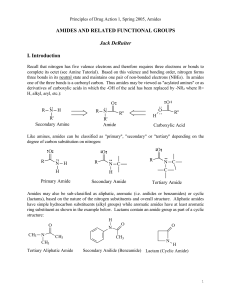
AMIDES AND RELATED FUNCTIONAL GROUPS
... While hydrogen bonding may enhance the water solubility of amides relative to hydrocarbons (alkanes, alkenes, alkynes and aromatic compounds), amides typically are regarded as compounds with low water solubility. They are significantly less water soluble than comparable acids or alcohols due to: 1). ...
... While hydrogen bonding may enhance the water solubility of amides relative to hydrocarbons (alkanes, alkenes, alkynes and aromatic compounds), amides typically are regarded as compounds with low water solubility. They are significantly less water soluble than comparable acids or alcohols due to: 1). ...
THE FRIEDEL-CRAFTS BENZYLATION OF ARENES AND THE
... opportunity to work in his research group. After the group arrived from UPenn, I was one of his first new PhD students at ETH and I feel really fortunate that I could join the group during this exciting time. Jeff, I want to thank you for all your support during the last 4 years, whether it was with ...
... opportunity to work in his research group. After the group arrived from UPenn, I was one of his first new PhD students at ETH and I feel really fortunate that I could join the group during this exciting time. Jeff, I want to thank you for all your support during the last 4 years, whether it was with ...
NAME Chem 204 Spring, 1990 Final Exam FINAL EXAM: Chaps. 1
... one of one reaction and one of two reactions) for converting a chiral alcohol into its inverted alkyl chloride, there is NO one reaction that will convert a chiral alcohol into its alkyl chloride with "retained" configuration. Thus, it is necessary to use several reaction steps, each of 100% inversi ...
... one of one reaction and one of two reactions) for converting a chiral alcohol into its inverted alkyl chloride, there is NO one reaction that will convert a chiral alcohol into its alkyl chloride with "retained" configuration. Thus, it is necessary to use several reaction steps, each of 100% inversi ...
Bottromycin

Bottromycin is a macrocyclic peptide with antibiotic activity. It was first discovered in 1957 as a natural product isolated from Streptomyces bottropensis. It has been shown to inhibit methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococci (VRE) among other Gram-positive bacteria and mycoplasma. Bottromycin is structurally distinct from both vancomycin, a glycopeptide antibiotic, and methicillin, a beta-lactam antibiotic.Bottromycin binds to the A site of the ribosome and blocks the binding of aminoacyl-tRNA, therefore inhibiting bacterial protein synthesis. Although bottromycin exhibits antibacterial activity in vitro, it has not yet been developed as a clinical antibiotic, potentially due to its poor stability in blood plasma. To increase its stability in vivo, some bottromycin derivatives have been explored.The structure of bottromycin contains a macrocyclic amidine as well as a thiazole ring. The absolute stereochemistry at several chiral centers has been determined as of 2009. In 2012, a three-dimensional solution structure of bottromycin was published. The solution structure revealed that several methyl groups are on the same face of the structure.Bottromycin falls within the ribosomally synthesized and post-translationally modified peptide class of natural product.











![[Ru(Triphos)H2(CO)] Characterisation - Durham e](http://s1.studyres.com/store/data/017676948_1-4352644236c53cc416f065328f560d26-300x300.png)